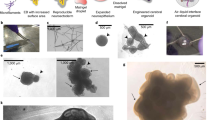
Abstract
When modeling disease in the laboratory, it is important to use clinically relevant models. Patient-derived human brain cells grown in vitro to study and test potential treatments provide such a model. Here, we present simple, highly reproducible coordinated procedures that can be used to routinely culture most cell types found in the human brain from single neurosurgically excised brain specimens. The cell types that can be cultured include dissociated cultures of neurons, astrocytes, microglia, pericytes and brain endothelial and neural precursor cells, as well as explant cultures of the leptomeninges, cortical slice cultures and brain tumor cells. The initial setup of cultures takes ~2 h, and the cells are ready for further experiments within days to weeks. The resulting cells can be studied as purified or mixed population cultures, slice cultures and explant-derived cultures. This protocol therefore enables the investigation of human brain cells to facilitate translation of neuroscience research to the clinic.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
Data availability
The data in our protocol figures are composed entirely of photomicrographs and electrophysiological recordings displaying the results anticipated with the use of the protocols. Previous uses of the protocols are available in the literature and clearly cited, providing further examples of the types of data that can be obtained from using this protocol.
References
Dragunow, M. The adult human brain in preclinical drug development. Nat. Rev. Drug Discov. 7, 659–666 (2008).
Gibbons, H. M. & Dragunow, M. Adult human brain cell culture for neuroscience research. Int. J. Biochem. Cell Biol. 42, 844–856 (2010).
Smith, A. M. & Dragunow, M. The human side of microglia. Trends Neurosci. 37, 125–135 (2014).
Dragunow, M. Human brain neuropharmacology: a platform for translational neuroscience. Trends Pharmacol. Sci. 41, 777–792 (2020).
Kim, D. et al. Generation of human induced pluripotent stem cells by direct delivery of reprogramming proteins. Cell Stem Cell 4, 472–476 (2009).
Park, I. H., Lerou, P. H., Zhao, R., Huo, H. & Daley, G. Q. Generation of human-induced pluripotent stem cells. Nat. Protoc. 3, 1180–1186 (2008).
Liu, Y. et al. Directed differentiation of forebrain GABA interneurons from human pluripotent stem cells. Nat. Protoc. 8, 1670–1679 (2013).
Moe, M. C. et al. Multipotent progenitor cells from the adult human brain: neurophysiological differentiation to mature neurons. Brain 128, 2189–2199 (2005).
Westerlund, U. et al. Stem cells from the adult human brain develop into functional neurons in culture. Exp. Cell Res. 289, 378–383 (2003).
Rustenhoven, J. et al. Modelling physiological and pathological conditions to study pericyte biology in brain function and dysfunction. BMC Neurosci. 19, 6 (2018).
Walsh, K., Megyesi, J. & Hammond, R. Human central nervous system tissue culture: a historical review and examination of recent advances. Neurobiol. Dis. 18, 2–18 (2005).
Brewer, G. J. et al. Culture and regeneration of human neurons after brain surgery. J. Neurosci. Methods 107, 15–23 (2001).
Sharif, A. & Prevot, V. Isolation and culture of human astrocytes. Methods Mol. Biol. 814, 137–151 (2012).
Gibbons, H. M. et al. Cellular composition of human glial cultures from adult biopsy brain tissue. J. Neurosci. Methods 166, 89–98 (2007).
De Groot, C. J. et al. Isolation and characterization of adult microglial cells and oligodendrocytes derived from postmortem human brain tissue. Brain Res. Brain Res. Protoc. 5, 85–94 (2000).
Rustenhoven, J. et al. Isolation of highly enriched primary human microglia for functional studies. Sci. Rep. 6, 19371 (2016).
Palmer, T. D. et al. Cell culture: progenitor cells from human brain after death. Nature 411, 42–43 (2001).
Kim, S. U., Moretto, G., Ruff, B. & Shin, D. H. Culture and cryopreservation of adult human oligodendrocytes and astrocytes. Acta Neuropathol. 64, 172–175 (1984).
Bernas, M. J. et al. Establishment of primary cultures of human brain microvascular endothelial cells to provide an in vitro cellular model of the blood-brain barrier. Nat. Protoc. 5, 1265–1272 (2010).
Park, T. I. et al. Cultured pericytes from human brain show phenotypic and functional differences associated with differential CD90 expression. Sci. Rep. 6, 26587 (2016).
Choi, P. J. et al. The synthesis of a novel Crizotinib heptamethine cyanine dye conjugate that potentiates the cytostatic and cytotoxic effects of Crizotinib in patient-derived glioblastoma cell lines. Bioorg. Med. Chem. Lett. 29, 2617–2621 (2019).
Cheng, L. et al. Glioblastoma stem cells generate vascular pericytes to support vessel function and tumor growth. Cell 153, 139–152 (2013).
Park, T. I.-H. et al. Isolation and culture of functional adult human neurons from neurosurgical brain specimens. Brain Commun. 2, fcaa171 (2020).
Smyth, L. C. D. et al. Unique and shared inflammatory profiles of human brain endothelia and pericytes. J. Neuroinflammation 15, 138 (2018).
Rustenhoven, J. et al. TGF-beta1 regulates human brain pericyte inflammatory processes involved in neurovasculature function. J. Neuroinflammation 13, 37 (2016).
Rustenhoven, J. et al. An anti-inflammatory role for C/EBPδ in human brain pericytes. Sci. Rep. 5, 12132 (2015).
Rustenhoven, J. et al. PU.1 regulates Alzheimer’s disease-associated genes in primary human microglia. Mol. Neurodegener. 13, 44 (2018).
Jansson, D. et al. Cardiac glycosides target barrier inflammation of the vasculature, meninges and choroid plexus. Commun. Biol. 4, 260 (2021).
Park, T. I. et al. Adult human brain neural progenitor cells (NPCs) and fibroblast-like cells have similar properties in vitro but only NPCs differentiate into neurons. PLoS One 7, e37742 (2012).
Choi, P. J. et al. PARP inhibitor cyanine dye conjugate with enhanced cytotoxic and antiproliferative activity in patient derived glioblastoma cell lines. Bioorg. Med. Chem. Lett. 30, 127252 (2020).
Brewer, G. J. & Torricelli, J. R. Isolation and culture of adult neurons and neurospheres. Nat. Protoc. 2, 1490–1498 (2007).
Ray, B., Chopra, N., Long, J. M. & Lahiri, D. K. Human primary mixed brain cultures: preparation, differentiation, characterization and application to neuroscience research. Mol. Brain 7, 63 (2014).
Jana, M., Jana, A., Pal, U. & Pahan, K. A simplified method for isolating highly purified neurons, oligodendrocytes, astrocytes, and microglia from the same human fetal brain tissue. Neurochem. Res. 32, 2015–2022 (2007).
Ayuso-Sacido, A., Roy, N. S., Schwartz, T. H., Greenfield, J. P. & Boockvar, J. A. Long-term expansion of adult human brain subventricular zone precursors. Neurosurgery 62, 223–231 (2008).
Gibbons, H. M. et al. Valproic acid induces microglial dysfunction, not apoptosis, in human glial cultures. Neurobiol. Dis. 41, 96–103 (2011).
Smith, A. M. et al. M-CSF increases proliferation and phagocytosis while modulating receptor and transcription factor expression in adult human microglia. J. Neuroinflammation 10, 85 (2013).
Guo, L. et al. Postmortem adult human microglia proliferate in culture to high passage and maintain their response to amyloid-β. J. Alzheimers Dis. 54, 1157–1167 (2016).
Cano-Abad, M. F. et al. New insights on culture and calcium signalling in neurons and astrocytes from epileptic patients. Int. J. Dev. Neurosci. 29, 121–129 (2011).
Williams, K., Dooley, N., Ulvestad, E., Becher, B. & Antel, J. P. IL-10 production by adult human derived microglial cells. Neurochem. Int. 29, 55–64 (1996).
Zhang, Y. et al. Purification and characterization of progenitor and mature human astrocytes reveals transcriptional and functional differences with mouse. Neuron 89, 37–53 (2016).
Lee, K. et al. Human in vitro systems for examining synaptic function and plasticity in the brain. J. Neurophysiol. 123, 945–965 (2020).
Paul, G. et al. The adult human brain harbors multipotent perivascular mesenchymal stem cells. PLoS One 7, e35577 (2012).
Verbeek, M. M., Westphal, J. R., Ruiter, D. J. & de Waal, R. M. T lymphocyte adhesion to human brain pericytes is mediated via very late antigen-4/vascular cell adhesion molecule-1 interactions. J. Immunol. 154, 5876–5884 (1995).
Arsenijevic, Y. et al. Isolation of multipotent neural precursors residing in the cortex of the adult human brain. Exp. Neurol. 170, 48–62 (2001).
Narayan, P. J., Gibbons, H. M., Mee, E. W., Faull, R. L. & Dragunow, M. High throughput quantification of cells with complex morphology in mixed cultures. J. Neurosci. Methods 164, 339–349 (2007).
Smith, A. M. et al. The transcription factor PU.1 is critical for viability and function of human brain microglia. Glia 61, 929–942 (2013).
Jansson, D. et al. A role for human brain pericytes in neuroinflammation. J. Neuroinflammation 11, 104 (2014).
Jansson, D. et al. Interferon-γ blocks signalling through PDGFRβ in human brain pericytes. J. Neuroinflammation 13, 249 (2016).
Rustenhoven, J., Jansson, D., Smyth, L. C. & Dragunow, M. Brain pericytes as mediators of neuroinflammation. Trends Pharmacol. Sci. 38, 291–304 (2017).
Stupp, R. et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 10, 459–466 (2009).
Hossain, A. et al. Mesenchymal stem cells isolated from human gliomas increase proliferation and maintain stemness of glioma stem cells through the IL-6/gp130/STAT3 pathway. Stem Cells 33, 2400–2415 (2015).
Perriot, S. et al. Human induced pluripotent stem cell-derived astrocytes are differentially activated by multiple sclerosis-associated cytokines. Stem Cell Rep. 11, 1199–1210 (2018).
Waldvogel, H. J., Curtis, M. A., Baer, K., Rees, M. I. & Faull, R. L. Immunohistochemical staining of post-mortem adult human brain sections. Nat. Protoc. 1, 2719–2732 (2006).
Mizee, M. R. et al. Isolation of primary microglia from the human post-mortem brain: effects of ante- and post-mortem variables. Acta Neuropathol. Commun. 5, 16 (2017).
Sabbagh, M. F. & Nathans, J. A genome-wide view of the de-differentiation of central nervous system endothelial cells in culture. eLife 9, e51276 (2020).
Ting, J. T. et al. Preparation of acute brain slices using an optimized N-methyl-D-glucamine protective recovery method. J. Vis. Exp. 2018, 53825 (2018).
Le Duigou, C. et al. Imaging pathological activities of human brain tissue in organotypic culture. J. Neurosci. Methods 298, 33–44 (2018).
Dragunow, M., Feng, S., Rustenhoven, J., Curtis, M. & Faull, R. Studying human brain inflammation in leptomeningeal and choroid plexus explant cultures. Neurochem. Res. 41, 579–588 (2016).
De Groot, C. J. et al. Establishment of human adult astrocyte cultures derived from postmortem multiple sclerosis and control brain and spinal cord regions: immunophenotypical and functional characterization. J. Neurosci. Res. 49, 342–354 (1997).
Armulik, A. et al. Pericytes regulate the blood-brain barrier. Nature 468, 557–561 (2010).
Smyth, L. C. D. et al. Markers for human brain pericytes and smooth muscle cells. J. Chem. Neuroanat. 92, 48–60 (2018).
Guo, W., Patzlaff, N. E., Jobe, E. M. & Zhao, X. Isolation of multipotent neural stem or progenitor cells from both the dentate gyrus and subventricular zone of a single adult mouse. Nat. Protoc. 7, 2005–2012 (2012).
Vinci, M. et al. Functional diversity and cooperativity between subclonal populations of pediatric glioblastoma and diffuse intrinsic pontine glioma cells. Nat. Med. 24, 1204–1215 (2018).
Lee, J. H. et al. Human glioblastoma arises from subventricular zone cells with low-level driver mutations. Nature 560, 243–247 (2018).
Andersson, M. et al. Optogenetic control of human neurons in organotypic brain cultures. Sci. Rep. 6, 24818 (2016).
Schwarz, N. et al. Long-term adult human brain slice cultures as a model system to study human CNS circuitry and disease. eLife 8, e48417 (2019).
Schwarz, N. et al. Human cerebrospinal fluid promotes long-term neuronal viability and network function in human neocortical organotypic brain slice cultures. Sci. Rep. 7, 12249 (2017).
Lun, M. P., Monuki, E. S. & Lehtinen, M. K. Development and functions of the choroid plexus-cerebrospinal fluid system. Nat. Rev. Neurosci. 16, 445–457 (2015).
Skipor, J. & Thiery, J. C. The choroid plexus-cerebrospinal fluid system: undervaluated pathway of neuroendocrine signaling into the brain. Acta Neurobiol. Exp. (Wars.) 68, 414–428 (2008).
Bjorefeldt, A., Illes, S., Zetterberg, H. & Hanse, E. Neuromodulation via the cerebrospinal fluid: insights from recent in vitro studies. Front. Neural Circuits 12, 5 (2018).
Bjorefeldt, A. et al. Human cerebrospinal fluid increases the excitability of pyramidal neurons in the in vitro brain slice. J. Physiol. 593, 231–243 (2015).
Smith, A. M., Gibbons, H. M., Lill, C., Faull, R. L. & Dragunow, M. Isolation and culture of adult human microglia within mixed glial cultures for functional experimentation and high-content analysis. Methods Mol. Biol. 1041, 41–51 (2013).
Acknowledgements
We are tremendously grateful to the generous neurosurgical brain tissue donors and their families, without whom this research could not be performed; thank you so much. They are the real drivers of this work, and it is because of their special gifts of brain tissue that we can undertake this work for the benefit of future patients. We also thank our collaborators and research technicians: R. Faull, M. Curtis, J. Montgomery, H. Gibbons, M. Cheung, Y. Jung, S.-L. Kim, I. Semenyajenko and M. Eszes from Auckland University; and C. Turner, P. Bergin, E. Walker, L. Roberts and S. Tharakan from Auckland Hospital and LabPlus. This work was supported by the Hugh Green Foundation (funders of the Hugh Green Biobank), a Programme Grant from the Health Research Council of New Zealand, the Sir Thomas and Lady Duncan Trust, the Coker Charitable Trust, and Brain Research New Zealand to M.D. T.I.-H.P., the Centre for Brain Research Douglas Neurosurgical Postdoctoral Research Fellow, is funded by the Douglas Charitable Trust through a School of Medicine Foundation Grant to M.D. (director of the Hugh Green Biobank).
Author information
Authors and Affiliations
Contributions
T.I.-H.P. conceived, optimized and established the neuronal, neural progenitor, microglia, pericyte and endothelial cultures; optimized the tissue-collection process and wrote most of the manuscript. L.C.D.S. conceived, optimized and established the pericyte and endothelial cultures and wrote parts of the manuscript. M.A. conceived, optimized and established the pericyte and mixed glial cultures and wrote parts of the manuscript. Z.R.W. optimized the microglial cultures and wrote parts of the manuscript. J.R. conceived, optimized and established the pericyte and microglial cultures and wrote parts of the manuscript. K.L. conceived, optimized and established the neuronal and brain slice cultures and wrote parts of the manuscript. D.J. conceived, optimized and established the mixed glial and explant cultures. A.S. optimized the mixed glial cultures and inflammatory experiments. S.F. conceived, optimized and established the mixed glial and explant cultures. J.C., P.H. and P.S. are neurosurgeons and were part of the clinical research team; they aided in design and coordination of experiments, consented and provided tissue and optimized the biopsy tissue-collection process. E.M.is a neurosurgeon and was part of the clinical research team. He conceived these projects with M.D. and consented and provided biopsy tissue. M.D.was the study leader and Principal Investigator. He conceived, led and funded the entire program and supervised T.I.-H.P., L.C.D.S., M.A., Z.R.W., J.R., K.L., D.J., A.S. and S.F. in establishing the Hugh Green Biobank.
Corresponding author
Ethics declarations
Competing interests
M.D. has developed a platform called ‘Neurovalida’ for CNS drug discovery and target validation.
Peer review
Peer review information
Nature Protocols thanks Henner Koch, Debomoy K. Lahiri, Jonas Ort and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Park, T. I.-H. et al. Brain Commun. 2, fcaa171 (2020): https://doi.org/10.1093/braincomms/fcaa171
Rustenhoven, J. et al. Sci. Rep. 6, 19371 (2016): https://doi.org/10.1038/srep19371
Jansson, D. et al. Commun. Biol. 4, 260 (2021): https://doi.org/10.1038/s42003-021-01787-x
Supplementary information
Supplementary Information
Supplementary Figs. 1 and 2, Supplementary Tables 1–3 and Supplementary Methods.
Supplementary Data 1
Raw data for Supplementary Tables 1–3.
Rights and permissions
About this article
Cite this article
Park, T.IH., Smyth, L.C.D., Aalderink, M. et al. Routine culture and study of adult human brain cells from neurosurgical specimens. Nat Protoc 17, 190–221 (2022). https://doi.org/10.1038/s41596-021-00637-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-021-00637-8
This article is cited by
-
Involvement of the tumour necrosis factor receptor system in glioblastoma cell death induced by palbociclib-heptamethine cyanine dye conjugate
Cell Communication and Signaling (2024)
-
Activation of HCA2 regulates microglial responses to alleviate neurodegeneration in LPS-induced in vivo and in vitro models
Journal of Neuroinflammation (2023)
-
Neural cell isolation from adult macaques for high-throughput analyses and neurosphere cultures
Nature Protocols (2023)
-
Brain pericytes in culture display diverse morphological and functional phenotypes
Cell Biology and Toxicology (2023)
-
Pericytes take up and degrade α-synuclein but succumb to apoptosis under cellular stress
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.