Abstract
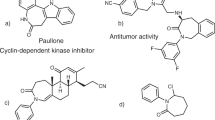
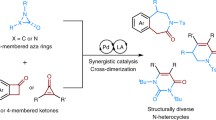
The synthesis of functionalized nitrogen heterocycles is integral to discovering, manufacturing and evolving high-value materials. The availability of effective strategies for heterocycle synthesis often biases the frequency of specific ring systems over others in the core structures of bioactive leads. For example, while the six- and five-membered piperidine and pyrrolidine are widespread in medicinal chemistry libraries, the seven-membered azepane is essentially absent and this leaves open a substantial area of three-dimensional chemical space. Here we report a strategy to prepare complex azepanes from simple nitroarenes by photochemical dearomative ring expansion centred on the conversion of the nitro group into a singlet nitrene. This process is mediated by blue light, occurs at room temperature and transforms the six-membered benzenoid framework into a seven-membered ring system. A following hydrogenolysis provides the azepanes in just two steps. We have demonstrated the utility of the strategy with the synthesis of several azepane analogues of piperidine drugs.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and the Supplementary Information. Computational data can be found through Figshare: https://doi.org/10.6084/m9.figshare.21432345.
References
Pennington, L. D. & Moustakas, D. T. The necessary nitrogen atom: a versatile high-impact design element for multiparameter optimization. J. Med. Chem. 60, 3552–3579 (2017).
Vitaku, E., Smith, D. T. & Njardarson, J. T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. J. Med. Chem. 57, 10257–10274 (2014).
Taylor, R. D., MacCoss, M. & Lawson, A. D. G. Rings in drugs. J. Med. Chem. 57, 5845–5859 (2014).
Shearer, J., Castro, J. L., Lawson, A. D. G., MacCoss, M. & Taylor, R. D. Rings in clinical trials and drugs: present and future. J. Med. Chem. 65, 8699–8712 (2022).
Aldeghi, M., Malhotra, S., Selwood, D. L. & Chan, A. W. E. Two- and three-dimensional rings in drugs. Chem. Biol. Drug Des. 83, 450–461 (2014).
Lovering, F., Bikker, J. & Humblet, C. Escape from flatland: increasing saturation as an approach to improving clinical success. J. Med. Chem. 52, 6752–6756 (2009).
Campos, K. R. et al. The importance of synthetic chemistry in the pharmaceutical industry. Science 363, eaat0805 (2019).
Blakemore, D. C. et al. Organic synthesis provides opportunities to transform drug discovery. Nat. Chem. 10, 383–394 (2018).
Jurczyk, J. et al. Single-atom logic for heterocycle editing. Nat. Synth. 1, 352–364 (2022).
Weintraub, P. M., Sabol, J. S., Kane, J. M. & Borcherding, D. R. Recent advances in the synthesis of piperidones and piperidines. Tetrahedron 59, 2953–2989 (2003).
Chiacchio, M. A., Legnani, L., Chiacchio, U. & Iannazzo, D. in More Synthetic Approaches to Nonaromatic Nitrogen Heterocycles (ed. Faisca Phillips, A. M.) 529–558 (2022).
Glorius, F. Asymmetric hydrogenation of aromatic compounds. Org. Biomol. Chem. 3, 4171–4175 (2005).
Mainolfi, N. et al. Discovery of 4-((2S,4S)-4-ethoxy-1-((5-methoxy-7-methyl-1H-indol-4-yl)methyl)piperidin-2-yl)benzoic acid (LNP023), a factor B inhibitor specifically designed to be applicable to treating a diverse array of complement mediated diseases. J. Med. Chem. 63, 5697–5722 (2020).
Zhou, Y.-G. Asymmetric hydrogenation of heteroaromatic compounds. Acc. Chem. Res. 40, 1357–1366 (2007).
Wang, D.-S., Chen, Q.-A., Lu, S.-M. & Zhou, Y.-G. Asymmetric hydrogenation of heteroarenes and arenes. Chem. Rev. 112, 2557–2590 (2012).
Huck, C. J. & Sarlah, D. Shaping molecular landscapes: recent advances, opportunities, and challenges in dearomatization. Chem 6, 1589–1603 (2020).
Gritsan, N. P. & Platz, M. S. Kinetics, spectroscopy, and computational chemistry of arylnitrenes. Chem. Rev. 106, 3844–3867 (2006).
Borden, W. T. et al. The interplay of theory and experiment in the study of phenylnitrene. Acc. Chem. Res. 33, 765–771 (2000).
Odum, R. A. & Aaronson, A. M. Intermolecular reaction of an aryl nitrene. J. Am. Chem. Soc. 91, 5680–5681 (1969).
Horner, L. & Christmann, A. Nitrenes. Angew. Chem. Int. Ed. 2, 599–608 (1963).
Patel, S. C. & Burns, N. Z. Conversion of aryl azides to aminopyridines. J. Am. Chem. Soc. 144, 17797–17802 (2022).
Brase, S. & Keck, D. Product class 35: aryl azides. In Science of Synthesis: Houben–Weyl Methods of Molecular Transformations (eds Ramsden, C. A. & Bellus, D.) 10.1055/sos-SD-031-02074 (2007).
Cadogan, J. I. G., Grace, D. S. B., Lim, P. K. K. & Tait, B. S. Reduction of nitro- and nitroso-compounds by tervalent phosphorus reagents. Part XII. Conversion of aryl 2-nitroaryl ethers into novel 3-aryl-2,3-dihydro-1,3,2-benzoxazaphosph(V)oles (oxazaphosphoranes) and their 2-oxo-derivatives. J. Chem. Soc. Perkin Trans. 1, 2376–2385 (1975).
Atherton, F. R. & Lambert, R. W. Nitrenes generated from nitro-compounds by various phosphorus reagents in heterocyclic synthesis. A convenient route to substituted 3H-azepines. J. Chem. Soc. Perkin Trans. 1, 1079–1084 (1973).
Li, G. et al. An improved PIII/PV═O-catalyzed reductive C–N coupling of nitroaromatics and boronic acids by mechanistic differentiation of rate- and product-determining steps. J. Am. Chem. Soc. 142, 6786–6799 (2020).
Li, G., Lavagnino, M. N., Ali, S. Z., Hu, S. & Radosevich, A. T. Tandem C/N-difunctionalization of nitroarenes: reductive amination and annulation by a ring expansion/contraction sequence. J. Am. Chem. Soc. 145, 41–46 (2023).
Ruffoni, A., Hampton, C., Simonetti, M. & Leonori, D. Photoexcited nitroarenes for the oxidative cleavage of alkenes. Nature 610, 81–86 (2022).
Cu, A. & Testa, A. C. Photochemistry of the nitro group in aromatic heterocyclic molecules. J. Phys. Chem. 79, 644–646 (1975).
Odum, R. A. & Brenner, M. Rearrangement on deoxygenation of nitrosobenzene. J. Am. Chem. Soc. 88, 2074–2075 (1966).
Iddon, B., Meth-Cohn, O., Scriven, E. F. V., Suschitzky, H. & Gallagher, P. T. Developments in arylnitrene chemistry: syntheses and mechanisms [new synthetic methods (31)]. Angew. Chem. Int. Ed. 18, 900–917 (1979).
Gritsan, N. P., Yuzawa, T. & Platz, M. S. Direct observation of singlet phenylnitrene and measurement of its rate of rearrangement. J. Am. Chem. Soc. 119, 5059–5060 (1997).
Marcinek, A. et al. Unusually long lifetimes of the singlet nitrenes derived from 4-azido-2,3,5,6-tetrafluorobenzamides. J. Phys. Chem. 98, 412–419 (1994).
DeGraff, B. A., Gillespie, D. W. & Sundberg, R. J. Phenyl nitrene. Flash photolytic investigation of the reaction with secondary amines. J. Am. Chem. Soc. 96, 7491–7496 (1974).
Chapman, O. L. & Le Roux, J. P. 1-Aza-1,2,4,6-cycloheptatetraene. J. Am. Chem. Soc. 100, 282–285 (1978).
Satake, K. et al. Synthesis and characterization of 2H-, 3H- and 4H-azepine: the first observation of the thermal distribution equilibrium of azepines. J. Chem. Soc. Chem. Commun. https://doi.org/10.1039/C39910001154 (1991).
Negele, S., Wieser, K. & Severin, T. Photochemical oxidation of hydrocarbons by nitropyridinium salts. J. Org. Chem. 63, 1138–1143 (1998).
Burdzinski, G. et al. Early events in the photochemistry of aryl azides from femtosecond UV/vis spectroscopy and quantum chemical calculations. J. Am. Chem. Soc. 128, 13402–13411 (2006).
Nairoukh, Z., Wollenburg, M., Schlepphorst, C., Bergander, K. & Glorius, F. The formation of all-cis-(multi)fluorinated piperidines by a dearomatization–hydrogenation process. Nat. Chem. 11, 264–270 (2019).
Blakemore, D. et al. (eds) in Synthetic Methods in Drug Discovery Vol. 1 1–69 (The Royal Society of Chemistry, 2016).
West, M. J., Fyfe, J. W. B., Vantourout, J. C. & Watson, A. J. B. Mechanistic development and recent applications of the Chan–Lam amination. Chem. Rev. 119, 12491–12523 (2019).
Leonori, D. & Aggarwal, V. K. Lithiation–borylation methodology and its application in synthesis. Acc. Chem. Res. 47, 3174–3183 (2014).
Matteson, D. S., Collins, B. S. L., Aggarwal, V. K. & Ciganek, E. in Organic Reactions 427–860.
Delhaye, L., Merschaert, A., Diker, K. & Houpis, I. N. Synthesis of 4,4′-disubstituted azepines via ring-closing metathesis reaction and asymmetric arylation of lactones. Synthesis 9, 1437–1442 (2006).
Guillemont, J. E. G., Lancois, D. F. A., Motte, M. M. S., Koul, A. & Balemans, W. M. A. PCT Int. Appl. WO 2013021051 A2013021051 2020130214 (2013).
Lamara, K. & Smalley, R. K. 3H-azepines and related systems. Part 4. Preparation of 3H-azepin-2-ones and 6H-azepino[2,1-b]quinazolin-12-ones by photo-induced ring expansions of aryl azides. Tetrahedron 47, 2277–2290 (1991).
Genovino, J., Sames, D., Hamann, L. G. & Touré, B. B. Accessing drug metabolites via transition-metal catalyzed C−H oxidation: the liver as synthetic inspiration. Angew. Chem. Int. Ed. 55, 14218–14238 (2016).
St. Jean, D. J. Jr & Fotsch, C. Mitigating heterocycle metabolism in drug discovery. J. Med. Chem. 55, 6002–6020 (2012).
Ertl, P., Altmann, E., Racine, S. & Lewis, R. Ring replacement recommender: ring modifications for improving biological activity. Eur. J. Med. Chem. 238, 114483 (2022).
Goldberg, F. W., Kettle, J. G., Kogej, T., Perry, M. W. D. & Tomkinson, N. P. Designing novel building blocks is an overlooked strategy to improve compound quality. Drug Discov. Today 20, 11–17 (2015).
Hu, Y., Stumpfe, D. & Bajorath, J. Recent advances in scaffold hopping. J. Med. Chem. 60, 1238–1246 (2017).
Karakas, E., Simorowski, N. & Furukawa, H. Subunit arrangement and phenylethanolamine binding in GluN1/GluN2B NMDA receptors. Nature 475, 249–253 (2011).
Schubart, A. et al. Small-molecule factor B inhibitor for the treatment of complement-mediated diseases. Proc. Natl Acad. Sci. USA 116, 7926–7931 (2019).
Acknowledgements
D.L. thanks the Engineering and Physical Sciences Research Council for a grant (EP/V046799/1), the European Research Council for a research grant (758427) and the Leverhulme Trust for additional support (Philip Leverhulme Prize to D.L.). R.S.-B. thanks the European Union Horizon 2020 innovation programme for the Marie Skłodowska-Curie grant, agreement no. 956324 (PhotoReAct). E.M. thanks Ministerio de Universidades of the Spanish Government and the European Union Funds for a Postdoctoral Margarita Salas (NextGenerationEU) Fellowship (MSALAS-2022-19993). We thank C. Vermeeren (RWTH Aachen University) for help with the purification of some of the products.
Author information
Authors and Affiliations
Contributions
A.R. and D.L. designed the project. R.M., R.S.-B., E.M., V.K.D., A.V., L.A. and J.L. performed the synthetic and mechanistic experiments. R.J.C. performed the docking experiments. All authors analysed and discussed the results and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–6, Schemes 1–4 and Tables 1–12.
Supplementary Data 1
Coordinates (xyz) of the computational section.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mykura, R., Sánchez-Bento, R., Matador, E. et al. Synthesis of polysubstituted azepanes by dearomative ring expansion of nitroarenes. Nat. Chem. (2024). https://doi.org/10.1038/s41557-023-01429-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41557-023-01429-1