Abstract
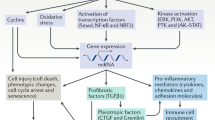
The kidney is permeated by a highly complex vascular system with glomerular and peritubular capillary networks that are essential for maintaining the normal functions of glomerular and tubular epithelial cells. The integrity of the renal vascular network depends on a balance of proangiogenic and antiangiogenic factors, and disruption of this balance has been identified in various kidney diseases. Decreased levels of the predominant proangiogenic factor, vascular endothelial growth factor A (VEGFA), can result in glomerular microangiopathy and contribute to the onset of preeclampsia, whereas upregulation of VEGFA has roles in diabetic kidney disease (DKD) and polycystic kidney disease (PKD). Other factors that regulate angiogenesis, such as angiopoietin 1 and vasohibin 1, have been shown to be protective in animal models of DKD and renal fibrosis. The renal lymphatic system is important for fluid homeostasis in the kidney, as well as the transport of immune cells and antigens. Experimental studies suggest that the lymphangiogenic factor VEGFC might have protective effects in PKD, DKD and renal fibrosis. Understanding the physiological and pathological roles of factors that regulate angiogenesis and lymphangiogenesis in the kidney has led to the development of novel therapeutic strategies for kidney diseases.
Key points
In the kidney, the glomerular and peritubular capillary networks are critical for normal nephron function; disruption of these networks can lead to the development and progression of various kidney diseases.
Normal expression of the proangiogenic factor vascular endothelial growth factor A (VEGFA), the vascular maturation factor angiopoietin 1 and the antiangiogenic factor vasohibin 1 is essential for maintaining capillary networks in the kidney.
The expression of proangiogenic and antiangiogenic factors can be altered in kidney diseases, including diabetic kidney disease (DKD) and renal fibrosis; targeting these factors is considered a promising therapeutic strategy.
Formation of abnormal lymphatic vessel networks accompanied by inflammatory infiltration is associated with various kidney diseases.
Experimental studies suggest that administration of VEGFC — the main regulator of lymphangiogenesis — might be beneficial in DKD, renal fibrosis and polycystic kidney disease.
Novel approaches to selectively delivering angiogenesis-regulating factors to the placenta and kidney have been developed as therapeutic strategies for preeclampsia and renovascular disease, respectively.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Webster, A. C., Nagler, E. V., Morton, R. L. & Masson, P. Chronic kidney disease. Lancet 389, 1238–1252 (2017).
Kida, Y., Tchao, B. N. & Yamaguchi, I. Peritubular capillary rarefaction: a new therapeutic target in chronic kidney disease. Pediatr. Nephrol. 29, 333–342 (2014).
Babickova, J. et al. Regardless of etiology, progressive renal disease causes ultrastructural and functional alterations of peritubular capillaries. Kidney Int. 91, 70–85 (2017).
Tanaka, T. & Nangaku, M. Angiogenesis and hypoxia in the kidney. Nat. Rev. Nephrol. 9, 211–222 (2013).
Zuazo-Gaztelu, I. & Casanovas, O. Unraveling the role of angiogenesis in cancer ecosystems. Front. Oncol. 8, 248 (2018).
De Palma, M., Biziato, D. & Petrova, T. V. Microenvironmental regulation of tumour angiogenesis. Nat. Rev. Cancer 17, 457–474 (2017).
Lugano, R., Ramachandran, M. & Dimberg, A. Tumor angiogenesis: causes, consequences, challenges and opportunities. Cell. Mol. Life Sci. https://doi.org/10.1007/s00018-019-03351-7 (2019).
Apte, R. S., Chen, D. S. & Ferrara, N. VEGF in signaling and disease: beyond discovery and development. Cell 176, 1248–1264 (2019).
Eremina, V. et al. Glomerular-specific alterations of VEGF-A expression lead to distinct congenital and acquired renal diseases. J. Clin. Invest. 111, 707–716 (2003).
Eremina, V. et al. VEGF inhibition and renal thrombotic microangiopathy. N. Engl. J. Med. 358, 1129–1136 (2008).
Russell, P. S., Hong, J., Windsor, J. A., Itkin, M. & Phillips, A. R. J. Renal lymphatics: anatomy, physiology, and clinical implications. Front. Physiol. 10, 251 (2019).
Rauniyar, K., Jha, S. K. & Jeltsch, M. Biology of vascular endothelial growth factor C in the morphogenesis of lymphatic vessels. Front. Bioeng. Biotechnol. 6, 7 (2018).
Graves, F. T. The anatomy of the intrarenal arteries and its application to segmental resection of the kidney. Br. J. Surg. 42, 132–139 (1954).
Macchi, V. et al. Anatomical study of renal arterial vasculature and its potential impact on partial nephrectomy. BJU Int. 120, 83–91 (2017).
Dimke, H., Maezawa, Y. & Quaggin, S. E. Crosstalk in glomerular injury and repair. Curr. Opin. Nephrol. Hypertens. 24, 231–238 (2015).
Chade, A. R. Small vessels, big role: renal microcirculation and progression of renal injury. Hypertension 69, 551–563 (2017).
Tanabe, K., Maeshima, Y., Sato, Y. & Wada, J. Antiangiogenic therapy for diabetic nephropathy. Biomed Res. Int. 2017, 5724069 (2017).
Simons, M., Gordon, E. & Claesson-Welsh, L. Mechanisms and regulation of endothelial VEGF receptor signalling. Nat. Rev. Mol. Cell Biol. 17, 611–625 (2016).
Tanabe, K., Sato, Y. & Wada, J. Endogenous antiangiogenic factors in chronic kidney disease: potential biomarkers of progression. Int. J. Mol. Sci. 19, 1859 (2018).
Peach, C. J. et al. Molecular pharmacology of VEGF-A isoforms: binding and signalling at VEGFR2. Int. J. Mol. Sci. 19, 1264 (2018).
Guo, H. F. & Vander Kooi, C. W. Neuropilin functions as an essential cell surface receptor. J. Biol. Chem. 290, 29120–29126 (2015).
Brown, L. F. et al. Vascular permeability factor mRNA and protein expression in human kidney. Kidney Int. 42, 1457–1461 (1992).
Kanellis, J., Mudge, S. J., Fraser, S., Katerelos, M. & Power, D. A. Redistribution of cytoplasmic VEGF to the basolateral aspect of renal tubular cells in ischemia-reperfusion injury. Kidney Int. 57, 2445–2456 (2000).
Wu, S., Kim, C., Baer, L. & Zhu, X. Bevacizumab increases risk for severe proteinuria in cancer patients. J. Am. Soc. Nephrol. 21, 1381–1389 (2010).
Moslehi, J. J. Cardiovascular toxic effects of targeted cancer therapies. N. Engl. J. Med. 375, 1457–1467 (2016).
Person, F. et al. Bevacizumab-associated glomerular microangiopathy. Mod. Pathol. 32, 684–700 (2019).
Kandula, P. & Agarwal, R. Proteinuria and hypertension with tyrosine kinase inhibitors. Kidney Int. 80, 1271–1277 (2011).
Izzedine, H. et al. Kidney diseases associated with anti-vascular endothelial growth factor (VEGF): an 8-year observational study at a single center. Medicine 93, 333–339 (2014).
Estrada, C. C., Maldonado, A. & Mallipattu, S. K. Therapeutic inhibition of VEGF signaling and associated nephrotoxicities. J. Am. Soc. Nephrol. 30, 187–200 (2019).
Dimke, H. et al. Tubulovascular cross-talk by vascular endothelial growth factor a maintains peritubular microvasculature in kidney. J. Am. Soc. Nephrol. 26, 1027–1038 (2015).
Hakroush, S. et al. Effects of increased renal tubular vascular endothelial growth factor (VEGF) on fibrosis, cyst formation, and glomerular disease. Am. J. Pathol. 175, 1883–1895 (2009).
Roth, L. et al. Neuropilin-1 mediates vascular permeability independently of vascular endothelial growth factor receptor-2 activation. Sci. Signal. 9, ra42 (2016).
Aggarwal, P. K. et al. Semaphorin3a promotes advanced diabetic nephropathy. Diabetes 64, 1743–1759 (2015).
Sawamiphak, S. et al. Ephrin-B2 regulates VEGFR2 function in developmental and tumour angiogenesis. Nature 465, 487–491 (2010).
Kida, Y., Ieronimakis, N., Schrimpf, C., Reyes, M. & Duffield, J. S. EphrinB2 reverse signaling protects against capillary rarefaction and fibrosis after kidney injury. J. Am. Soc. Nephrol. 24, 559–572 (2013).
Becerra, S. P. & Notario, V. The effects of PEDF on cancer biology: mechanisms of action and therapeutic potential. Nat. Rev. Cancer 13, 258–271 (2013).
Fujimura, T. et al. Administration of pigment epithelium-derived factor (PEDF) reduces proteinuria by suppressing decreased nephrin and increased VEGF expression in the glomeruli of adriamycin-injected rats. Nephrol. Dial. Transplant. 24, 1397–1406 (2009).
Saharinen, P., Eklund, L. & Alitalo, K. Therapeutic targeting of the angiopoietin-TIE pathway. Nat. Rev. Drug. Discov. 16, 635–661 (2017).
Woolf, A. S., Gnudi, L. & Long, D. A. Roles of angiopoietins in kidney development and disease. J. Am. Soc. Nephrol. 20, 239–244 (2009).
Parikh, S. M. The angiopoietin-Tie2 signaling axis in systemic inflammation. J. Am. Soc. Nephrol. 28, 1973–1982 (2017).
Winderlich, M. et al. VE-PTP controls blood vessel development by balancing Tie-2 activity. J. Cell Biol. 185, 657–671 (2009).
Souma, T. et al. Context-dependent functions of angiopoietin 2 are determined by the endothelial phosphatase VEPTP. Proc. Natl Acad. Sci. USA 115, 1298–1303 (2018).
Bafunno, V. et al. Mutation of the angiopoietin-1 gene (ANGPT1) associates with a new type of hereditary angioedema. J. Allergy Clin. Immunol. 141, 1009–1017 (2018).
d’Apolito, M. et al. Angiopoietin-1 haploinsufficiency affects the endothelial barrier and causes hereditary angioedema. Clin. Exp. Allergy 49, 626–635 (2019).
Yuan, H. T., Suri, C., Yancopoulos, G. D. & Woolf, A. S. Expression of angiopoietin-1, angiopoietin-2, and the Tie-2 receptor tyrosine kinase during mouse kidney maturation. J. Am. Soc. Nephrol. 10, 1722–1736 (1999).
Jeansson, M. et al. Angiopoietin-1 is essential in mouse vasculature during development and in response to injury. J. Clin. Invest. 121, 2278–2289 (2011).
Loganathan, K. et al. Angiopoietin-1 deficiency increases renal capillary rarefaction and tubulointerstitial fibrosis in mice. PLoS One 13, e0189433 (2018).
Kenig-Kozlovsky, Y. et al. Ascending vasa recta are angiopoietin/Tie2-dependent lymphatic-like vessels. J. Am. Soc. Nephrol. 29, 1097–1107 (2018).
Davis, B. et al. Podocyte-specific expression of angiopoietin-2 causes proteinuria and apoptosis of glomerular endothelia. J. Am. Soc. Nephrol. 18, 2320–2329 (2007).
Allegretti, A. S. et al. Serum angiopoietin-2 predicts mortality and kidney outcomes in decompensated cirrhosis. Hepatology 69, 729–741 (2019).
Araujo, C. B. et al. Angiopoietin-2 as a predictor of acute kidney injury in critically ill patients and association with ARDS. Respirology 24, 345–351 (2019).
El-Kenawi, A. E. & El-Remessy, A. B. Angiogenesis inhibitors in cancer therapy: mechanistic perspective on classification and treatment rationales. Br. J. Pharmacol. 170, 712–729 (2013).
Walia, A. et al. Endostatin’s emerging roles in angiogenesis, lymphangiogenesis, disease, and clinical applications. Biochim. Biophys. Acta 1850, 2422–2438 (2015).
Marneros, A. G. & Olsen, B. R. Physiological role of collagen XVIII and endostatin. FASEB J. 19, 716–728 (2005).
Utriainen, A. et al. Structurally altered basement membranes and hydrocephalus in a type XVIII collagen deficient mouse line. Hum. Mol. Genet. 13, 2089–2099 (2004).
Hamano, Y. et al. Lack of collagen XVIII/endostatin exacerbates immune-mediated glomerulonephritis. J. Am. Soc. Nephrol. 21, 1445–1455 (2010).
Chauhan, K. et al. Plasma endostatin predicts kidney outcomes in patients with type 2 diabetes. Kidney Int. 95, 439–446 (2019).
Watanabe, K. et al. Vasohibin as an endothelium-derived negative feedback regulator of angiogenesis. J. Clin. Invest. 114, 898–907 (2004).
Miyashita, H. et al. Angiogenesis inhibitor vasohibin-1 enhances stress resistance of endothelial cells via induction of SOD2 and SIRT1. PLoS One 7, e46459 (2012).
Kimura, H. et al. Distinctive localization and opposed roles of vasohibin-1 and vasohibin-2 in the regulation of angiogenesis. Blood 113, 4810–4818 (2009).
Hosaka, T. et al. Vasohibin-1 expression in endothelium of tumor blood vessels regulates angiogenesis. Am. J. Pathol. 175, 430–439 (2009).
Koyanagi, T. et al. Downregulation of vasohibin-2, a novel angiogenesis regulator, suppresses tumor growth by inhibiting angiogenesis in endometrial cancer cells. Oncol. Lett. 5, 1058–1062 (2013).
Olmer, R. et al. Long term expansion of undifferentiated human iPS and ES cells in suspension culture using a defined medium. Stem Cell Res. 5, 51–64 (2010).
Norita, R. et al. Vasohibin-2 is required for epithelial-mesenchymal transition of ovarian cancer cells by modulating transforming growth factor-beta signaling. Cancer Sci. 108, 419–426 (2017).
Takeda, E., Suzuki, Y., Yamada, T., Katagiri, H. & Sato, Y. Knockout of vasohibin-1 gene in mice results in healthy longevity with reduced expression of insulin receptor, insulin receptor substrate 1, and insulin receptor substrate 2 in their white adipose tissue. J. Aging Res. 2017, 9851380 (2017).
Tanimura, S. et al. Renal tubular injury exacerbated by vasohibin-1 deficiency in a murine cisplatin-induced acute kidney injury model. Am. J. Physiol. Renal Physiol. 317, F264–F274 (2019).
Masuda, K. et al. Deletion of pro-angiogenic factor vasohibin-2 ameliorates glomerular alterations in a mouse diabetic nephropathy model. PLoS One 13, e0195779 (2018).
Hinamoto, N. et al. Urinary and plasma levels of vasohibin-1 can predict renal functional deterioration in patients with renal disorders. PLoS One 9, e96932 (2014).
Ren, H. et al. Expression levels of serum vasohibin-1 and other biomarkers in type 2 diabetes mellitus patients with different urinary albumin to creatinine ratios. J. Diabetes Complicat. 33, 477–484 (2019).
Meng, X. M., Nikolic-Paterson, D. J. & Lan, H. Y. TGF-beta: the master regulator of fibrosis. Nat. Rev. Nephrol. 12, 325–338 (2016).
Pardali, E., Goumans, M. J. & ten Dijke, P. Signaling by members of the TGF-beta family in vascular morphogenesis and disease. Trends Cell Biol. 20, 556–567 (2010).
Cunha, S. I. & Pietras, K. ALK1 as an emerging target for antiangiogenic therapy of cancer. Blood 117, 6999–7006 (2011).
Lopez-Novoa, J. M. & Bernabeu, C. The physiological role of endoglin in the cardiovascular system. Am. J. Physiol. Heart Circ. Physiol. 299, H959–H974 (2010).
Ruiz-Llorente, L. et al. Endoglin and alk1 as therapeutic targets for hereditary hemorrhagic telangiectasia. Expert. Opin. Ther. Targets 21, 933–947 (2017).
Wang, X. et al. LRG1 promotes angiogenesis by modulating endothelial TGF-beta signalling. Nature 499, 306–311 (2013).
Haku, S. et al. Early enhanced leucine-rich alpha-2-glycoprotein-1 expression in glomerular endothelial cells of type 2 diabetic nephropathy model mice. Biomed Res. Int. 2018, 2817045 (2018).
Hong, Q. et al. LRG1 promotes diabetic kidney disease progression by enhancing TGF-beta-induced angiogenesis. J. Am. Soc. Nephrol. 30, 546–562 (2019).
Karaman, S., Leppanen, V. M. & Alitalo, K. Vascular endothelial growth factor signaling in development and disease. Development 145, dev.151019 (2018).
Jha, S. K. et al. KLK3/PSA and cathepsin D activate VEGF-C and VEGF-D. eLife 8, e44478 (2019).
Dellinger, M. T., Hunter, R. J., Bernas, M. J., Witte, M. H. & Erickson, R. P. Chy-3 mice are Vegfc haploinsufficient and exhibit defective dermal superficial to deep lymphatic transition and dermal lymphatic hypoplasia. Dev. Dyn. 236, 2346–2355 (2007).
Balboa-Beltran, E. et al. A novel stop mutation in the vascular endothelial growth factor-C gene (VEGFC) results in Milroy-like disease. J. Med. Genet. 51, 475–478 (2014).
Nurmi, H. et al. VEGF-C is required for intestinal lymphatic vessel maintenance and lipid absorption. EMBO Mol. Med. 7, 1418–1425 (2015).
Baldwin, M. E. et al. Vascular endothelial growth factor D is dispensable for development of the lymphatic system. Mol. Cell. Biol. 25, 2441–2449 (2005).
Koch, M. et al. VEGF-D deficiency in mice does not affect embryonic or postnatal lymphangiogenesis but reduces lymphatic metastasis. J. Pathol. 219, 356–364 (2009).
Haiko, P. et al. Deletion of vascular endothelial growth factor C (VEGF-C) and VEGF-D is not equivalent to VEGF receptor 3 deletion in mouse embryos. Mol. Cell. Biol. 28, 4843–4850 (2008).
Zarjou, A. et al. Dynamic signature of lymphangiogenesis during acute kidney injury and chronic kidney disease. Lab. Invest. 99, 1376–1388 (2019).
Foster, R. R. et al. VEGF-C promotes survival in podocytes. Am. J. Physiol. Renal Physiol. 291, F196–F207 (2006).
Foster, R. R. et al. Vascular endothelial growth factor-C, a potential paracrine regulator of glomerular permeability, increases glomerular endothelial cell monolayer integrity and intracellular calcium. Am. J. Pathol. 173, 938–948 (2008).
Bartlett, C. S., Jeansson, M. & Quaggin, S. E. Vascular growth factors and glomerular disease. Annu. Rev. Physiol. 78, 437–461 (2016).
Armaly, Z., Jadaon, J. E., Jabbour, A. & Abassi, Z. A. Preeclampsia: novel mechanisms and potential therapeutic approaches. Front. Physiol. 9, 973 (2018).
Stillman, I. E. & Karumanchi, S. A. The glomerular injury of preeclampsia. J. Am. Soc. Nephrol. 18, 2281–2284 (2007).
Levine, R. J. et al. Circulating angiogenic factors and the risk of preeclampsia. N. Engl. J. Med. 350, 672–683 (2004).
Agrawal, S., Cerdeira, A. S., Redman, C. & Vatish, M. Meta-analysis and systematic review to assess the role of soluble FMS-like tyrosine kinase-1 and placenta growth factor ratio in prediction of preeclampsia: the SaPPPhirE study. Hypertension 71, 306–316 (2018).
Zeisler, H. et al. Predictive value of the sFlt-1:PlGF ratio in women with suspected preeclampsia. N. Engl. J. Med. 374, 13–22 (2016).
Rana, S., Lemoine, E., Granger, J. & Karumanchi, S. A. Preeclampsia: pathophysiology, challenges, and perspectives. Circ. Res. 124, 1094–1112 (2019).
Spradley, F. T. et al. Placental growth factor administration abolishes placental ischemia-induced hypertension. Hypertension 67, 740–747 (2016).
Makris, A. et al. Placental growth factor reduces blood pressure in a uteroplacental ischemia model of preeclampsia in nonhuman primates. Hypertension 67, 1263–1272 (2016).
Ren, Z., Cui, N., Zhu, M. & Khalil, R. A. Placental growth factor reverses decreased vascular and uteroplacental MMP-2 and MMP-9 and increased MMP-1 and MMP-7 and collagen types I and IV in hypertensive pregnancy. Am. J. Physiol. Heart Circ. Physiol. 315, H33–H47 (2018).
Logue, O. C., Mahdi, F., Chapman, H., George, E. M. & Bidwell, G. L., III. A maternally sequestered, biopolymer-stabilized vascular endothelial growth factor (VEGF) chimera for treatment of preeclampsia. J. Am. Heart Assoc. 6, e007216 (2017).
Parchem, J. G. et al. Loss of placental growth factor ameliorates maternal hypertension and preeclampsia in mice. J. Clin. Invest. 128, 5008–5017 (2018).
Bergmann, A. et al. Reduction of circulating soluble Flt-1 alleviates preeclampsia-like symptoms in a mouse model. J. Cell. Mol. Med. 14, 1857–1867 (2010).
Jin, J. et al. Soluble FLT1 binds lipid microdomains in podocytes to control cell morphology and glomerular barrier function. Cell 151, 384–399 (2012).
Turanov, A. A. et al. RNAi modulation of placental sFLT1 for the treatment of preeclampsia. Nat. Biotechnol. 36, 1164–1173 (2018).
Robertson, S. A. Preventing preeclampsia by silencing soluble Flt-1? N. Engl. J. Med. 380, 1080–1082 (2019).
Trapiella-Alfonso, L. et al. VEGF (vascular endothelial growth factor) functionalized magnetic beads in a microfluidic device to improve the angiogenic balance in preeclampsia. Hypertension 74, 145–153 (2019).
Brenner, B. M. et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N. Engl. J. Med. 345, 861–869 (2001).
Perkovic, V. et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N. Engl. J. Med. 380, 2295–2306 (2019).
Tervaert, T. W. et al. Pathologic classification of diabetic nephropathy. J. Am. Soc. Nephrol. 21, 556–563 (2010).
Reidy, K., Kang, H. M., Hostetter, T. & Susztak, K. Molecular mechanisms of diabetic kidney disease. J. Clin. Invest. 124, 2333–2340 (2014).
Nakagawa, T. et al. Endothelial dysfunction as a potential contributor in diabetic nephropathy. Nat. Rev. Nephrol. 7, 36–44 (2011).
Nakagawa, T. et al. Diabetic endothelial nitric oxide synthase knockout mice develop advanced diabetic nephropathy. J. Am. Soc. Nephrol. 18, 539–550 (2007).
Mohan, S. et al. Diabetic eNOS knockout mice develop distinct macro- and microvascular complications. Lab. Invest. 88, 515–528 (2008).
Kanesaki, Y. et al. Vascular endothelial growth factor gene expression is correlated with glomerular neovascularization in human diabetic nephropathy. Am. J. Kidney Dis. 45, 288–294 (2005).
Veron, D. et al. Podocyte vascular endothelial growth factor (VEGF(1)(6)(4)) overexpression causes severe nodular glomerulosclerosis in a mouse model of type 1 diabetes. Diabetologia 54, 1227–1241 (2011).
Veron, D. et al. Podocyte-specific VEGF-a gain of function induces nodular glomerulosclerosis in eNOS null mice. J. Am. Soc. Nephrol. 25, 1814–1824 (2014).
Baelde, H. J. et al. Reduction of VEGF-A and CTGF expression in diabetic nephropathy is associated with podocyte loss. Kidney Int. 71, 637–645 (2007).
Sivaskandarajah, G. A. et al. Vegfa protects the glomerular microvasculature in diabetes. Diabetes 61, 2958–2966 (2012).
Dessapt-Baradez, C. et al. Targeted glomerular angiopoietin-1 therapy for early diabetic kidney disease. J. Am. Soc. Nephrol. 25, 33–42 (2014).
Carota, I. A. et al. Targeting VE-PTP phosphatase protects the kidney from diabetic injury. J. Exp. Med. 216, 936–949 (2019).
Hinamoto, N. et al. Exacerbation of diabetic renal alterations in mice lacking vasohibin-1. PLoS One 9, e107934 (2014).
Nasu, T. et al. Vasohibin-1, a negative feedback regulator of angiogenesis, ameliorates renal alterations in a mouse model of diabetic nephropathy. Diabetes 58, 2365–2375 (2009).
Bus, P. et al. Endoglin mediates vascular endothelial growth factor-A-induced endothelial cell activation by regulating Akt signaling. Am. J. Pathol. 188, 2924–2935 (2018).
Onions, K. L. et al. VEGFC reduces glomerular albumin permeability and protects against alterations in VEGF receptor expression in diabetic nephropathy. Diabetes 68, 172–187 (2019).
Floege, J. & Amann, K. Primary glomerulonephritides. Lancet 387, 2036–2048 (2016).
Avihingsanon, Y. et al. Decreased renal expression of vascular endothelial growth factor in lupus nephritis is associated with worse prognosis. Kidney Int. 75, 1340–1348 (2009).
Sato, W. et al. The pivotal role of VEGF on glomerular macrophage infiltration in advanced diabetic nephropathy. Lab. Invest. 88, 949–961 (2008).
Keir, L. S. et al. VEGF regulates local inhibitory complement proteins in the eye and kidney. J. Clin. Invest. 127, 199–214 (2017).
Schmitt, R. & Melk, A. Molecular mechanisms of renal aging. Kidney Int. 92, 569–579 (2017).
Glassock, R. J. & Rule, A. D. The implications of anatomical and functional changes of the aging kidney: with an emphasis on the glomeruli. Kidney Int. 82, 270–277 (2012).
Leonard, E. C., Friedrich, J. L. & Basile, D. P. VEGF-121 preserves renal microvessel structure and ameliorates secondary renal disease following acute kidney injury. Am. J. Physiol. Renal Physiol. 295, F1648–F1657 (2008).
Basile, D. P. et al. Impaired endothelial proliferation and mesenchymal transition contribute to vascular rarefaction following acute kidney injury. Am. J. Physiol. Renal Physiol. 300, F721–F733 (2011).
Liu, E. et al. Increased expression of vascular endothelial growth factor in kidney leads to progressive impairment of glomerular functions. J. Am. Soc. Nephrol. 18, 2094–2104 (2007).
Singh, S. et al. Tubular overexpression of angiopoietin-1 attenuates renal fibrosis. PLoS One 11, e0158908 (2016).
Takeda, E., Suzuki, Y. & Sato, Y. Age-associated downregulation of vasohibin-1 in vascular endothelial cells. Aging Cell 15, 885–892 (2016).
Watatani, H. et al. Vasohibin-1 deficiency enhances renal fibrosis and inflammation after unilateral ureteral obstruction. Physiol. Rep. 2 (2014).
Lee, A. S. et al. Vascular endothelial growth factor-C and -D are involved in lymphangiogenesis in mouse unilateral ureteral obstruction. Kidney Int. 83, 50–62 (2013).
Kinashi, H. et al. Connective tissue growth factor regulates fibrosis-associated renal lymphangiogenesis. Kidney Int. 92, 850–863 (2017).
Schwager, S. & Detmar, M. Inflammation and lymphatic function. Front. Immunol. 10, 308 (2019).
Hasegawa, S. et al. Vascular endothelial growth factor-C ameliorates renal interstitial fibrosis through lymphangiogenesis in mouse unilateral ureteral obstruction. Lab. Invest. 97, 1439–1452 (2017).
Ruge, T. et al. Endostatin level is associated with kidney injury in the elderly: findings from two community-based cohorts. Am. J. Nephrol. 40, 417–424 (2014).
Lin, C. H. et al. Endostatin and kidney fibrosis in aging: a case for antagonistic pleiotropy? Am. J. Physiol. Heart Circ. Physiol. 306, H1692–H1699 (2014).
Faye, C. et al. Transglutaminase-2: a new endostatin partner in the extracellular matrix of endothelial cells. Biochem. J. 427, 467–475 (2010).
Lin, C. H. et al. Endostatin and transglutaminase 2 are involved in fibrosis of the aging kidney. Kidney Int. 89, 1281–1292 (2016).
Leonhard, W. N., Happe, H. & Peters, D. J. Variable cyst development in autosomal dominant polycystic kidney disease: the biologic context. J. Am. Soc. Nephrol. 27, 3530–3538 (2016).
Wei, W., Popov, V., Walocha, J. A., Wen, J. & Bello-Reuss, E. Evidence of angiogenesis and microvascular regression in autosomal-dominant polycystic kidney disease kidneys: a corrosion cast study. Kidney Int. 70, 1261–1268 (2006).
Suwabe, T. et al. Suitability of patients with autosomal dominant polycystic kidney disease for renal transcatheter arterial embolization. J. Am. Soc. Nephrol. 27, 2177–2187 (2016).
Song, X. et al. Systems biology of autosomal dominant polycystic kidney disease (ADPKD): computational identification of gene expression pathways and integrated regulatory networks. Hum. Mol. Genet. 18, 2328–2343 (2009).
Tao, Y. et al. VEGF receptor inhibition slows the progression of polycystic kidney disease. Kidney Int. 72, 1358–1366 (2007).
Raina, S. et al. Anti-VEGF antibody treatment accelerates polycystic kidney disease. Am. J. Physiol. Renal Physiol. 301, F773–F783 (2011).
Huang, J. L. et al. Vascular endothelial growth factor C for polycystic kidney diseases. J. Am. Soc. Nephrol. 27, 69–77 (2016).
Chade, A. R. Renovascular disease, microcirculation, and the progression of renal injury: role of angiogenesis. Am. J. Physiol. Regulatory, Integr. Comp. Physiol. 300, R783–R790 (2011).
Cooper, C. J. et al. Stenting and medical therapy for atherosclerotic renal-artery stenosis. N. Engl. J. Med. 370, 13–22 (2014).
Eirin, A. et al. Mesenchymal stem cell-derived extracellular vesicles attenuate kidney inflammation. Kidney Int. 92, 114–124 (2017).
Eirin, A. et al. Mesenchymal stem cell-derived extracellular vesicles improve the renal microvasculature in metabolic renovascular disease in swine. Cell Transplant. 27, 1080–1095 (2018).
Chade, A. R. et al. Systemic biopolymer-delivered vascular endothelial growth factor promotes therapeutic angiogenesis in experimental renovascular disease. Kidney Int. 93, 842–854 (2018).
Bidwell, G. L., 3rd et al. A kidney-selective biopolymer for targeted drug delivery. Am. J. Physiol. Renal Physiol. 312, F54–F64 (2017).
Engel, J. E., Williams, E., Williams, M. L., Bidwell, G. L., 3rd & Chade, A. R. Targeted VEGF (vascular endothelial growth factor) therapy induces long-term renal recovery in chronic kidney disease via macrophage polarization. Hypertension 74, 1113–1123 (2019).
Author information
Authors and Affiliations
Contributions
K.T. wrote the text. J.W. and Y.S. reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Nephrology thanks Abolfazl Zarjou and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Capillary rarefaction
-
A reduction in capillary vessel density.
- Pericytes
-
Cells that enwrap capillaries and microvessels.
- Vascular hyperpermeability
-
Excessive leakage of fluids and proteins from the blood vessel owing to an enhanced ability of the vessel walls to enable the passage of small molecules.
- Intracapillary hyaline pseudothrombi
-
Intracapillary thrombi-like plugs that do not contain platelets and fibrin.
- Occipital encephalocele
-
A defect in the back (occipital area) of the skull that results in a sac-like protrusion of the brain and meninges.
- Chylous ascites
-
Retention of triglyceride-rich lymph in the peritoneal cavity.
Rights and permissions
About this article
Cite this article
Tanabe, K., Wada, J. & Sato, Y. Targeting angiogenesis and lymphangiogenesis in kidney disease. Nat Rev Nephrol 16, 289–303 (2020). https://doi.org/10.1038/s41581-020-0260-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41581-020-0260-2
This article is cited by
-
The cytoskeleton adaptor protein Sorbs1 controls the development of lymphatic and venous vessels in zebrafish
BMC Biology (2024)
-
Ocular and systemic vascular endothelial growth factor ligand inhibitor use and nephrotoxicity: an update
International Urology and Nephrology (2024)
-
Notoginsenoside R1 can inhibit the interaction between FGF1 and VEGFA to retard podocyte apoptosis
BMC Endocrine Disorders (2023)
-
Targeted VEGFA therapy in regulating early acute kidney injury and late fibrosis
Acta Pharmacologica Sinica (2023)
-
Identifying key genes related to the peritubular capillary rarefaction in renal interstitial fibrosis by bioinformatics
Scientific Reports (2023)