Abstract
Kaposi’s sarcoma associated herpesvirus (KSHV) a gammaherpesvirus establishes perennial latency in the host with periodic reactivation. Occasionally change in the physiological condition like hypoxia, host cell differentiation can trigger the lytic switch and reactivation of the virus. The biologically active form of 1, 25(OH)2 D3 plays a critical role in the regulation of various physiological processes (e.g. regulation of mineral homeostasis and control of bone metabolism). Apart from its role in host physiology, 1, 25(OH)2 D3 has been implicated as a potential agent for the prevention and/or treatment of many a tumors. Here we show that 1, 25(OH)2 D3 induces both death of Kaposi sarcoma associated herpesvirus infected PEL cells and KSHV replication. 1, 25(OH)2 D3 mediated inhibition of proliferation was associated with apoptosis of the PEL cells, and virus reactivation. In addition, p38 signalling is required for KSHV reactivation. Furthermore, treatment of PEL cells with p38 inhibitor abrogated the expression of ORF57, thus blocking lytic switch. Furthermore, silencing of VDR resulted in reduced ORF57 expression compared to the control cells, signifying the potential role of 1, 25(OH)2 D3 in KSHV reactivation. Thus, our studies have revealed a novel role of 1, 25(OH)2 D3 in the regulation of KSHV reactivation and PEL cell death.
Similar content being viewed by others
Introduction
Kaposi’s sarcoma associated herpesvirus (KSHV) is a DNA tumor viruses belonging to a member of gammaherpesvirus family and is associated with Kaposi sarcoma (KS), Primary effusion lymphoma (PEL) and a subtype of multicentric castleman disease (MCD)1,2,3,4. KSHV like other herpesvirus exhibits two different life cycles, latent and lytic. During latent infection, only a subset of genes are expressed, which enable KSHV to evade immune system and promote viral persistence5,6,7. While lytic cycle, lytic proteins are expressed in an ordered cascade to produce virons for their efficient propagation and transmission8,9. Studying induction of lytic switch provides an opportunity to understand the infection and pathogenesis of KSHV associated diseases. The switch from latent to lytic replication is an active area of research and has contributed to a large extent information about the cellular factors with possible roles in reactivation mechanisms. However the regulation of KSHV pathogenesis by metabolic pathways is still only sparsely understood.
Primary effusion lymphoma (PEL) is a rare HIV-associated non-Hodgkin’s lymphoma (NHL), resembles a transformed post-germinal center (GC) B cell10,11,12. PEL typically presents with lymphomatous body cavity effusions in the absence of solid tumor masses harbouring KSHV episomes and arise preferentially within the pleural or peritoneal cavities of approximately 4% of all HIV associated NHLs13,14,15. KSHV infection of PEL cell is predominantly latent, which makes PEL cells an ideal cell lines to study two phases of its life cycle16. Therapeutic induction of virus replication is necessary to target and eliminate KSHV associated tumor cells. Earlier studies have attempted induction of KSHV reactivation with a different compounds or drugs17,18,19.
Vitamin D3 was originally identified as a key regulator of bone metabolism and calcium homeostasis20. Most of the biological action of 1, 25(OH)2 D3 are exerted through nuclear receptor vitamin D receptor (VDR)21. Apart from bone metabolism and calcium homeostasis, 1, 25(OH)2 D3 has been shown to be involved in the control of angiogenesis, apoptosis, Immunomodulation, growth and differentiation of many cell types, including lymphoma cells22,23,24,25,26. VDR expression is reported in many cancers types including breast, prostrate, pancreas, colon, leukaemia’s and lymphomas27,28,29,30,31,32. Exposure of these cells to 1, 25(OH)2 D3 induces apoptosis in cells. However, studies are lacking on the role of 1, 25(OH)2 D3 in viral pathogenesis, only very few studies have indicated that vitamin D3 deficiency may confer increased risk of influenza and respiratory tract infection33,34. In vitro studies have demonstrated the effect of 1, 25(OH)2 D3 in susceptibility and control of HIV infection35. Furthermore, pre-treatment of human monoblastoid U937 cell line and monocyte derived macrophages in cell culture model of HIV infection have demonstrated anti-viral effects36. However, the underlying mechanism or pathways involving these functions is unclear, due to varied activities and functions. In addition, it remains to be identified whether 1, 25(OH)2 D3 is protective or pathogenic in cases of viral infection.
Effect of 1, 25(OH)2 D3 on downregulation of NF-κB pathway in endothelial cells transformed by Kaposi sarcoma associated herpes virus G protein coupled receptor is known37. Further, it has been shown that 1, 25(OH)2 D3 also has anti-proliferative effect on KSHV GPCR transformed endothelial cells38. Gene expression profiling of PEL cells have demonstrated that VDR is highly expressed in PEL cells as compared to normal B and T cell lymphoma and their sensitivity to vitamin D analogue EB1089, implicates a role for VDR in KSHV pathogenesis11. In view of these facts, the current investigations were taken up to dissect the mechanism (s) of action of 1, 25(OH)2 D3 on PEL cells, in particular its effect on apoptosis and reactivation.
Material and Methods
Cells and Reagents
PEL cells (JSC-1 and HBL-6) were kindly provided by Erle Robertson (University of Pennsylvania). These cells were cultured in RPMI 1640 supplemented with 10% foetal bovine serum glutamine (300 mg/mL) and streptomycin (100 mg/mL) and penicillin (100 U/mL) under 5% CO2 at 37 °C. 1, 25(OH)2 D3 was purchased from Sigma-Aldrich and was reconstituted in 90% ethanol and stored at −80 °C in an inert atmosphere in the dark. In all experiments, equal amount of 90% ethanol were added to control cultures. Pan caspase inhibitor Z-VAD-FMK was purchased from R&D system. FITC annexin V apoptosis detection kit was purchased from BD Biosciences, SB203580 (p38 inhibitor) and PD98059 (ERK inhibitor) were purchased from InvivoGen, phorbol 12-myristate 13-acetate, sodium Butyrate and MTT reagent were purchased from Sigma-Aldrich.
Cell viability assay
All cells were plated in 96 well culture plate in complete medium at a density of 5 × 104 cells per well and treated with or without increasing concentration of 1, 25(OH)2 D3(10, 50, 100, 200 nM). The plates were incubated at 37 °C, 5% CO2, for 24, 48 and 72 hours, respectively. Then, MTT solution (10 μL) for a total volume of 100 μL was added in every well and incubated for 4 hours at 37 °C with 5% CO2. Subsequently, MTT-containing medium was removed gently and replaced with DMSO (100 μL per well) and absorbance was obtained at 570 nm on a microtiter plate reader.
shRNA mediated VDR knockdown
To knock down VDR expression, two validated lentiviral constructs expressing small hairpin RNA (shRNA) sequences to targeting 2 different regions of the human VDR transcript were used. The constructs were obtained from (Sigma-Aldrich). Details of the clones and target sequences are given in Table 1. Lentiviral particles were prepared using standard protocols, resuspended in serum-free media and used to transduce JSC-1 cells. After 48 h, stably transduced cells were selected for puromycin resistance (2.5 μg/mL) for 20 days.
Quantitative Real Time RT-PCR (qRT-PCR)
Total RNA was extracted from cells using TRIzol reagent (Invitrogen, Life Technologies, USA) as per manufacturer’s instruction, followed by treatment with DNase 1. One microgram total RNA was reverse transcribed using cDNA synthesis kit (Thermo Fischer, USA). Syber green PCR was performed using primer specific for KSHV ORF57, RTA and the human GAPDH gene. Sequence of primers are mentioned in Table 2.
Cell death assay using flowcytometry
JSC-1 and HBL-6 cell were treated with 10 nM of 1, 25(OH)2 D3 for 48hr. The cells were harvested and the percentage of cells undergoing apoptosis was measured by flow cytometry after staining with fluorescein-conjugated Annexin V and propidium iodide (BD Pharmingen, USA), according to the manufacturer’s recommendation The effect of 1, 25(OH)2 D3 (24 h post treatment) in presence or absence of Z-VAD-fmk was examined. The stained cells were acquired using BD LSRFortessa and analysed with BD FACS Diva software.
Western blot analysis
Cells were lysed in modified RIPA buffer containing 150 mM NaCl, 1% NP-40, 50 mM Tris-HCl (pH 8), 0.5% deoxycholic acid, 0.1% SDS, 1% Triton X-100, protease and phosphatase inhibitors. Lysates were placed on ice for 45 minutes and then clarified by centrifugation. Supernatants were removed and total protein measured by Bradford assay. Forty microgram of protein lysate per lane was electrophoresed on 12% SDS-PAGE and transferred to nitrocellulose membranes. The membranes were blocked for 1 h in TBST blocking solution, containing 5% bovine serum albumin and then incubated with a primary antibody overnight at 4 °C. The membranes were washed at least 3 times with each wash for 10 min with washing solution (TBS and 0.1% Tween 20) and incubated for 45 min with appropriate horseradish peroxidase-conjugated secondary antibodies. The washed membranes were developed using ECL Blotting Substrate (Thermo Scientific). The β-actin,VDR, ORF57, K8α and ERK antibodies were purchased from Santa Cruz Biotechnology. The phospho-p38 mitogen-activated protein kinase (MAPK), LANA and caspase-3 antibodies were purchased from Imgenex.
Viral Load Assay
For intracellular Viral load assay, DNA was isolated using Gene elute mammalian genomic DNA isolation kit according to manufacturer’s instructions (Sigma-Aldrich) and KSHV replication was determined by qPCR using SYBR green PCR master mix (Agilent technology, USA). KSHV ORF57 gene expression compared to vehicle controls. KSHV ORF57 gene expression was compared to vehicle controls. The qPCR reactions were carried out using LC480 machine (Roche life science, USA). Relative fold expressions were determined by the ∆∆CT method39.
KSHV infection assay
JSC-1 cells were treated with 1, 25(OH)2 D3, vehicle, or TPA for 48 h. Supernatants were harvested and added to confluent monolayers of uninfected 293 cells in a 24 well dish. Polybrene (8 µg/mL) was added to each well and the plate was spinoculated at 2500 rpm for 2 h at 26 °C as previously described40. Ninety-six hours post-infection, intracellular viral loaded was determined by real time PCR. Furthermore, infection was validated by checking the expression of LANA by western blot.
Results
Antiproliferative effect of 1, 25(OH)2 D3 in PEL cell lines
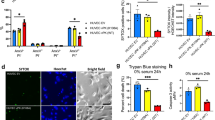
The demonstration of VDR expression in diverse tumors and cancers has emphasized that the effect of 1, 25(OH)2 D3 is not limited to VDR expression only but also display a range of antiproliferative activities. JSC-1, HBL-6 and DG-75 cells were exposed to different 1, 25(OH)2 D3 concentrations, (0 to 200 nM), for 48 h and cell viability was tested. 1, 25(OH)2 D3 induced a dose-dependent loss of viability in JSC-1 and HBL-6 PEL cells as compared to control cells DG-75 (Fig. 1A). A time-kinetic investigation showed that 1, 25(OH)2 D3 treatments (10 nM) increased cell death between 24 and 48 h in PEL cell lines but not in DG-75cells (Fig. 1B). In general, JSC-1 cells showed higher sensitivity to 1, 25(OH)2 D3 as compared to HBL-6 cells. This varied sensitivity to the 1, 25(OH)2 D3 treatment correlated to the level of VDR expression (Fig. 1C). The expression of VDR was slightly upregulated by treatment with 1, 25(OH)2 D3. Flowcytometry showed higher VDR expression in JSC-1 and DG-75 cells with 1, 25(OH)2 D3 (Fig. 1C). In contrast, VDR levels were lower in HBL-6 cells (Fig. 1C). Collectively, the above data shows that the PEL derived cell lines are remarkably sensitive to 1, 25(OH)2 D3 induced growth inhibition.
1, 25(OH)2 D3 induced loss of viability in PEL cell lines. (A) JSC-1, HBL-6 and DG-75 were treated for 48 h with different 1, 25(OH) 2 D3 concentrations (10, 50,100,150 and 200 nM) or (B) for different times (24, 48 and 72 h) with 10 nM of 1, 25(OH)2 D3, and cell viability was measured by MTT assays. The data shown here are representative of 3 independent experiments. Error bars indicate standard deviations. (C) JSC-1, HBL-6 and DG-75 cells were assessed for expression of vitamin D receptor by immunofluorescent flow cytometric assay.
1, 25(OH)2 D3 induces caspase-3 dependent cell apoptosis
To determine whether the inhibitory effects of 1, 25(OH)2 D3 on viability was associated with the induction of apoptosis, we evaluated the percentage of apoptotic cells by annexinV/PI staining. 1, 25(OH)2 D3 treated JSC-1 cells showed significantly higher percentage of apoptotic cell after 48 h (Fig. 2A). On the other hand, HBL-6 did not show any significant change in apoptosis (Fig. 2A). Simultaneously we also evaluated the effect of 1, 25(OH)2 D3 on the expression of pro and anti-apoptotic proteins. Significant increase in the cleaved PARP and caspase-3 was observed in JSC-1 cells (Fig. 2B). However, only a modest change in the level of these proteins was found in HBL-6 cells (Fig. 2C) indicating higher sensitivity of JSC-1 cells to 1, 25(OH)2 D3. Thus suggesting that VDR contributes to the cellular apoptosis induced by 1, 25(OH)2 D3 in PEL cells. Further to prove the involvement of caspases in 1, 25(OH)2 D3 induced cell death pan-caspase inhibitor Z-VAD-FMK was used. Pan Caspase inhibitor Z-VAD-FMK completely repressed cellular death as evidenced by reversal of capspase-3 and cleaved PARP to the basal levels (Fig. 3A and B). Treatment of cells with z-VAD-FMK efficiently blocked cleavage of PARP-1 and Caspase-3 in 1, 25(OH)2 D3 treated JSC-1 and HBL-6 cells. Importantly however, it did not prevent expression of lytic protein ORF57 and K8.1 in 1, 25(OH)2 D3 treated cells (Fig. 3C and D).
1, 25(OH)2 D3 induces apoptosis of PEL cells. PEL cells were cultured in the presence or absence of 1, 25(OH)2 D3 (10 nM). (A) After 48 h in culture, cells were washed and stained with Annexin V and PI and analyzed by flow cytometry. The numbers in the lower right quadrant represent the percent of apoptotic cells in culture. (B and C ). Cleaved caspase-3, and cleaved PARP expressions were detected by Western blot in JSC-1 and HBL-6 cells after treatment with 1, 25(OH)2 D3. β-actin was used to normalize protein loading.
Effects of caspases inhibition by Z-VAD-FMK on apoptosis in PEL cells. JSC-1 cells were pre-treated with the caspase inhibitor Z-VAD-FMK (20 µM) for 2 h, followed by treatment with 10 nM 1, 25(OH)2 D3 for 24 h. (A) Cells were stained with Annexin V/PI, and apoptosis was determined using flow cytometry. Values for cells treated with DIM and Z-VAD-FMK were significantly reduced as compared to treatment with 1, 25(OH)2 D3 alone. Total protein extracts were prepared and subjected to Western blot assay for cleaved PARP and cleaved caspase-3 (B). Cell lysates collected 24 h after induction with the 1, 25(OH)2 D3 (10 nm) were immunoblotted with anti-ORF57 and anti K8.1 antibody. Effect of Caspase inhibitor on the expression of ORF57 or K8.1 in cells treated with 1, 25(OH)2 D3 alone or both 1, 25(OH)2 D3 and Z-VAD-FMK (C and D). β-actin was used to normaliz protein loading.
1, 25(OH)2 D3 induces lytic gene expression and virus production in KSHV infected PEL cell lines
To determine whether VDR activation induces expression of KSHV lytic genes and virus production in 1, 25(OH)2 D3 treated PEL cells, JSC-1 and HBL-6 cells were treated with cognate 1, 25(OH)2 D3 for 48 h and monitored for the expression of LANA, RTA, ORF57 and K8.1. Significant increase in RTA, ORF57 and K8.1 were observed upon addition of 1, 25(OH)2 D3 (Fig. 4A and B). We then tested whether the increase in the expression of lytic genes also correlated to progeny virus production. For this latently infected JSC-1 cells were used, wherein cell-free virus was isolated from JSC-1 supernatants 2 days post 1, 25(OH)2 D3 treatment,.Viral DNA was extracted, and viral genome copy number determined by qPCR(4D,E and F). Figure 4G clearly demonstrates that 1, 25(OH)2 D3 induced virus production when compared to the controls. However, KSHV reactivation was comparatively lower in 1, 25(OH)2 D3 treatment than in the positive control, TPA (Fig. 4G). To validate further, a time course treatment of JSC-1 and HBL-6 cells with 1, 25(OH)2 D3 or a combination of both TPA and sodium butyrate (NaB) as a potent positive control for KSHV reactivation was performed [0,6,12,24,36 and 48 h] (Fig. 4C). Immunobloting, for expression of ORF57 and K8α showed lytic replication at the 24 h and 36 h with peak activation at 24 h (Fig. 4C). These results indicate that 1, 25(OH)2 D3 induces expression of lytic genes and progeny virus production.
1, 25(OH)2 D3 induces viral lytic gene expression in KSHV-infected PEL cells. JSC-1 (A) and HBL-6 (B) cells were treated with either 1, 25(OH)2 D3 (10 mM), or vehicle for 48 h, then viral latent (LANA) and lytic gene (RTA, K8.1, ORF57) transcripts were quantified using qRT-PCR. Error bars represent the S.E.M for three independent experiments. (C) JSC-1 and HBL-6 cells were treated as above and TPA (20 ng/mL used as positive control) and then the expression of viral lytic protein K8.1 and ORF57 was checked by western blotting. β actin was used as loading control. (D) Standard curves for the quantification of Viral DNA (KSHV), Dilution series containing between 101 and 109 copies of LANA plasmid were used as quantification standards. (E and F) Absolute copy numbers of viral DNA derived from standard curve was plotted for controls and 1, 25(OH)2 D3 treated JSC-1 and HBl-6 cells (G) JSC-1 cells were treated by vehicle, 1, 25(OH)2 D3 (10 mM), and TPA (20 ng/mL as a positive control), respectively, for 48 h, then the virions were collected as described in Methods, followed by infection of 293 cells. LANA transcripts from each group were quantified by qRT-PCR. Error bars represent the S.E.M. for 3 independent experiments.
1, 25(OH)2 D3 activates KSHV replication involving MAPK signalling pathway
To gain insights into the mechanism underlying the role of 1, 25(OH)2 D3 in the induction of reactivation, we explored cellular signalling pathways that may mediate KSHV reactivation downstream of VDR signalling. Previous studies have shown that the mitogen-activated protein kinase (MAPK) signalling pathways play important roles in KSHV reactivation induced by phorbol esters, Ras and oxidative stress41,42,43,44. We therefore examined whether MAPK signalling is required for KSHV reactivation induced by 1, 25(OH)2 D3. PD 98059, a specific inhibitor of ERK, and SB 203580, a specific inhibitor of p38 MAPK, significantly inhibited KSHV reactivation induced by 1, 25(OH)2 D3, as indicated ORF57 protein levels (Fig. 5B). In contrast, the JNK inhibitor SP 600125 did not significantly affect KSHV reactivation induced by 1, 25(OH)2 D3 (Fig. 5B). Since 1, 25(OH)2 D3 upregulated RTA transcription (Fig. 4A and B), we further tested whether ERK or p38 signaling is involved in this upregulation. Both PD 98059 and SB 203580 inhibited RTA upregulation but not SP 600125 (Fig. 5C). Thus, ERK and p38 signalling are involved in the upregulation of RTA and KSHV reactivation downstream of VDR signalling.
Induction of MAPK signalling by 1, 25(OH)2 D3. 1, 25(OH)2 D3 induces MAPK signalling pathway in PEL cells. (A) Western blotting with anti-phospho-p38, p-JNK,and anti p-ERK and anti β-actin antibodies of cell lysates of JSC-1 and HBL-6 cells treated with 10 nM D3 for 0 to 48hr showed phosphorylation of p-38 and induction of MAPK activity. (B) Cells were induced with 1, 25(OH)2 D3 in the presence of inhibitor of p38 (SB203580), ERK (PD98059) and JNK (SP600125), and analysed for the expression of ORF57 protein at 24 h. (C) Total RNA was extracted and subjected to qRT-PCR with the indicated primers to examine RTA expression in JSC-1 and HBL-6 cells. All the experiments were carried out three times, each with three replicates.
Effect of VDR knockdown on 1, 25(OH)2 D3 mediated KSHV Reactivation
To further confirm the role of VDR in KSHV reactivation, we also used lentiviral shVDR, a plasmid that express short hairpin RNA (shRNA) targeting VDR, to examine the effect of VDR depletion on KSHV reactivation. Table 1 shows the 2 shRNA constructs targeting 2 discrete regions of the human VDR transcript. We selected transfected cells with puromycin resistance gene to obtain stably transduced cells JSC-1-VDRKO. VDR knock-down JSC-1 cells were used in 1, 25(OH)2 D3 reactivation assays. Viral reactivation was measured by western analysis for the lytic protein, ORF-57. The control cell line, pTRCJSC-1 showed significant reactivation with 1, 25(OH)2 D3 (Fig. 6D) as compared VDR knockdown cells. Notably, all knock-down cell lines were responsive to lytic reactivation by TPA, and showed significant reactivation (Fig. 6D). In summary, VDR knock-down lowers 1, 25(OH)2 D3 induced viral reactivation.
VDR knockdown inhibits KSHV reactivation. JSC-1 cells were transduced with lentiviral particles containing either scrambled shRNA or shRNAs directed against the human VDR transcript. (A) Western blot analysis of stably transduced cells shows near complete absence of VDR protein in JSC-1 cells. (B and C) Cell apoptosis was significantly decreased in JSC-1 cells with VDR knockdown as compared to control shRNA transduced cells. (D) Western blotting with anti caspase-3, anti ORF57 and β-actin antibodies of cell lysates of JSC-1 VDR KO cell line and JSC-1 Ctrl treated with 10 nM of 1, 25(OH)2 D3 and 20 ng/ml of TPA used as positive control.
Discussion
In summary, we report here compelling evidence to KSHV reactivation through VDR signalling. VDR mediated reactivation from latency offers a paradigm for how KSHV may initiate lytic replication in vivo. The mechanisms controlling KSHV latncy and lytic replication are complex. Whether KSHV undergoes latent or lytic replication might depend on diverse factors including: the status of cellular signalling pathways, cell cycle, extracellular factors, cell types, stages of viral infection, and viral regulatory factors, and susceptibility to disease development45,46,47,48,49,50,51. Understanding the key cellular and molecular basis of KSHV latency and reactivation may provide newer control stategies. In this report, we demonstrate the involvement of VDR-dependent signal transduction in KSHV reactivation in latently infected cells.
The present work dissects the action of 1, 25(OH)2 D3 in PEL cells. Vitamin D receptor belongs to the superfamily of steroid receptors, which act as ligand dependent transcription factor. It is reported that VDR is constitutively expressed in primary effusion lymphoma B cells at high levels11,52. It has been previously demonstrated that active form of 1, 25(OH)2 D3 promotes growth inhibition in lymphocytes and in a variety of human cancer cell lines53,54,55,56,57. PEL cell line used in this study responds robustly to 1, 25(OH)2 D3 during a 48 h treatment period (Fig. 1). We observed strong growth inhibition at early time point from 24 h. However, this response was lost with incubation beyond 72 hours in JSC-1 cells (Fig. 1A). In contrast, a delayed response was noted in HBL-6 cells and growth inhibition starts 72 h post treatment (Fig. 1B). On the other hand, DG-75 cells showed no inhibition. Difference in sensitivity of these (PEL) cells to ligand may be due to the difference in receptor expression (Fig. 1C).
Reduction in growth, proliferation and induction of apoptosis are likely to cause Herpes virus reactivation58,59,60. Previous studies have shown that two seemingly conflicting phenotypes of KSHV reactivation and the death of PEL cells occur simultaneously43. Notably, 1, 25(OH)2 D3 mediated inhibition of proliferation was associated with apoptosis of the PEL cells (Fig. 2). On the other hand, the reactivation of PEL cells observed (expression of lytic transcripts ORF57 and K8.1) was not affected by pan caspase inhibitor (Fig. 3C and D) although it was able to suppress apoptosis (Fig. 3A), clearly indicating that these two actions are independent of each other. Even though the extent of reactivation by 1, 25(OH)2 D3 is comparatively lower than those caused by strong inducers, such as TPA. More importantly, unlike TPA and butyrate, 1, 25(OH)2 D3 is a natural product of cellular metabolism and plays a critical role in several physiological and pathological conditions. It is likely that 1, 25(OH)2 D3 may play a pivotal role in regulation and equilibrium between latent and lytic replication in PEL cells. Thus, our findings elucidate one of the possible mechanisms for the pathogenesis and reactivation associated with KSHV infection. To identify the mechanisms of reactivation of KSHV in PEL cells, several different signalling pathways have been investigated. Several authors have shown the involvement of MAPK pathways in 1, 25(OH)2 D3 treated cells. MAPK p38 has been shown to be involved skeletal and intestinal cells, thereby affecting cell cycle, growth and differentiation61,62. Previous reports have also shown the involvement of MAPK pathways in KSHV lytic replication during productive primary infection and reactivation from latency63,64. In this study, the p38 MAPK pathway however appears to be generally activated by 1, 25(OH)2 D3 in PEL cells as assessed by expression of p-p38 protein (Fig. 6A). Most importantly, this activation of p38 MAPK by 1, 25(OH)2 D3 led to reactivation of KSHV in PEL cells. Furthermore, the p38 kinase inhibitor SB203580 not only prevented p38 phosphorylation but also abrogated KSHV reactivation (Fig. 5B). On the other hand, ERK inhibitor, PD98059 only partially suppressed KSHV reactivation, while there was no change with JNK inhibitor (Fig. 5A and B) suggesting that MAPK pathways p38 and ERK may be involved in switching from latency to lytic phase in KSHV.
Treatment of 1, 25(OH)2 D3 in PEL cells caused p38, ERK expression and caspase activation indicating that signalling events bifurcate downstream of VDR in mediating these two processes, i.e., virus reactivation and cell death. KSHV reactivation from latency depends on the expression of RTA. Our finding mirrors this as RTA expression occurred following stimulation by 1, 25(OH)2 D3 (Fig. 4A and B). As RTA does not contain any VDRE, it is speculated that 1, 25(OH)2 D3 indirectly increases the RTA expression most likely via p38. Thus, increase in the RTA expression induced by 1, 25(OH)2 D3 may lead to a greater degree of KSHV reactivation, making RTA a sensitive regulator between latency and reactivation as also confirmed through infection of 293 cells (Fig. 4G).
Lastly, we determined the effects of VDR knockdown on PEL cell proliferation and KSHV reactivation. VDR Knockdown rendered JSC-1 cells significantly less susceptible to 1, 25(OH)2 D3 mediated KSHV reactivation, while virus reactivation by phorbol esters remained intact (Fig. 6D). The findings further suggest that 1, 25(OH)2 D3 may activate latent KSHV in vivo. Thus, our findings clearly establish a key role in which VDR signalling allows the virus to escape a cell that is destined to die and induces KSHV reactivation and lytic replication. These findings imply a cross talk between a host cell and a latent KSHV that determine the clinical consequences.
References
Mesri, E. A., Cesarman, E. & Boshoff, C. Kaposi’s sarcoma and its associated herpesvirus. Nat Rev Cancer 10, 707–719, https://doi.org/10.1038/nrc2888 (2010).
Cathomas, G. Kaposi’s sarcoma-associated herpesvirus (KSHV)/human herpesvirus 8 (HHV-8) as a tumour virus. Herpes 10, 72–77 (2003).
Cesarman, E., Chang, Y., Moore, P. S., Said, J. W. & Knowles, D. M. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in AIDS-related body-cavity-based lymphomas. N Engl J Med 332, 1186–1191, https://doi.org/10.1056/NEJM199505043321802 (1995).
Soulier, J. et al. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in multicentric Castleman’s disease. Blood 86, 1276–1280 (1995).
Dittmer, D. et al. A cluster of latently expressed genes in Kaposi’s sarcoma-associated herpesvirus. J Virol 72, 8309–8315 (1998).
Lee, H. R., Brulois, K., Wong, L. & Jung, J. U. Modulation of Immune System by Kaposi’s Sarcoma-Associated Herpesvirus: Lessons from Viral Evasion Strategies. Front Microbiol 3, 44, https://doi.org/10.3389/fmicb.2012.00044 (2012).
Coscoy, L. Immune evasion by Kaposi’s sarcoma-associated herpesvirus. Nat Rev Immunol 7, 391–401, https://doi.org/10.1038/nri2076 (2007).
Purushothaman, P., Uppal, T. & Verma, S. C. Molecular biology of KSHV lytic reactivation. Viruses 7, 116–153, https://doi.org/10.3390/v7010116 (2015).
Miller, G., El-Guindy, A., Countryman, J., Ye, J. & Gradoville, L. Lytic cycle switches of oncogenic human gammaherpesviruses. Adv Cancer Res 97, 81–109, https://doi.org/10.1016/S0065-230X(06)97004-3 (2007).
Chen, Y. B., Rahemtullah, A. & Hochberg, E. Primary effusion lymphoma. Oncologist 12, 569–576, https://doi.org/10.1634/theoncologist.12-5-569 (2007).
Jenner, R. G. et al. Kaposi’s sarcoma-associated herpesvirus-infected primary effusion lymphoma has a plasma cell gene expression profile. Proc Natl Acad Sci USA 100, 10399–10404, https://doi.org/10.1073/pnas.1630810100 (2003).
Cesarman, E. & Knowles, D. M. The role of Kaposi’s sarcoma-associated herpesvirus (KSHV/HHV-8) in lymphoproliferative diseases. Semin Cancer Biol 9, 165–174, https://doi.org/10.1006/scbi.1998.0118 (1999).
Ansari, M. Q. et al. Primary body cavity-based AIDS-related lymphomas. Am J Clin Pathol 105, 221–229, https://doi.org/10.1093/ajcp/105.2.221 (1996).
Gaidano, G. & Carbone, A. Primary effusion lymphoma: a liquid phase lymphoma of fluid-filled body cavities. Adv Cancer Res 80, 115–146, https://doi.org/10.1016/S0065-230X(01)80014-2 (2001).
Carbone, A. et al. Primary effusion lymphoma cell lines harbouring human herpesvirus type-8. Leuk Lymphoma 36, 447–456, https://doi.org/10.3109/10428190009148391 (2000).
Drexler, H. G., Uphoff, C. C., Gaidano, G. & Carbone, A. Lymphoma cell lines: in vitro models for the study of HHV-8+ primary effusion lymphomas (body cavity-based lymphomas). Leukemia 12, 1507–1517 (1998).
Brown, H. J., McBride, W. H., Zack, J. A. & Sun, R. Prostratin and bortezomib are novel inducers of latent Kaposi’s sarcoma-associated herpesvirus. Antivir Ther 10, 745–751 (2005).
Zoeteweij, J. P., Rinderknecht, A. S., Davis, D. A., Yarchoan, R. & Blauvelt, A. Minimal reactivation of Kaposi’s sarcoma-associated herpesvirus by corticosteroids in latently infected B cell lines. J Med Virol 66, 378–383, https://doi.org/10.1002/jmv.2155 (2002).
Shaw, R. N., Arbiser, J. L. & Offermann, M. K. Valproic acid induces human herpesvirus 8 lytic gene expression in BCBL-1 cells. AIDS 14, 899–902, https://doi.org/10.1097/00002030-200005050-00021 (2000).
Bouillon, R. & Suda, T. Vitamin D: calcium and bone homeostasis during evolution. Bonekey Rep 3, 480, https://doi.org/10.1038/bonekey.2013.214 (2014).
Mangelsdorf, D. J. et al. The nuclear receptor superfamily: the second decade. Cell 83, 835–839, https://doi.org/10.1016/0092-8674(95)90199-X (1995).
Mantell, D. J., Owens, P. E., Bundred, N. J., Mawer, E. B. & Canfield, A. E. 1 alpha,25-dihydroxyvitamin D(3) inhibits angiogenesis in vitro and in vivo. Circ Res 87, 214–220, https://doi.org/10.1161/01.RES.87.3.214 (2000).
Grundmann, M. et al. Vitamin D improves the angiogenic properties of endothelial progenitor cells. Am J Physiol Cell Physiol 303, C954–962, https://doi.org/10.1152/ajpcell.00030.2012 (2012).
McGuire, T. F., Trump, D. L. & Johnson, C. S. Vitamin D(3)-induced apoptosis of murine squamous cell carcinoma cells. Selective induction of caspase-dependent MEK cleavage and up-regulation of MEKK-1. J Biol Chem 276, 26365–26373, https://doi.org/10.1074/jbc.M010101200 (2001).
Mathiasen, I. S., Lademann, U. & Jaattela, M. Apoptosis induced by vitamin D compounds in breast cancer cells is inhibited by Bcl-2 but does not involve known caspases or p53. Cancer Res 59, 4848–4856 (1999).
Di Rosa, M., Malaguarnera, M., Nicoletti, F. & Malaguarnera, L. Vitamin D3: a helpful immuno-modulator. Immunology 134, 123–139, https://doi.org/10.1111/j.1365-2567.2011.03482.x (2011).
Mangelsdorf, D. J., Koeffler, H. P., Donaldson, C. A., Pike, J. W. & Haussler, M. R. 1,25-Dihydroxyvitamin D3-induced differentiation in a human promyelocytic leukemia cell line (HL-60): receptor-mediated maturation to macrophage-like cells. J Cell Biol 98, 391–398, https://doi.org/10.1083/jcb.98.2.391 (1984).
Hendrickson, W. K. et al. Vitamin D receptor protein expression in tumor tissue and prostate cancer progression. J Clin Oncol 29, 2378–2385, https://doi.org/10.1200/JCO.2010.30.9880 (2011).
Wada, K. et al. Vitamin D receptor expression is associated with colon cancer in ulcerative colitis. Oncol Rep 22, 1021–1025, https://doi.org/10.3892/or_00000530 (2009).
Albrechtsson, E. et al. Vitamin D receptor is expressed in pancreatic cancer cells and a vitamin D3 analogue decreases cell number. Pancreatology 3, 41–46, https://doi.org/10.1159/000069149 (2003).
Hickish, T. et al. The effect of 1,25-dihydroxyvitamin D3 on lymphoma cell lines and expression of vitamin D receptor in lymphoma. Br J Cancer 68, 668–672, https://doi.org/10.1038/bjc.1993.406 (1993).
Ruggiero, M. et al. Vitamin D receptor gene polymorphism is associated with metastatic breast cancer. Oncol Res 10, 43–46 (1998).
Aloia, J. F. & Li-Ng, M. Re: epidemic influenza and vitamin D. Epidemiol Infect 135, 1095–1096; author reply 1097–1098, https://doi.org/10.1017/S0950268807008308 (2007).
Roth, D. E., Jones, A. B., Prosser, C., Robinson, J. L. & Vohra, S. Vitamin D receptor polymorphisms and the risk of acute lower respiratory tract infection in early childhood. J Infect Dis 197, 676–680, https://doi.org/10.1086/527488 (2008).
Villamor, E. A potential role for vitamin D on HIV infection? Nutr Rev 64, 226–233, https://doi.org/10.1111/j.1753-4887.2006.tb00205.x (2006).
Pauza, C. D., Kornbluth, R., Emau, P., Richman, D. D. & Deftos, L. J. Vitamin D3 compounds regulate human immunodeficiency virus type 1 replication in U937 monoblastoid cells and in monocyte-derived macrophages. J Leukoc Biol 53, 157–164, https://doi.org/10.1038/bjc.1993.406 (1993).
Gonzalez-Pardo, V., Verstuyf, A., Boland, R. & Russo de Boland, A. Vitamin D analogue TX 527 down-regulates the NF-kappaB pathway and controls the proliferation of endothelial cells transformed by Kaposi sarcoma herpesvirus. Br J Pharmacol 169, 1635–1645, https://doi.org/10.1111/bph.12219 (2013).
Gonzalez-Pardo, V. et al. 1 Alpha,25-dihydroxyvitamin D3 and its TX527 analog inhibit the growth of endothelial cells transformed by Kaposi sarcoma-associated herpes virus G protein-coupled receptor in vitro and in vivo. Endocrinology 151, 23–31, https://doi.org/10.1210/en.2009-0650 (2010).
Dittmer, D. P. Transcription profile of Kaposi’s sarcoma-associated herpesvirus in primary Kaposi’s sarcoma lesions as determined by real-time PCR arrays. Cancer Res 63, 2010–2015 (2003).
West, J. & Damania, B. Upregulation of the TLR3 pathway by Kaposi’s sarcoma-associated herpesvirus during primary infection. J Virol 82, 5440–5449, https://doi.org/10.1128/JVI.02590-07 (2008).
Yu, F. et al. Systematic identification of cellular signals reactivating Kaposi sarcoma-associated herpesvirus. PLoS Pathog 3, e44, https://doi.org/10.1371/journal.ppat.0030044 (2007).
McAllister, S. C. et al. Increased efficiency of phorbol ester-induced lytic reactivation of Kaposi’s sarcoma-associated herpesvirus during S phase. J Virol 79, 2626–2630, https://doi.org/10.1128/JVI.79.4.2626-2630.2005 (2005).
Li, X., Feng, J. & Sun, R. Oxidative stress induces reactivation of Kaposi’s sarcoma-associated herpesvirus and death of primary effusion lymphoma cells. J Virol 85, 715–724, https://doi.org/10.1128/JVI.01742-10 (2011).
Cohen, A., Brodie, C. & Sarid, R. An essential role of ERK signalling in TPA-induced reactivation of Kaposi’s sarcoma-associated herpesvirus. J Gen Virol 87, 795–802, https://doi.org/10.1099/vir.0.81619-0 (2006).
Chang, M. et al. beta-Adrenoreceptors reactivate Kaposi’s sarcoma-associated herpesvirus lytic replication via PKA-dependent control of viral RTA. J Virol 79, 13538–13547, https://doi.org/10.1128/JVI.79.21.13538-13547.2005 (2005).
Davis, D. A. et al. Hypoxia induces lytic replication of Kaposi sarcoma-associated herpesvirus. Blood 97, 3244–3250, https://doi.org/10.1182/blood.V97.10.3244 (2001).
Wilson, S. J. et al. X box binding protein XBP-1s transactivates the Kaposi’s sarcoma-associated herpesvirus (KSHV) ORF50 promoter, linking plasma cell differentiation to KSHV reactivation from latency. J Virol 81, 13578–13586, https://doi.org/10.1128/JVI.01663-07 (2007).
Siegel, A. M., Rangaswamy, U. S., Napier, R. J. & Speck, S. H. Blimp-1-dependent plasma cell differentiation is required for efficient maintenance of murine gammaherpesvirus latency and antiviral antibody responses. J Virol 84, 674–685, https://doi.org/10.1128/JVI.01306-09 (2010).
Dalton-Griffin, L., Wilson, S. J. & Kellam, P. X-box binding protein 1 contributes to induction of the Kaposi’s sarcoma-associated herpesvirus lytic cycle under hypoxic conditions. J Virol 83, 7202–7209, https://doi.org/10.1128/JVI.00076-09 (2009).
Chang, J., Renne, R., Dittmer, D. & Ganem, D. Inflammatory cytokines and the reactivation of Kaposi’s sarcoma-associated herpesvirus lytic replication. Virology 266, 17–25, https://doi.org/10.1006/viro.1999.0077 (2000).
Chen, J. et al. Activation of latent Kaposi’s sarcoma-associated herpesvirus by demethylation of the promoter of the lytic transactivator. Proc Natl Acad Sci USA 98, 4119–4124, https://doi.org/10.1073/pnas.051004198 (2001).
Capello, D. et al. Genome wide DNA-profiling of HIV-related B-cell lymphomas. Br J Haematol 148, 245–255, https://doi.org/10.1111/j.1365-2141.2009.07943.x (2010).
Chen, S., Sims, G. P., Chen, X. X., Gu, Y. Y. & Lipsky, P. E. Modulatory effects of 1,25-dihydroxyvitamin D3 on human B cell differentiation. J Immunol 179, 1634–1647, https://doi.org/10.4049/jimmunol.179.3.1634 (2007).
Li, H. X. et al. Vitamin D3 potentiates the growth inhibitory effects of metformin in DU145 human prostate cancer cells mediated by AMPK/mTOR signalling pathway. Clin Exp Pharmacol Physiol 42, 711–717, https://doi.org/10.1111/1440-1681.12409 (2015).
Park, W. H. et al. The induction of apoptosis by a combined 1,25(OH)2D3 analog, EB1089 and TGF-beta1 in NCI-H929 multiple myeloma cells. Int J Oncol 20, 533–542, https://doi.org/10.3892/ijo.20.3.533 (2002).
James, S. Y., Mackay, A. G. & Colston, K. W. Effects of 1,25 dihydroxyvitamin D3 and its analogues on induction of apoptosis in breast cancer cells. J Steroid Biochem Mol Biol 58, 395–401, https://doi.org/10.1016/0960-0760(96)00048-9 (1996).
Bruns, H. et al. Vitamin D-dependent induction of cathelicidin in human macrophages results in cytotoxicity against high-grade B cell lymphoma. Sci Transl Med 7, 282ra247, https://doi.org/10.1126/scitranslmed.aaa3230 (2015).
Prasad, A., Remick, J. & Zeichner, S. L. Activation of human herpesvirus replication by apoptosis. J Virol 87, 10641–10650, https://doi.org/10.1128/JVI.01178-13 (2013).
Du, T., Zhou, G. & Roizman, B. Induction of apoptosis accelerates reactivation of latent HSV-1 in ganglionic organ cultures and replication in cell cultures. Proc Natl Acad Sci USA 109, 14616–14621, https://doi.org/10.1073/pnas.1212661109 (2012).
Hughes, D. J., Wood, J. J., Jackson, B. R., Baquero-Perez, B. & Whitehouse, A. NEDDylation is essential for Kaposi’s sarcoma-associated herpesvirus latency and lytic reactivation and represents a novel anti-KSHV target. PLoS Pathog 11, e1004771, https://doi.org/10.1371/journal.ppat.1004771 (2015).
Pardo, V. G., Boland, R. & de Boland, A. R. 1alpha,25(OH)(2)-Vitamin D(3) stimulates intestinal cell p38 MAPK activity and increases c-Fos expression. Int J Biochem Cell Biol 38, 1181–1190, https://doi.org/10.1016/j.biocel.2005.12.018 (2006).
Irazoqui, A. P., Boland, R. L. & Buitrago, C. G. Actions of 1,25(OH)2-vitamin D3 on the cellular cycle depend on VDR and p38 MAPK in skeletal muscle cells. J Mol Endocrinol 53, 331–343, https://doi.org/10.1530/JME-14-0102 (2014).
Pan, H., Xie, J., Ye, F. & Gao, S. J. Modulation of Kaposi’s sarcoma-associated herpesvirus infection and replication by MEK/ERK, JNK, and p38 multiple mitogen-activated protein kinase pathways during primary infection. J Virol 80, 5371–5382, https://doi.org/10.1128/JVI.02299-05 (2006).
Xie, J., Ajibade, A. O., Ye, F., Kuhne, K. & Gao, S. J. Reactivation of Kaposi’s sarcoma-associated herpesvirus from latency requires MEK/ERK, JNK and p38 multiple mitogen-activated protein kinase pathways. Virology 371, 139–154, https://doi.org/10.1016/j.virol.2007.09.040 (2008).
Acknowledgements
This work was supported by the funding of Department of Biotechnology (DBT)(BT/PR7044/MED/29/855/2014), Govt. of India and University Grant Commission Govt of India(MRP-MAJOR-BIOT-2013-32241). We are grateful to Prof. David J Blackbourn, University of Surrey, for the kind gift of JSC-1 cell line and Prof. Erle S Robertson, the University of Pennsylvania for providing BC-3 cells. We also like to thank Bhabani Sahoo for his help in confocal microscopy.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: A.K., T.C. Performed the experiments: A.K., S.M., and S.K.S. Analysed the data: S.M., A.K., S.K.S., P.D., T.C. Contributed Reagents/materials/analysis tools: S.R., T.C. Wrote the paper: A.K., T.C.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kumar, A., Mohanty, S., Das, P. et al. 1, 25(OH)2 D3 Induces Reactivation and Death of Kaposi’s Sarcoma-Associated Herpesvirus of Primary Effusion Lymphoma cells. Sci Rep 7, 12438 (2017). https://doi.org/10.1038/s41598-017-12676-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-12676-x
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.