Abstract
We assessed the associations of genetically instrumented blood sucrose with risk of coronary heart disease (CHD) and its risk factors (i.e., type 2 diabetes, adiposity, blood pressure, lipids, and glycaemic traits), using two-sample Mendelian randomization. We used blood fructose as a validation exposure. Dental caries was a positive control outcome. We selected genetic variants strongly (P < 5 × 10–6) associated with blood sucrose or fructose as instrumental variables and applied them to summary statistics from the largest available genome-wide association studies of the outcomes. Inverse-variance weighting was used as main analysis. Sensitivity analyses included weighted median, MR-Egger and MR-PRESSO. Genetically higher blood sucrose was positively associated with the control outcome, dental caries (odds ratio [OR] 1.04 per log10 transformed effect size [median-normalized standard deviation] increase, 95% confidence interval [CI] 1.002–1.08, P = 0.04), but this association did not withstand allowing for multiple testing. The estimate for blood fructose was in the same direction. Genetically instrumented blood sucrose was not clearly associated with CHD (OR 1.01, 95% CI 0.997–1.02, P = 0.14), nor with its risk factors. Findings were similar for blood fructose. Our study found some evidence of the expected detrimental effect of sucrose on dental caries but no effect on CHD. Given a small effect on CHD cannot be excluded, further investigation with stronger genetic predictors is required.
Similar content being viewed by others
Introduction
Sugar, a common component of diet, is known to cause dental caries1, and has been thought to have adverse health effects, particularly on obesity and type 2 diabetes (T2D) for decades2. Whether sugar causes cardiovascular disease (CVD) was heavily debated in the context of whether dietary sugar or fat contributed to the rise in coronary heart disease (CHD) in the mid to late twentieth century in many Western countries3,4. Despite some initial concern about the role of protein5, dietary guidelines since the 1980s have largely focused on promoting a low-fat, high-carbohydrate diet to lower blood cholesterol and prevent CVD5. However, with the upward trend in obesity and T2D6 the role of sugar is being reappraised. In 2015, the World Health Organization (WHO) recommended limiting free sugar intake to less than 10% of total energy intake7.
Observationally, systematic reviews and meta-analyses of prospective cohort studies suggest associations of sugar intake, mostly in the form of sugar sweetened beverages (SSB), with higher risks of CHD8, T2D9, weight gain10,11, hypertension12, and elevated triglycerides, low-density lipoprotein cholesterol (LDL-C) and blood glucose13. However, observational studies, largely from Western settings, may be biased by confounding by socioeconomic position and lifestyle14. In contrast, our previous findings from a birth cohort of Hong Kong Chinese children, where adiposity is not strongly confounded by socio-economic position15, suggested no association of SSB consumption with adiposity16. Randomized controlled trials (RCT) of the effect of sugar intake on CVD or T2D are lacking. Meta-analyses of RCTs on adiposity tend to show a positive effect of sugar or SSB consumption on weight gain in trials of ad libitum diets10,11, but not in trials of isoenergetic exchange of sugar with other carbohydrates10,17. Most of the trials were small and short-term10,11,17, with a meta-analysis possibly biased by publication bias and heterogeneity10. Evidence from RCTs on the effects of sugar intake on blood pressure, lipids, or glycaemic traits is limited and mixed. Some meta-analyses of RCTs have found sugar intake raised blood pressure and lipids18 but others showed no effect on blood pressure19,20 or lipids20,21 when sugars were substituted isoenergetically for other carbohydrates. Adding fructose-containing sugars to the diet appears to have a harmful effect on fasting insulin but not when substituting sugar for other macronutrients with differences by food sources22.
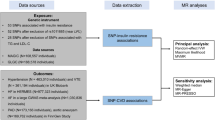
Observationally it is challenging to disentangle the role of a single dietary component, such as sugar, on CVD, while long-term RCTs have not been conducted. In this situation, Mendelian randomization provides an alternative means of examining causal effects by using genetic instruments predicting the exposures to identify effects of single items without confounding. Here, we used two-sample Mendelian randomization23 to estimate the effects of blood sucrose24, which may reflect sucrose intake because blood sucrose is mainly of dietary origin and metabolized very slowly25, on CHD and its risk factors, including T2D, adiposity, blood pressure, lipids, and glycaemic traits using the largest available genome-wide association studies (GWAS). We similarly used blood fructose (one of the two monosaccharides that form sucrose)26 as a validation exposure, which we expected to have directionally similar estimates as sucrose. We did not consider blood glucose as an exposure because blood glucose reflects a number of physiological processes as well as dietary intake of sugar and starch. As sugar is known to cause dental caries1, we used it as a positive control outcome.
Results
We obtained 9 and 2 uncorrelated (r2 < 0.001) single nucleotide polymorphisms (SNP) strongly (P < 5 × 10–6) predicting blood sucrose and fructose, respectively (Supplementary Table 1). Of the 9 SNPs for blood sucrose, only 2, 3 and 8 were available for lipids in the Global Lipids Genetics Consortium (GLGC), glycaemic traits, and blood pressure, respectively, while all were available for other outcomes. We used rs3821975 as a proxy for rs4693441 (r2 = 1.0) for lipids in GLGC and glycaemic traits, but the other SNPs had no proxy, giving 3, 4, and 8 SNPs for lipids in GLGC, glycaemic traits, and blood pressure, and 9 SNPs for all other outcomes. Both of the 2 SNPs for blood fructose were available for all outcomes. The 9 and 2 SNPs explained 6.2% and 0.6% of the variance in blood sucrose and fructose, respectively. The mean F-statistic was 15.7 and 23.6, respectively. None of the SNPs was related to key confounders in the UK Biobank or had known pleiotropic effects according to Ensembl or PhenoScanner.
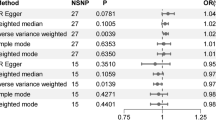
Genetically higher blood sucrose was positively associated with dental caries, but this association did not remain after accounting for multiple testing (Table 1). Genetically higher blood sucrose was suggestively positively associated with decayed and filled tooth surfaces divided by tooth surfaces (DFSS) but not with decayed, missing and filled tooth surfaces (DMFS). A directionally similar estimate with a much wider confidence interval (CI) was found for blood fructose with dental caries (Supplementary Table 2). Genetically instrumented blood sucrose was not significantly associated with CHD but the direction was positive, as it was for fructose (Table 1 and Supplementary Table 2). Genetically instrumented blood sucrose or fructose were not associated with T2D (Table 1 and Supplementary Table 2), adiposity, blood pressure, lipids, or glycaemic traits (Table 2 and Supplementary Table 3) using any method, with estimates in an inverse direction for adiposity, blood pressure and lipids.
Discussion
Our Mendelian randomization study suggests a positive association of genetically higher blood sucrose with dental caries, but this association did not withstand allowing for multiple testing. We found no significant association of genetically predicted sucrose with CHD, but a small effect of sucrose on CHD cannot be excluded. Genetically predicted blood sucrose was not associated with risk of T2D, adiposity, blood pressure, lipids, or glycaemic traits. No associations were found for blood fructose.
Our finding of no significant association of blood sucrose with risk of CHD is inconsistent with some meta-analyses of observational studies suggesting a positive association of SSB with CHD8 or myocardial infarction27 and an observational study suggesting a positive association of sucrose intake with coronary events28. However, the observed associations in Western populations may be confounded by socioeconomic position, diet and lifestyle marked by SSB consumption8,27. In contrast, a Japanese cohort of 39,786 participants followed up for 18 years suggested no association of soft drink consumption with CHD29. Nevertheless, our finding of a non-significant positive association might be owing to inadequate power. Further Mendelian Randomization studies with stronger instruments for sugar intake or biomarkers would be very helpful about the specific role of sugar. More broadly genetic evidence seems to be emerging suggesting that protein rather than fat or sugar is the key dietary element affecting health30.
No association was found of genetically predicted blood sucrose or fructose with T2D, which is consistent with a meta-analysis of observational studies of sugar or fructose intake31 but not with others suggesting positive associations of SSB with T2D9,32. However, the studies of SSB also found positive associations of artificially sweetened beverages with T2D9,32, implying that the associations might be due to confounding rather than to sugar itself. The null association with adiposity is concordant with our previous finding of SSB with adiposity in Hong Kong Chinese children16, but inconsistent with positive associations of sugar or SSB intake with weight gain in observational studies in Western populations10,11. The finding is more consistent with meta-analyses of RCTs showing no effect of substituting sugar for other carbohydrates on weight10,17,20. Sucrose or fructose intake, particularly in the form of SSB, may cause adiposity by adding to extra energy intake but inducing less satiety33 thus promoting positive energy balance. However, an experimental study in mice found no difference in adiposity by sucrose intake but did find differences by fat intake34, suggesting that fat rather than sucrose drives over-consumption. Sensitivity analyses suggested a possible inverse association of blood sucrose with BMI, which might be chance or because sucrose might be at the expense of a lower fat intake. The lack of associations with blood pressure or lipids is consistent with meta-analyses of trials substituting sugar for complex carbohydrates on systolic blood pressure (SBP) and diastolic blood pressure (DBP)20 and of trials substituting fructose for glucose on triglyceride35, but inconsistent with another showing positive effects of sugar intake on blood pressure and all lipids with or without energy control18. However, our MR findings differ from observed positive associations of SSB with hypertension12, triglycerides and LDL-C13. The lack of associations with glycaemic traits is consistent with a meta-analysis of RCTs showing no effect of sugar intake on fasting insulin when substituting sugar for other macronutrients or subtracting sugar from the diet22. Nevertheless, most of the trials had small sample sizes and short duration18,20,22,35, making the findings less certain.
Sugar may cause CVD through several mechanisms. First, sugar may promote CVD through its effect on weight gain. Observational studies have suggested a weakened association of SSB with CHD after adjusting for body mass index (BMI) and energy intake8. However, while sugar at high doses in hypercaloric trials did increase weight17, sugar at usual intake especially as part of an isocaloric diet showed no effect on weight gain10,17. Second, sugar may cause CVD through metabolic effects of fructose. Unlike glucose, fructose can bypass phosphofructokinase and be metabolised to substrates for lipid synthesis in unregulated glycolysis36, thus leading to increased de novo lipogenesis, dyslipidaemia, and insulin resistance after excess intake36. Chronic fructose intake may activate gene expression of lipogenic enzymes to further enhance lipogenesis36. Unregulated fructose metabolism can also raise uric acid concentration, which has been linked to endothelial dysfunction, hypertension, and increased risk of CVD33. It is also possible that sugar intake might affect glucagon or insulin which may cause CHD37,38. However, we did not find any evidence of these pathways, which need validation using stronger instruments predicting sugar.
Our study has some limitations. First, we used genetic variants predicting sucrose and fructose from metabolomics studies using mass spectrometry24,26. Blood sucrose and fructose may not be perfect biomarkers of sugar intake. However, blood sucrose is mainly of dietary origin and metabolized very slowly25, so blood sucrose reflects sucrose intake. High fructose corn syrup (42% or 55% fructose, glucose and water) is another commonly used sweetener other than sucrose, which may be captured by blood fructose. Many other factors, including intestinal permeability and liver metabolism39, may also influence blood levels of sucrose and fructose, but are unlikely to confound the genetic associations. Second, we used the genetic variants associated with blood sucrose and fructose at less than genome-wide significance (P < 5 × 10–6) and the variance of sucrose and fructose explained by the genetic variants were small (6.2% and 0.6%). Weak instrument bias could attenuate the causal estimates towards the null23. However, the SNPs had a mean F-statistic > 10. Weak instrument bias is expected to be small if the p-value is below 1 × 10–540. Third, confounding by population stratification is possible. However, the genetic associations concern people of European ancestry generated with genomic control. The GWAS of blood sucrose suggested no inflation or deflation due to population stratification24. The genetic variants were not related to key confounders in the UK Biobank. Fourth, genetic predictors of sucrose and fructose may have pleiotropic effects. However, we found no unknown pleiotropy from MR-Egger and MR-PRESSO and no known pleiotropy in Ensembl or PhennoScanner. All methods gave similar results. Fifth, our study was mainly limited to people of European ancestry and might not apply to other populations. However, causes would be expected to be consistent41, although might not always be relevant42. Sixth, the instruments predicting blood sucrose were obtained from primarily women, which may not be applicable to men. However, there is no evidence that genetic predisposition to blood sucrose or fructose differs by sex or has different effects by sex. The estimates for blood sucrose or fructose with dental caries, BMI and blood pressure in women were similar to those in both sexes. Seventh, we could not check whether the associations varied by age, or for non-linear effects. Eighth, canalization, i.e., compensatory mechanisms driving the association of genetic variants with sucrose or fructose, may result in different associations in Mendelian randomization than would be observed from interventions. However, whether that exists is unknown. Finally, although the estimates were close to the null, we cannot rule out a small effect for CHD, which should be investigated further. We only have 80% power to detect an OR of about 1.05 for sucrose on CHD. However, the estimates for adiposity, blood pressure and lipids were in the opposite direction to those expected.
Conclusions
Our study found some evidence of a detrimental effect of blood sucrose on dental caries. However, our study suggests no effect of blood sucrose on T2D, adiposity, blood pressure, lipids, or glycaemic traits, whilst it remains possible that there could be a small effect of sucrose on CHD risk.
Methods
Genetic instruments for blood sucrose and fructose
Genetic variants, i.e., SNP, strongly (P < 5 × 10–6) predicting serum sucrose (log10 transformed median-normalized values) were extracted from an existing GWAS of serum metabolites adjusted for sex and age, obtained from non-targeted metabolomics using four independent ultra-high-performance liquid chromatography-tandem mass spectrometry24, in 1439 individuals (median age 58 years, 96.6% women) of European ancestry24. These associations were not adjusted for principal components because there was no evidence of inflation or deflation due to population stratification24. SNPs strongly (P < 5 × 10–6) predicting blood fructose (log10 transformed normalized) were extracted from an existing GWAS of blood metabolites adjusted for age, sex, batch and genomic control26, obtained using liquid-phase chromatography, gas chromatography separation and tandem mass spectrometry26 in 7824 individuals (mean age 55 years, 83.5% women) of European ancestry26. SNPs were discarded if they were non-biallelic or had a minor allele frequency less than 0.01.
Genetic associations with dental caries, CHD, and T2D
Genetic associations with dental caries (ncase = 2110, ncontrol = 359,084) were extracted from genetic summary statistics from the UK Biobank in ~ 361,194 white British individuals (mean age 57.5 years)43 provided by Neale Lab (http://www.nealelab.is/uk-biobank/). Dental caries was defined as hospital inpatient admission due to International Classification of Diseases (ICD) 10 K02. These genetic associations were obtained from linear regresstion adjusted for age, age2, sex, interactions of sex with age and age2, and 20 principal components. The linear regression estimates were transformed to log odds ratios (OR) using an approximation44. Genetic associations with DFSS (inverse normal rank transformed) and DMFS (standard deviation of residuals) were taken from summary statistics from the Gene-Lifestyle Interactions in Dental Endpoints (GLIDE) consortium in up to 26,792 individuals of European ancestry adjusted for age, sex, and study-specific covariates45. Genetic associations with CHD were taken from summary statistics of the Coronary Artery DIsease Genome wide Replication and Meta-analysis plus Coronary Artery Disease (CARDIoGRAMplusC4D) consortium from a meta-analysis (ncase = ~ 76,014, ncontrol = ~ 264,785) of CARDIoGRAMplusC4D 1000 Genomes, the Myocardial Infarction Genetics and CARDIoGRAM Exome, the UK Biobank SOFT CAD GWAS, and two small case–control studies in individuals of mainly European ancestry (77% in CARDIoGRAMplusC4D 1000 Genomes and 94% in the UK Biobank SOFT CAD GWAS)46 with genetic control. CHD cases included fatal or nonfatal myocardial infarction, percutaneous transluminal coronary angioplasty, coronary artery bypass grafting, chronic ischemic heart disease and angina determined from clinical diagnosis, medical records and self-report of medication usage. Controls were those who were not cases. Genetic associations with T2D were obtained from summary statistics from the DIAbetes Genetics Replication And Meta-analysis (DIAGRAM) consortium from a meta-analysis of case (n = 74,124)-control (n = 824,006) studies47 in individuals of European ancestry adjusted for study-specific covariates and population structure but not BMI.
Genetic associations with adiposity, blood pressure, lipids, and glycaemic traits
Genetic associatons with inverse-normally transformed BMI were obtained from summary statitsics from a meta-analysis of the Genetic Investigation of ANthropometric Traits (GIANT) Consortium and the UK Biobank in up to 806,834 individuals of European ancestry adjusted for age, age2, assessment centre, and sex48. Genetic associations with inverse-rank normalized waist circumference (WC), body fat percentage (BFP), high-density lipoprotein cholesterol (HDL-C), LDL-C, and triglyceride were obtained from UK Biobank summary statitsics43 provided by Neale Lab (http://www.nealelab.is/uk-biobank/) in ~ 361,194 white British individuals adjusted for age, age2, and 20 principal components, sex and interactions of sex with age and age2. Genetic associations with inverse-normally transformed HDL-C, LDL-C, and triglyceride were also taken from GLGC in ~ 188,578 individuals of European ancestry adjusted for age, age2, and sex49. Genetic associations with SBP and DBP were obtained from a meta-analysis of the UK Biobank and the International Consortium of Blood Pressure-Genome Wide Association Studies (ICBP) in ~ 757,601 individuals of European ancestry adjusted for sex, age, age2, BMI, and genotyping chips50. Genetic associations with glycaemic traits, including indices of β-cell function (HOMA-B) and insulin resistance (HOMA-IR), and fasting insulin (all naturally log transformed), were taken from a meta-analysis of GWAS including the UK Biobank in up to 46,186 individuals of European ancestry adjusted for age, sex and study specific covariates with genomic control51 provided by the Meta-Analyses of Glucose and Insulin-related traits Consortium (MAGIC).
Statistical analysis
We obtained uncorrelated SNPs (r2 ≤ 0.001) using the “ld_clump” function of “ieugwasr” R package. For SNPs not available for an outcome, we used proxies (r2 ≥ 0.8) identified from LDlink (https://analysistools.nci.nih.gov/LDlink/). We aligned genetic variants for exposure and outcome on effect allele as none were palindromic. We estimated the F-statistic for each SNP using an approximation52, where a F-statistic of 10 or above indicated a lower risk of weak instrument bias. We checked whether any of the SNPs were associated with key confounders (i.e., Townsend deprivation index, education, smoking, alcohol drinking, and moderate physical activity) in the UK Biobank at Bonferroni corrected significance (0.05 divided by the number of SNPs times the number of confounders) (http://www.nealelab.is/uk-biobank). We checked for known pleiotropic effects using two curated genotype-to-phenotype cross-references, Ensembl 99 (http://www.ensembl.org/index.html, accessed April 15, 2020), and PhenoScanner (http://www.phenoscanner.medschl.cam.ac.uk, accessed April 15, 2020).
We obtained Mendelian randomization estimates by meta-analysing SNP-specific Wald estimates (ratio of SNP on outcome to SNP on exposure)23 using inverse variance weighting (IVW) with multiplicative random effects which allows for heterogeneity but assumes balanced pleiotropy. We also used a weighted median which is valid as long as < 50% of the weight are contributed by invalid SNPs53, and MR-Egger which is valid as long as the instrument strength independent of direct effect assumption (i.e., the size of the pleiotropic effects is independent of their strength as instruments) holds52. MR-Egger provides a check on the validity of the IVW estimate, with a significant p-value (< 0.05) for the intercept indicating that the IVW estimate may be invalid52. We also used the Mendelian randomization pleiotropy residual sum and outlier (MR-PRESSO) test which can correct for potential horizontal pleiotropy (i.e., the variant has an effect on disease outside of its effect on the exposure)54.
We estimated the expected effect size of the association of blood sucrose with CHD for 80% power. At an R2 (the proportion of variance in blood sucrose explained by the genetic variants) of 6.2%, we have 80% power to detect an OR of ∼1.05 for sucrose on CHD. Here, we are testing a hypothesis, the effect on CHD with related secondary outcomes (CHD risk factors) and a control outcome (dental caries), for comprehensiveness we considered a p-value of 0.05 and a Bonferroni-corrected p-value of 0.05/7 = 0.007 (given 7 outcomes including CHD, 5 CHD risk factors and dental caries) as significant. All statistical analyses were performed using R version 3.6.3 (The R Foundation for Statistical Computing, Vienna, Austria)55 and the “MendelianRandomization” and “MRPRESSO” R packages. This study requires no ethical approval because only publicly available summary data was used.
Data availability
The data are available in the consortia.
Abbreviations
- BFP:
-
Body fat percentage
- BMI:
-
Body mass index
- CARDIoGRAMplusC4D:
-
The Coronary Artery DIsease Genome wide Replication and Meta-analysis plus Coronary Artery Disease consortium
- CHD:
-
Coronary heart disease
- CI:
-
Confidence interval
- DBP:
-
Diastolic blood pressure
- DFSS:
-
Decayed and filled tooth surfaces divided by tooth surfaces
- DIAGRAM:
-
The DIAbetes Genetics Replication And Meta-analysis consortium
- DMFS:
-
Decayed, missing and filled tooth surfaces
- GLIDE:
-
The Gene-Lifestyle Interactions in Dental Endpoints consortium
- GLGC:
-
The Global Lipids Genetics Consortium
- GWAS:
-
Genome-wide association study
- HDL-C:
-
High-density lipoprotein cholesterol
- ICBP:
-
International Consortium of Blood Pressure-Genome Wide Association Studies
- ICD:
-
International Classification of Diseases
- IVW:
-
Inverse variance weighting
- LDL-C:
-
Low-density lipoprotein cholesterol
- MAGIC:
-
The Meta-Analyses of Glucose and Insulin-related traits Consortium
- OR:
-
Odds ratio
- RCT:
-
Randomized controlled trial
- SBP:
-
Systolic blood pressure
- SD:
-
Standard deviation
- SNP:
-
Single nucleotide polymorphism
- SSB:
-
Sugar-sweetened beverage
- T2D:
-
Type 2 diabetes
- WC:
-
Waist circumference
- WHO:
-
World Health Organization
References
Moynihan, P. J. & Kelly, S. A. Effect on caries of restricting sugars intake: Systematic review to inform WHO guidelines. J. Dent. Res. 93, 8–18. https://doi.org/10.1177/0022034513508954 (2014).
Yudkin, J. Pure, White and Deadly: How Sugar is Killing Us and What We Can Do to Stop It (Penguin, New York, 1988).
Yudkin, J. Patterns and trends in carbohydrate consumption and their relation to disease. Proc. Nutr. Soc. 23, 149–162. https://doi.org/10.1079/pns19640028 (1964).
Keys, A. Diet and the epidemiology of coronary heart disease. J. Am. Med. Assoc. 164, 1912–1919. https://doi.org/10.1001/jama.1957.62980170024007e (1957).
Oppenheimer, G. M. & Benrubi, I. D. McGovern’s Senate Select Committee on Nutrition and Human Needs versus the meat industry on the diet-heart question (1976–1977). Am. J. Public Health 104, 59–69. https://doi.org/10.2105/ajph.2013.301464 (2014).
Geiss, L. S. et al. Prevalence and incidence trends for diagnosed diabetes among adults aged 20 to 79 years, United States, 1980–2012. JAMA 312, 1218–1226. https://doi.org/10.1001/jama.2014.11494 (2014).
World Health Organization. Guideline: Sugars Intake for Adults and Children (World Health Organization, Geneva, 2015).
Huang, C., Huang, J., Tian, Y., Yang, X. & Gu, D. Sugar sweetened beverages consumption and risk of coronary heart disease: A meta-analysis of prospective studies. Atherosclerosis 234, 11–16. https://doi.org/10.1016/j.atherosclerosis.2014.01.037 (2014).
Imamura, F. et al. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: Systematic review, meta-analysis, and estimation of population attributable fraction. Br. J. Sports Med. 50, 496–504. https://doi.org/10.1136/bjsports-2016-h3576rep (2016).
Te Morenga, L., Mallard, S. & Mann, J. Dietary sugars and body weight: Systematic review and meta-analyses of randomised controlled trials and cohort studies. BMJ (Clinical research ed.) 346, e7492. https://doi.org/10.1136/bmj.e7492 (2012).
Malik, V. S., Pan, A., Willett, W. C. & Hu, F. B. Sugar-sweetened beverages and weight gain in children and adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 98, 1084–1102. https://doi.org/10.3945/ajcn.113.058362 (2013).
Kim, Y. & Je, Y. Prospective association of sugar-sweetened and artificially sweetened beverage intake with risk of hypertension. Arch. Cardiovasc. Dis. 109, 242–253. https://doi.org/10.1016/j.acvd.2015.10.005 (2016).
Keller, A., Heitmann, B. L. & Olsen, N. Sugar-sweetened beverages, vascular risk factors and events: A systematic literature review. Public Health Nutr. 18, 1145–1154. https://doi.org/10.1017/s1368980014002122 (2015).
Ioannidis, J. P. A. The challenge of reforming nutritional epidemiologic research. JAMA 320, 969–970. https://doi.org/10.1001/jama.2018.11025 (2018).
Schooling, C. M., Yau, C., Cowling, B. J., Lam, T. H. & Leung, G. M. Socio-economic disparities of childhood Body Mass Index in a newly developed population: Evidence from Hong Kong’s “Children of 1997” birth cohort. Arch. Dis. Child. 95, 437–443. https://doi.org/10.1136/adc.2009.168542 (2010).
Zhang, T. et al. Association of sugar-sweetened beverage frequency with adiposity: Evidence from the “Children of 1997” Birth Cohort. Nutrients. https://doi.org/10.3390/nu12041015 (2020).
Sievenpiper, J. L. et al. Effect of fructose on body weight in controlled feeding trials: A systematic review and meta-analysis. Ann. Intern. Med. 156, 291–304. https://doi.org/10.7326/0003-4819-156-4-201202210-00007 (2012).
Te Morenga, L. A., Howatson, A. J., Jones, R. M. & Mann, J. Dietary sugars and cardiometabolic risk: Systematic review and meta-analyses of randomized controlled trials of the effects on blood pressure and lipids. Am. J. Clin. Nutr. 100, 65–79. https://doi.org/10.3945/ajcn.113.081521 (2014).
Ha, V. et al. Effect of fructose on blood pressure: A systematic review and meta-analysis of controlled feeding trials. Hypertension 59, 787–795. https://doi.org/10.1161/hypertensionaha.111.182311 (2012).
Fattore, E., Botta, F., Agostoni, C. & Bosetti, C. Effects of free sugars on blood pressure and lipids: A systematic review and meta-analysis of nutritional isoenergetic intervention trials. Am. J. Clin. Nutr. 105, 42–56. https://doi.org/10.3945/ajcn.116.139253 (2017).
Chiavaroli, L. et al. Effect of fructose on established lipid targets: A systematic review and meta-analysis of controlled feeding trials. J. Am. Heart Assoc. 4, e001700. https://doi.org/10.1161/jaha.114.001700 (2015).
Choo, V. L. et al. Food sources of fructose-containing sugars and glycaemic control: Systematic review and meta-analysis of controlled intervention studies. BMJ 363, k4644. https://doi.org/10.1136/bmj.k4644 (2018).
Burgess, S., Scott, R. A., Timpson, N. J., Davey Smith, G. & Thompson, S. G. Using published data in Mendelian randomization: A blueprint for efficient identification of causal risk factors. Eur. J. Epidemiol. 30, 543–552. https://doi.org/10.1007/s10654-015-0011-z (2015).
Long, T. et al. Whole-genome sequencing identifies common-to-rare variants associated with human blood metabolites. Nat. Genet. 49, 568–578. https://doi.org/10.1038/ng.3809 (2017).
Menzies, I. S. Absorption of intact oligosaccharide in health and disease. Biochem. Soc. Trans. 2, 1042–1047. https://doi.org/10.1042/bst0021042 (1974).
Shin, S. Y. et al. An atlas of genetic influences on human blood metabolites. Nat. Genet. 46, 543–550. https://doi.org/10.1038/ng.2982 (2014).
Narain, A., Kwok, C. S. & Mamas, M. A. Soft drinks and sweetened beverages and the risk of cardiovascular disease and mortality: A systematic review and meta-analysis. Int. J. Clin. Pract. 70, 791–805. https://doi.org/10.1111/ijcp.12841 (2016).
Warfa, K., Drake, I., Wallstrom, P., Engstrom, G. & Sonestedt, E. Association between sucrose intake and acute coronary event risk and effect modification by lifestyle factors: Malmo Diet and Cancer Cohort Study. Br. J. Nutr. 116, 1611–1620. https://doi.org/10.1017/s0007114516003561 (2016).
Eshak, E. S. et al. Soft drink intake in relation to incident ischemic heart disease, stroke, and stroke subtypes in Japanese men and women: The Japan Public Health Centre-based study cohort I. Am. J. Clin. Nutr. 96, 1390–1397. https://doi.org/10.3945/ajcn.112.037903 (2012).
Meddens, S. F. W. et al. Genomic analysis of diet composition finds novel loci and associations with health and lifestyle. Mol. Psychiatry https://doi.org/10.1038/s41380-020-0697-5 (2020).
Tsilas, C. S. et al. Relation of total sugars, fructose and sucrose with incident type 2 diabetes: A systematic review and meta-analysis of prospective cohort studies. CMAJ 189, E711-e720. https://doi.org/10.1503/cmaj.160706 (2017).
Greenwood, D. C. et al. Association between sugar-sweetened and artificially sweetened soft drinks and type 2 diabetes: Systematic review and dose-response meta-analysis of prospective studies. Br. J. Nutr. 112, 725–734. https://doi.org/10.1017/s0007114514001329 (2014).
Johnson, R. J. et al. Potential role of sugar (fructose) in the epidemic of hypertension, obesity and the metabolic syndrome, diabetes, kidney disease, and cardiovascular disease. Am. J. Clin. Nutr. 86, 899–906. https://doi.org/10.1093/ajcn/86.4.899 (2007).
Hu, S. et al. Dietary fat, but not protein or carbohydrate, regulates energy intake and causes adiposity in mice. Cell Metab. https://doi.org/10.1016/j.cmet.2018.06.010 (2018).
Livesey, G. & Taylor, R. Fructose consumption and consequences for glycation, plasma triacylglycerol, and body weight: Meta-analyses and meta-regression models of intervention studies. Am. J. Clin. Nutr. 88, 1419–1437. https://doi.org/10.3945/ajcn.2007.25700 (2008).
Herman, M. A. & Samuel, V. T. The sweet path to metabolic demise: Fructose and lipid synthesis. Trends Endocrinol. Metab. 27, 719–730. https://doi.org/10.1016/j.tem.2016.06.005 (2016).
Ng, J. C. M. & Schooling, C. M. Effect of glucagon on ischemic heart disease and its risk factors: A Mendelian randomization study. J. Clin. Endocrinol. Metab. https://doi.org/10.1210/clinem/dgaa259 (2020).
Zhao, J. V., Luo, S. & Schooling, C. M. Sex-specific Mendelian randomization study of genetically predicted insulin and cardiovascular events in the UK Biobank. Commun. Biol. 2, 332. https://doi.org/10.1038/s42003-019-0579-z (2019).
Sundborn, G. et al. Are liquid sugars different from solid sugar in their ability to cause metabolic syndrome?. Obesity (Silver Spring, Md.) 27, 879–887. https://doi.org/10.1002/oby.22472 (2019).
Burgess, S., Butterworth, A. & Thompson, S. G. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet. Epidemiol. 37, 658–665. https://doi.org/10.1002/gepi.21758 (2013).
Rothman, K. J., Gallacher, J. E. & Hatch, E. E. Why representativeness should be avoided. Int. J. Epidemiol. 42, 1012–1014. https://doi.org/10.1093/ije/dys223 (2013).
Lopez, P., Subramanian, S. V. & Schooling, C. M. Effect measure modification conceptualized using selection diagrams as medication by mechanisms of varying population-level relevance. J. Clin. Epidemiol. https://doi.org/10.1016/j.jclinepi.2019.05.005 (2019).
Bycroft, C. et al. The UK Biobank resource with deep phenotyping and genomic data. Nature 562, 203–209. https://doi.org/10.1038/s41586-018-0579-z (2018).
Lloyd-Jones, L. R., Robinson, M. R., Yang, J. & Visscher, P. M. Transformation of summary statistics from linear mixed model association on all-or-none traits to odds ratio. Genetics 208, 1397–1408. https://doi.org/10.1534/genetics.117.300360 (2018).
Shungin, D. et al. Genome-wide analysis of dental caries and periodontitis combining clinical and self-reported data. Nat. Commun. 10, 2773. https://doi.org/10.1038/s41467-019-10630-1 (2019).
Nelson, C. P. et al. Association analyses based on false discovery rate implicate new loci for coronary artery disease. Nat. Genet. 49, 1385–1391. https://doi.org/10.1038/ng.3913 (2017).
Mahajan, A. et al. Fine-mapping type 2 diabetes loci to single-variant resolution using high-density imputation and islet-specific epigenome maps. Nat. Genet. 50, 1505–1513. https://doi.org/10.1038/s41588-018-0241-6 (2018).
Pulit, S. L. et al. Meta-analysis of genome-wide association studies for body fat distribution in 694,649 individuals of European ancestry. Hum. Mol. Genet. 28, 166–174. https://doi.org/10.1093/hmg/ddy327 (2019).
Willer, C. J. et al. Discovery and refinement of loci associated with lipid levels. Nat. Genet. 45, 1274–1283. https://doi.org/10.1038/ng.2797 (2013).
Evangelou, E. et al. Genetic analysis of over 1 million people identifies 535 new loci associated with blood pressure traits. Nat. Genet. 50, 1412–1425. https://doi.org/10.1038/s41588-018-0205-x (2018).
Dupuis, J. et al. New genetic loci implicated in fasting glucose homeostasis and their impact on type 2 diabetes risk. Nat. Genet. 42, 105–116. https://doi.org/10.1038/ng.520 (2010).
Bowden, J. et al. Assessing the suitability of summary data for two-sample Mendelian randomization analyses using MR-Egger regression: the role of the I2 statistic. Int. J. Epidemiol. 45, 1961–1974. https://doi.org/10.1093/ije/dyw220 (2016).
Burgess, S. & Thompson, S. G. Interpreting findings from Mendelian randomization using the MR-Egger method. Eur. J. Epidemiol. 32, 377–389. https://doi.org/10.1007/s10654-017-0255-x (2017).
Verbanck, M., Chen, C. Y., Neale, B. & Do, R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat. Genet. 50, 693–698. https://doi.org/10.1038/s41588-018-0099-7 (2018).
R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. (2020).
Acknowledgements
Data on DFSS and DMFS have been contributed by the GLIDE consortium (https://data.bris.ac.uk/data/dataset/2j2rqgzedxlq02oqbb4vmycnc2). Data on CHD have been contributed by the CARDIoGRAMplusC4D consortium (http://www.cardiogramplusc4d.org). Data on T2D have been contributed by the DIAGRAM Consortium (http://www.diagram-consortium.org). Data on BMI have been contributed by the GIANT Consortium (http://portals.broadinstitute.org/collaboration/giant/index.php/GIANT_consortium_data_files). Data on dental caries and lipids have been contributed by the UK Biobank (http://www.nealelab.is/uk-biobank). Additional data on lipids have been contributed by GLGC. Data on blood pressure have been contributed by ICBP. Data on glycaemic traits have been contributed by the MAGIC (www.magicinvestigators.org). This study was partly funded by the Health and Medical Research Fund, Food and Health Bureau, HKSAR Government, Hong Kong, China (CFS-HKU1). The funder had no role in the design, analyses, interpretation of results or writing of the paper.
Author information
Authors and Affiliations
Contributions
C.M.S. conceptualized the study. T.Z. conducted the analysis and drafted the manuscript. C.M.S. directed the analytic strategy and supervised the study. C.M.S. and S.L.A.Y. critically reviewed the manuscript. All authors approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, T., Au Yeung, S. & Schooling, C.M. Association of genetically predicted blood sucrose with coronary heart disease and its risk factors in Mendelian randomization. Sci Rep 10, 21588 (2020). https://doi.org/10.1038/s41598-020-78685-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-78685-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.