Abstract
We investigated the prognostic influences of BCL1 and BCL2 expression on disease-free survival in breast cancer patients. BCL1 and BCL2 expression statuses were assessed by immunohistochemistry using tissue microarrays from 393 breast cancer patients. The Kaplan–Meier estimator and log-rank test were used for survival analyses. The Cox proportional hazards model was used to calculate hazard ratio (HR) and the 95% confidence interval (CI) of survival analyses. BCL1 expression revealed no impact on survival. The high BCL2 group showed superior disease-free survival compared with the low BCL2 group (p = 0.002), especially regarding local recurrence-free survival (p = 0.045) and systemic recurrence-free survival (p = 0.002). BCL2 expression was a significant prognostic factor by univariable analysis (HR, 0.528; 95% CI, 0.353–0.790; p = 0.002) and by multivariable analysis (HR, 0.547; 95% CI, 0.362–0.826; p = 0.004). High BCL2 expression was associated with higher disease-free survival in the hormone receptor (HRc)-positive and human epidermal growth factor receptor 2 (HER2)-negative (HRc(+)/HER2(−)) subtype only (p = 0.002). The high BCL2 group was associated with positive estrogen receptor (ER), positive progesterone receptor (PR), low histologic grade, and age ≤ 50 years. BCL1 expression had no prognostic impact, but BCL2 expression was a significant independent prognostic factor. High BCL2 expression was associated with higher disease-free survival, especially regarding local recurrence and systemic recurrence. The prognostic effect of BCL2 expression was effective only in the HRc(+)/HER2(−) subtype. Favorable clinicopathologic features and a strong association with the ER/PR status could partly explain the superior prognosis of the high BCL2 group. BCL2 expression could be utilized to assess the prognosis of breast cancer patients in clinical settings.
Similar content being viewed by others
Introduction
Both BCL1 and BCL2 are known to have oncogenic activities in various human malignancies and are key proteins in the induction of cellular proliferation and cancer progression1,2,3,4,5,6. Previous studies have investigated the prognostic roles of BCL1 and BCL2 in various human cancers including breast cancer. Although BCL1 and BCL2 were expected to be adverse prognostic indicators in breast cancer, the results have been largely inconsistent.
BCL1 is reported to be a major oncogene in various human solid cancers1,2,3,4. BCL1 is located on chromosome 11q13.3 and is also known as CCND1, cyclin D1, and PRAD1. The BCL1 protein, otherwise known as cyclin D1, is one of the D-type cyclins and is a crucial protein for cell cycle regulation, especially in the process of the G0/S transition. BCL1 overexpression, which is frequently observed in human malignancies, induces shortening of the G0/S transition time, activating rapid cell growth and neoplastic growth1,2,3,4.
BCL2 is also regarded to be an oncogene in various malignancies, especially in leukemia and lymphoma, and is a key factor for the regulation of cellular apoptosis. BCL2 is located on chromosome 18q21 and encodes an integral outer mitochondrial membrane protein that blocks the apoptotic death of cells. BCL2 overexpression inhibits apoptotic cell death and activates cellular proliferation and tumor progression5,6. BCL1 and BCL2 also reportedly interact: BCL2 overexpression induces BCL1 expression in human breast epithelial cells7.
Although both BCL1 and BCL2 are predicted to be associated with an adverse prognosis in breast cancer, previous studies have reported inconsistent results. Some studies reported the adverse effect of BCL1 on breast cancer prognosis8,9,10, whereas others reported no association between BCL1 expression and survival11,12. Furthermore, a favorable prognostic impact of BCL1 was also described13,14,15. BCL2 is also believed to have an adverse influence on breast cancer survival, but most previous studies have reported a favorable prognostic effect16,17,18,19,20. However, some studies reported no association between BCL2 expression and survival21, and several studies reported that BCL2 is an adverse prognostic indicator22.
This study investigates the prognostic effects of BCL1 and BCL2 expression in breast cancer by analyzing tissue microarray data of primary breast cancers. The association of BCL1 and BCL2 expression with various clinicopathologic features as well as different breast cancer subtypes was also investigated.
Methods
Study subjects
Primary invasive breast cancer patients have been prospectively registered in the Boramae Hospital Breast Cancer Registry. In 2012, we made tissue microarrays using cancer tissues from 420 registered patients23. Among these, 13 patients were excluded due to insufficient information from immunostaining results. Three patients were further excluded because of duplication. An additional 11 patients, who were initially diagnosed as stage IV, were also excluded. Therefore, 393 operable primary breast cancer patients were enrolled in this study. This study was approved by the Institutional Review Boards of the Seoul Metropolitan Government Seoul National University Boramae Medical Center (approval number: 16-2016-82). All methods including data acquisition and analysis were performed in compliance with protocols approved by the Institutional Review Boards. Written informed consent was obtained from all participants prior to the study.
Preparation of tissue microarrays and immunohistochemistry of BCL1 and BCL2
Tissue microarrays were constructed with 2 mm diameter cores of representative tumor areas from formalin-fixed paraffin-embedded tissue blocks. Immunohistochemical staining was performed using rabbit anti-Cyclin D1 (SP4-R) monoclonal antibody (ready-to-use format, Ventana-Roche Diagnostics, Indianapolis, IN) for BCL1 and mouse anti-BCL2 (Clone 124) monoclonal antibody (1:200, DAKO-Agilent Technologies, Santa Clara, CA) for BCL2.
BCL1 staining was not detected in normal breast cancer tissues (Fig. S1A). BCL1 showed heterogeneous nuclear staining in cases with positive staining of BCL1 in breast cancer tissues (Fig. S1C). BCL2 staining was detected in normal breast cancer tissues (Fig. S1D). BCL2 showed homogenous cytoplasmic staining both in normal breast tissues (Fig. S1D) and breast cancer tissues with positive BCL2 staining (Fig. S1F). BCL2 staining was detected in terminal duct lobular units and reactive lymphocytes but not in the stromal cells of normal or cancer tissues (Fig. S1D, F). Staining results were evaluated based on the proportion and average intensity of nuclear staining for BCL1 and cytoplasmic staining for BCL2 in stained tumor cells. Proportion scores were dichotomously classified as follows: ≤ 10% of tumor cells vs. > 10% of tumor cells. Intensity scores were scored as follows: 0, negative; 1, weak; 2, moderate; 3, strong (Figs. S2 and S3). The Allred score was calculated as the sum of the proportion score and the intensity score, according to previous reports24,25.
Defining low vs. high expression of BCL1 and BCL2
Significant survival differences for BCL1 were not observed at any cut-off values of the Allred scores (Fig. S4). In this study, high BCL1 expression was designated when the Allred score was greater than 6 because the distribution of subject numbers into two groups was most even under this condition. For BCL2, the high BCL2 group showed significantly superior survival compared with the low BCL2 group when cut-off values for the Allred scores were set at 2 (p = 0.002), 3 (p = 0.001), and 4 (p = 0.009) (Fig. S5). In this study, high BCL2 expression was defined when the Allred score was greater than 2 because the distribution of subject numbers was most even under this criterion. Significant survival differences were not observed with regards to either the intensity score or the proportion score in terms of BCL1. The high BCL2 group revealed better survival with regards to both the BCL2 intensity (p = 0.002; intensity score 1, 2, 3 vs. 0) score and proportion score (p = 0.002; proportion score > 10% vs. ≤ 10%) (Fig. S6).
Definition of variables
The stage of the cancer was described according to the 8th edition of the American Joint Committee on Cancer. The status of the estrogen receptor (ER) and the progesterone receptor (PR) was defined based on the result of the immunohistochemical test25,26. The human epidermal growth factor receptor 2 (HER2) status was defined as positive or negative based on the immunohistochemical results and the in situ hybridization test27. The hormone receptor (HRc) status was defined as positive when the immunohistochemical test for either ER or PR was positive. Breast cancer subtypes were classified into four groups according to HRc and HER2: HRc(+)/HER2(−), HRc(+)/HER2(+), HRc(−)/HER2(+), and HRc(−)/HER2(−). Histologic grade followed the modified Scarff-Bloom-Richardson grading system. Body mass index was defined as the ratio of body weight in kilograms to height in square meters.
Statistical analyses
A two-sample t-test was used to determine statistical differences between mean ages, and Pearson’s χ2 test was used to determine statistical differences between all other baseline characteristics. Recurrence was defined as any first event of local, regional, systemic, and/or contralateral breast cancer relapse. Time duration for disease-free survival was defined as the time difference from the operation to any recurrence. The Kaplan–Meier estimator was used to analyze survival rates, and a log-rank test was used to determine the significance of differences between survival curves. The Cox proportional hazards model was used to calculate the hazard ratio (HR) and 95% confidence interval (CI). All statistical analyses were carried out using IBM SPSS Statistics, version 26.0 (IBM Corp., Armonk, NY). All tests were two-sided, and statistical significance was defined as when the p value was less than 0.05.
Results
Baseline characteristics of study subjects
The mean age of the 393 study subjects was 53.4 ± 12.3 years (median, 51.0 years; range, 25–87 years). Operation dates occurred between July 1999 and April 2012. The mean durations for post-operative disease-free survival were 87.5 ± 50.3 months (median, 90.0 months; range, 1–216 months). There were 103 recurrences, and the numbers of local, regional, systemic, and contralateral breast cancer recurrence were 18, 16, 64, and 8, respectively. The numbers of subjects with low and high BCL1 expression were 174 (44.3%) and 219 (55.7%), respectively. The numbers of subjects with low and high BCL2 expression were 205 (52.2%) and 188 (47.8%), respectively (Table 1). The high BCL1 group included higher proportions of patients with ER-positive, PR-positive, and low histologic grade compared with the low BCL1 group. The high BCL2 group showed higher proportions of patients with ER-positive, PR-positive, low histologic grade, and age ≤ 50 years compared with the low BCL2 group. The high BCL1 and high BCL2 groups showed higher proportions of the HRc(+)/HER2(−) and HRc(+)/HER2(+) subtypes and lower proportions of the HRc(−)/HER2(+) and HRc(−)/HER2(−) subtypes. The high BCL1 group received more anti-HER2 therapy and endocrine therapy. The high BCL2 group received more endocrine therapy. High BCL1 expression was associated with high BCL2 expression (p < 0.001). Baseline characteristics of study subjects according to BCL1 and BCL2 statuses are summarized in Table 1.
Survival analysis according to BCL1 and BCL2 statuses
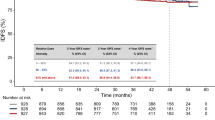
There was no significant association between BCL1 expression and disease-free survival (Fig. 1A). BCL1 expression revealed no survival differences regarding any recurrence types including local recurrence, regional recurrence, systemic recurrence, and contralateral breast cancer (data not shown). A prognostic effect of BCL1 expression was not observed in unselected breast cancers as well as in HRc-positive or HRc-negative breast cancers (data not shown). However, the high BCL2 group showed superior disease-free survival (p = 0.002) compared with the low BCL2 group (Fig. 1B). The high BCL2 group showed superior local recurrence-free survival (p = 0.045) and systemic recurrence-free survival (p = 0.002). However, BCL2 status did not reveal a significant survival difference in terms of regional recurrence-free survival and contralateral breast cancer-free survival (Fig. 2). The high BCL2 group showed better disease-free survival (p = 0.004) in HRc-positive breast cancers but there was no prognostic significance of BCL2 expression in HRc-negative breast cancers (Fig. S7). Detailed survival rates according to BCL2 status for disease-free survival are described in Table S1.
Univariable and multivariable analyses according to BCL2 status
BCL2 expression was a significant prognostic factor by univariable analysis (HR, 0.528; 95% CI, 0.353–0.790; p = 0.002; Table 2). By multivariable analyses, BCL2 expression was also an independent prognostic factor in both model 1 (HR, 0.488; 95% CI, 0.305–0.782; p = 0.003) and model 2 (HR, 0.532; 95% CI, 0.353–0.801; p = 0.003).
Subgroup survival analyses according to BCL2 status
High BCL2 expression was a significant favorable prognostic indicator in all subjects (Fig. 3) regardless of nodal positivity, PR, age group, and chemotherapy. BCL2 expression was also a significant prognostic indicator in subgroups including tumor size > 2 cm, stage II, ER-positive, HER2-negative, histologic grade 1 or 2, positive lymphovascular invasion, and body mass index ≤ 25 kg/m2. Additionally, BCL2 expression was a favorable prognostic factor in patients who received lumpectomy, radiation therapy or endocrine therapy, and in patients who did not receive anti-HER2 therapy. Regarding breast cancer subtype, high BCL2 expression was associated with higher disease-free survival in the HRc(+)/HER2(−) subtype only (p = 0.001; Fig. 4). BCL2 expression had no association with disease-free survival in the other subtypes including HRc(+)/HER2(+), HRc(−)/HER2(+), and HRc(−)/HER2(−). Of note, prudent interpretation of the negative results in the subtypes of HRc(+)/HER2(+), HRc(−)/HER2(+), and HRc(−)/HER2(−) should be exercised, as the subject numbers of respective subtypes are small.
Subgroup analyses by Cox proportional hazards model according to BCL2 status regarding disease-free survival. Abbreviation: CI, confidence interval; HER2, human epidermal growth factor receptor 2; HR, hazard ratio. a)HRs are the relative risks of the high BCL2 group with reference of the low BCL2 group by Cox proportional hazards model. b)In the forest plot, a HR value of less than 1 favors the high BCL2 group against the low BCL2 group. The red circles mean statistical significance and the blue squares mean no statistical significance. The green diamond means the result of total subjects.
Discussion
This study investigated the prognostic roles of BCL1 and BCL2 expression in breast cancer using tissue microarrays from 393 operable primary breast cancer patients. BCL1 expression had no prognostic impact at all but BCL2 expression had significant prognostic effects. The high BCL2 group showed higher disease-free survival compared with the low BCL2 group, especially regarding local recurrence and systemic recurrence. BCL2 expression was a significant independent prognostic factor in terms of disease-free survival.
BCL1 is thought to be a driving oncogene in various solid tumors including breast cancer1,2,3,4,28,29. BCL1, or cyclin D1, is amplified in the G1 phase and coordinates the G1 to S phase entry2. DNA synthesis of cyclin D1 is complexed with cyclin D-dependent kinases (CDKs), especially with CDK4 and CDK6, and regulates phosphorylation of the retinoblastoma protein, resulting in the release of transcription factors such as E2F-130. These transcription factors then allow cell cycle progression from G1 to S phase. Cyclin D1 and the CDKs are presumably activated in the majority of human tumors and enable cancer cells to enter the cell cycle continuously with a dramatically shortened G1 phase2. Several previous studies have proposed the CDK4/6 axis as a rational therapeutic target for cancer treatment2,31,32.
As the biological function of BCL1 has been reported to be closely associated with oncogenic activity in human cancer cells, BCL1 was generally predicted to have an adverse effect on the prognosis of breast cancer. Accordingly, some previous studies have reported an adverse prognostic effect of BCL1 in breast cancer patients8,9,10. A study analyzed data from 364 breast cancer patients and reported that high BCL1 expression was associated with worse overall survival in ER-positive cancers8. Another study performed meta-analysis using data from 9238 breast cancer patients from 21 different studies and reported that BCL1 amplification was associated with a higher risk of recurrence and mortality9. However, other studies have reported no association between BCL1 and breast cancer prognosis11,12. A study that analyzed 1014 breast cancer patients reported that BCL1 amplification was not associated with significant increases in relapse or death11. Another study performed meta-analysis using data from 9189 breast cancer patients from 34 studies and found that high expression of BCL1 was not significantly related to overall survival12. Furthermore, other studies have reported that BCL1 is a favorable prognostic indicator13,14,15. A study analyzed data from 179 breast cancer patients and reported that overexpression of BCL1 was associated with higher overall survival13. Another study analyzed data from 102 breast cancer patients and reported increased breast cancer-specific survival and increased overall survival in patients with BCL1 expression14. As a whole, the prognostic role of BCL1 in breast cancer is largely inconsistent to date.
In this study, although high BCL1 expression was significantly associated with favorable clinicopathologic features such as ER-positive, PR-positive, and low histologic grade, BCL1 expression had no prognostic effect in unselected breast cancers as well as in HRc-positive or HRc-negative breast cancers. While BCL1 has adverse prognostic effects as an oncogene, it has also favorable prognostic effects in breast cancer. BCL1 expression is reported to be associated with well-differentiated breast cancers having favorable clinicopathologic features such as small tumor size, positive ER/PR, and low histologic grade14,28. Additionally, BCL1 has been reported to be associated with endocrine therapy resistance in breast cancer10,33,34. Favorable effects and adverse effects of BCL1 expression on the breast cancer prognosis could cancel each other, As a whole, BCL1 expression might have no prognostic effect on breast cancer in this study. However, the role of BCL1 as a prognostic indicator in breast cancer is still controversial, and further study is required to conclusively elucidate its effect.
Since BCL2 is a putative oncogene in various malignant tumors, BCL2 expression has been hypothesized to have an adverse prognostic effect in breast cancers. However, most previous studies have reported a favorable prognostic effect of BCL216,17,18,19,20. Callagy et al. analyzed data from 930 breast cancers using tissue microarrays and reported that BCL2 expression is an independent favorable prognostic marker regarding overall survival16. They also performed a meta-analysis of 18 studies including 5892 breast cancers and reported the same results17. Dawson et al. analyzed 5 studies of 11,212 breast cancers and reported that BCL2 is a powerful favorable prognostic marker irrespective of molecular subtypes or adjuvant therapies received18. Hwang et al. also reported that BCL2 is a powerful independent favorable prognostic factor in breast cancer, but the impact of BCL2 was different across breast cancer subtypes19,20. Although most previous studies have reported favorable prognostic effects of BCL2, some studies have reported different results. A study analyzed data from 414 breast cancers and reported that BCL2 positivity was not an independent prognosticator in unselected breast cancers21. Another study analyzed data from 64 triple-negative breast cancers and reported that high expression of BCL2 is an independent adverse prognostic factor regarding overall survival22.
The prognostic influence of BCL2 expression might be different across breast cancer subtypes. Hwang et al. reported that BCL2 expression was a strong prognostic factor in the HRc(+)/HER2(−) subtype20, whereas BCL2 expression had only marginal significance in the HRc(+)/HER2(+) subtype, and it was not a significant prognosticator in the HRc(−)/HER2(+) and HRc(−)/HER2(−) subtypes. Dawson et al. reported that BCL2 is a powerful favorable prognostic marker regardless of ER or HER2 status18. Another study analyzed data of 2399 breast cancer patients and reported that BCL2 expression is an independent favorable prognostic factor only in the HRc(+)/HER2(−) subtype but not in the other subtypes35. In this study, BCL2 expression was a significant prognostic factor only in the HRc(+)/HER2(−) subtype.
The mechanisms through which BCL2 might exert its protective effect in breast cancer are unclear16,17. Inhibition of apoptosis is oncogenic, whereas promotion of cell cycle arrest is tumor suppressive. Although BCL2 is known as an anti-apoptotic factor in human malignancies, paradoxical inhibition of solid tumor cell growth by BCL2 has been proposed36. BCL2 family members can be both oncogenic and tumor suppressive, and which of the dual functions predominates is lineage-specific and context-dependent37. The BCL2 pathway has also been reported to be closely associated with resistance mechanisms to chemotherapy, endocrine therapy, anti-HER2 therapy, and radiation therapy in breast cancer38,39,40,41. Clinically, BCL2 expression is associated with favorable clinicopathologic features such as small tumor size, negative node, and low histologic grade. BCL2 expression has a strong correlation with HRc-positive and HER2-negative subtypes. These favorable clinicopathologic features could partly explain the superior prognosis of BCL2-positive breast cancers19,20. Additionally, BCL2 positivity could be closely related to markers that denote better differentiation of breast cancer cells16,20,42,43. High protein expression of BCL2 is observed in luminal subtypes from The Cancer Genome Atlas data42,43, and BCL2 expression is dominant in luminal subtypes, especially in luminal A from clinical data20. BCL2 expression is down-regulated in non-luminal subtypes such as HER2 and triple-negative subtypes which are related to worse prognoses43.
This study investigated the prognostic impacts of BCL1 and BCL2 expression in breast cancer by utilizing tissue microarray data of breast cancer patients. While detailed clinicopathologic information and long-term follow-up duration are the strengths of our study, it is important to denote the limitations. First, the number of subjects is relatively small, which might lessen the statistical power, especially in subgroup analyses. In particular, the negative results in the subtypes of HRc(+)/HER2(+), HRc(−)/HER2(+), and HRc(−)/HER2(−) might be rather inconclusive which need to be validated using larger number of subjects. Second, the cut-off values of the BCL1 and BCL2 expression are arbitrary because there are no standardized methods. Further studies are needed to overcome these limitations and validate the prognostic roles of BCL1 and BCL2.
In conclusion, BCL1 expression had no prognostic impact on disease-free survival in breast cancer, whereas BCL2 expression was a significant independent prognostic factor in terms of disease-free survival. The high BCL2 group displayed superior disease-free survival compared with the low BCL2 group, especially regarding local recurrence and systemic recurrence. The favorable prognostic effect of BCL2 expression was detected only in the HRc(+)/HER2(−) subtype, but not in the other subtypes including HRc(+)/HER2(+), HRc(−)/HER2(+), and HRc(−)/HER2(−). Favorable clinicopathologic features and strong association with ER/PR status could partly explain the superior prognosis of the high BCL2 group. BCL2 expression could be utilized to predict prognosis of operable breast cancer patients in clinical settings. As low BCL2 expression is associated with unfavorable clinical outcomes, more active adjuvant treatment might be recommended for breast cancer patients with low BCL2 expression. Further studies are required to validate the usefulness of BCL2 expression as a prognostic marker in breast cancer.
Abbreviations
- CDK:
-
Cyclin D-dependent kinase
- CI:
-
Confidence interval
- ER:
-
Estrogen receptor
- HER2:
-
Human epidermal growth factor receptor 2
- HR:
-
Hazard ratio
- HRc:
-
Hormone receptor
- PR:
-
Progesterone receptor
References
Kim, J. K. & Diehl, J. A. Nuclear cyclin D1: an oncogenic driver in human cancer. J. Cell Physiol. 220, 292–296. https://doi.org/10.1002/jcp.21791 (2009).
Choi, Y. J. & Anders, L. Signaling through cyclin D-dependent kinases. Oncogene 33, 1890–1903. https://doi.org/10.1038/onc.2013.137 (2014).
Casimiro, M. C., Velasco-Velazquez, M., Aguirre-Alvarado, C. & Pestell, R. G. Overview of cyclins D1 function in cancer and the CDK inhibitor landscape: past and present. Expert Opin. Investig. Drugs 23, 295–304. https://doi.org/10.1517/13543784.2014.867017 (2014).
Qie, S. & Diehl, J. A. Cyclin D1, cancer progression, and opportunities in cancer treatment. J. Mol. Med. (Berl.) 94, 1313–1326. https://doi.org/10.1007/s00109-016-1475-3 (2016).
Adams, J. M. & Cory, S. The BCL-2 arbiters of apoptosis and their growing role as cancer targets. Cell Death Differ. 25, 27–36. https://doi.org/10.1038/cdd.2017.161 (2018).
Czabotar, P. E., Lessene, G., Strasser, A. & Adams, J. M. Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat. Rev. Mol. Cell Biol. 15, 49–63. https://doi.org/10.1038/nrm3722 (2014).
Lin, H. M., Lee, Y. J., Li, G., Pestell, R. G. & Kim, H. R. Bcl-2 induces cyclin D1 promoter activity in human breast epithelial cells independent of cell anchorage. Cell Death Differ. 8, 44–50. https://doi.org/10.1038/sj.cdd.4400770 (2001).
Ahlin, C. et al. High expression of cyclin D1 is associated to high proliferation rate and increased risk of mortality in women with ER-positive but not in ER-negative breast cancers. Breast Cancer Res. Treat. 164, 667–678. https://doi.org/10.1007/s10549-017-4294-5 (2017).
He, Q. et al. Clinicopathological and prognostic significance of cyclin D1 amplification in patients with breast cancer: a meta-analysis. J. BUON 22, 1209–1216 (2017).
Kenny, F. S. et al. Overexpression of cyclin D1 messenger RNA predicts for poor prognosis in estrogen receptor-positive breast cancer. Clin. Cancer Res. 5, 2069–2076 (1999).
Seshadri, R. et al. Cyclin DI amplification is not associated with reduced overall survival in primary breast cancer but may predict early relapse in patients with features of good prognosis. Clin. Cancer Res. 2, 1177–1184 (1996).
Moradi Binabaj, M. et al. The prognostic value of cyclin D1 expression in the survival of cancer patients: a meta-analysis. Gene 728, 144283. https://doi.org/10.1016/j.gene.2019.144283 (2020).
Ortiz, A. B. et al. Prognostic significance of cyclin D1 protein expression and gene amplification in invasive breast carcinoma. PLoS ONE 12, e0188068. https://doi.org/10.1371/journal.pone.0188068 (2017).
Peurala, E., Koivunen, P., Haapasaari, K. M., Bloigu, R. & Jukkola-Vuorinen, A. The prognostic significance and value of cyclin D1, CDK4 and p16 in human breast cancer. Breast Cancer Res. 15, R5. https://doi.org/10.1186/bcr3376 (2013).
Bilalovic, N., Vranic, S., Basic, H., Tatarevic, A. & Selak, I. Immunohistochemical evaluation of cyclin D1 in breast cancer. Croat. Med. J. 46, 382–388 (2005).
Callagy, G. M. et al. Bcl-2 is a prognostic marker in breast cancer independently of the Nottingham prognostic index. Clin. Cancer Res. 12, 2468–2475. https://doi.org/10.1158/1078-0432.CCR-05-2719 (2006).
Callagy, G. M., Webber, M. J., Pharoah, P. D. & Caldas, C. Meta-analysis confirms BCL2 is an independent prognostic marker in breast cancer. BMC Cancer 8, 153. https://doi.org/10.1186/1471-2407-8-153 (2008).
Dawson, S. J. et al. BCL2 in breast cancer: a favourable prognostic marker across molecular subtypes and independent of adjuvant therapy received. Br. J. Cancer 103, 668–675. https://doi.org/10.1038/sj.bjc.6605736 (2010).
Hwang, K. T. et al. Prognostic influence of BCL2 expression in breast cancer. Int. J. Cancer 131, E1109-1119. https://doi.org/10.1002/ijc.27539 (2012).
Hwang, K. T. et al. Prognostic influence of BCL2 on molecular subtypes of breast cancer. J. Breast Cancer 20, 54–64. https://doi.org/10.4048/jbc.2017.20.1.54 (2017).
Jalava, P. J., Collan, Y. U., Kuopio, T., Juntti-Patinen, L. & Kronqvist, P. Bcl-2 immunostaining: a way to finding unresponsive postmenopausal N+ breast cancer patients. Anticancer Res. 20, 1213–1219 (2000).
Ozretic, P. et al. Apoptosis regulator Bcl-2 is an independent prognostic marker for worse overall survival in triple-negative breast cancer patients. Int. J. Biol. Markers 33, 109–115. https://doi.org/10.5301/ijbm.5000291 (2018).
Hwang, K. T. et al. Influence of androgen receptor on the prognosis of breast cancer. J. Clin. Med. https://doi.org/10.3390/jcm9041083 (2020).
Allred, D. C., Harvey, J. M., Berardo, M. & Clark, G. M. Prognostic and predictive factors in breast cancer by immunohistochemical analysis. Mod. Pathol. 11, 155–168 (1998).
Hammond, M. E. et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. Arch. Pathol. Lab. Med. 134, 907–922. https://doi.org/10.1043/1543-2165-134.6.907 (2010).
Allison, K. H. et al. Estrogen and progesterone receptor testing in breast cancer: ASCO/CAP guideline update. J. Clin. Oncol. 38, 1346–1366. https://doi.org/10.1200/JCO.19.02309 (2020).
Wolff, A. C. et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. Arch. Pathol. Lab. Med. 138, 241–256. https://doi.org/10.5858/arpa.2013-0953-SA (2014).
Barnes, D. M. & Gillett, C. E. Cyclin D1 in breast cancer. Breast Cancer Res. Treat. 52, 1–15. https://doi.org/10.1023/a:1006103831990 (1998).
Arnold, A. & Papanikolaou, A. Cyclin D1 in breast cancer pathogenesis. J. Clin. Oncol. 23, 4215–4224. https://doi.org/10.1200/JCO.2005.05.064 (2005).
Narasimha, A. M. et al. Cyclin D activates the Rb tumor suppressor by mono-phosphorylation. Elife https://doi.org/10.7554/eLife.02872 (2014).
VanArsdale, T., Boshoff, C., Arndt, K. T. & Abraham, R. T. Molecular pathways: targeting the cyclin D-CDK4/6 axis for cancer treatment. Clin. Cancer Res. 21, 2905–2910. https://doi.org/10.1158/1078-0432.CCR-14-0816 (2015).
Sherr, C. J., Beach, D. & Shapiro, G. I. Targeting CDK4 and CDK6: from discovery to therapy. Cancer Discov. 6, 353–367. https://doi.org/10.1158/2159-8290.CD-15-0894 (2016).
Stendahl, M. et al. Cyclin D1 overexpression is a negative predictive factor for tamoxifen response in postmenopausal breast cancer patients. Br. J. Cancer 90, 1942–1948. https://doi.org/10.1038/sj.bjc.6601831 (2004).
Jirstrom, K. et al. Adverse effect of adjuvant tamoxifen in premenopausal breast cancer with cyclin D1 gene amplification. Cancer Res. 65, 8009–8016. https://doi.org/10.1158/0008-5472.CAN-05-0746 (2005).
Seong, M. K. et al. Bcl-2 is a highly significant prognostic marker of hormone-receptor-positive, human epidermal growth factor receptor-2-negative breast cancer. Breast Cancer Res. Treat. 150, 141–148. https://doi.org/10.1007/s10549-015-3305-7 (2015).
Pietenpol, J. A. et al. Paradoxical inhibition of solid tumor cell growth by bcl2. Cancer Res. 54, 3714–3717 (1994).
Zinkel, S., Gross, A. & Yang, E. BCL2 family in DNA damage and cell cycle control. Cell Death Differ. 13, 1351–1359. https://doi.org/10.1038/sj.cdd.4401987 (2006).
Hwang, E. et al. ABT-737 ameliorates docetaxel resistance in triple negative breast cancer cell line. Ann. Surg. Treat. Res. 95, 240–248. https://doi.org/10.4174/astr.2018.95.5.240 (2018).
Merino, D., Lok, S. W., Visvader, J. E. & Lindeman, G. J. Targeting BCL-2 to enhance vulnerability to therapy in estrogen receptor-positive breast cancer. Oncogene 35, 1877–1887. https://doi.org/10.1038/onc.2015.287 (2016).
Saatci, O. et al. Targeting PLK1 overcomes T-DM1 resistance via CDK1-dependent phosphorylation and inactivation of Bcl-2/xL in HER2-positive breast cancer. Oncogene 37, 2251–2269. https://doi.org/10.1038/s41388-017-0108-9 (2018).
Lu, L. et al. Activation of STAT3 and Bcl-2 and reduction of reactive oxygen species (ROS) promote radioresistance in breast cancer and overcome of radioresistance with niclosamide. Oncogene 37, 5292–5304. https://doi.org/10.1038/s41388-018-0340-y (2018).
The Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature 490, 61–70. https://doi.org/10.1038/nature11412 (2012).
Hwang, K. T. et al. BCL2 regulation according to molecular subtype of breast cancer by analysis of the cancer genome atlas database. Cancer Res. Treat. 50, 658–669. https://doi.org/10.4143/crt.2017.134 (2018).
Acknowledgements
This research was supported by a multidisciplinary research Grant-in-aid from the Seoul Metropolitan Government Seoul National University Boramae Medical Center (02-2018-4) and by Research Program 2019 funded by Seoul National University College of Medicine Research Foundation (800-20190513).
We appreciate valuable discussion from the members of the Boramae hospital Breast cancer Study group (BBS). Ki-Tae Hwang, MD, PhD (Department of Surgery); Bo Kyung Koo, MD, PhD (Department of Internal Medicine); Byoung Hyuck Kim, MD (Department of Radiation Oncology); Young A Kim, MD, PhD (Department of Pathology); Jongjin Kim, MD (Department of Surgery); Eun Youn Roh, MD, PhD (Department of Laboratory Medicine); Sejung Maeng, PhD (Department of Surgery); Sung Bae Park, MD (Department of Neurosurgery); Jeong Hwan Park, MD, PhD (Department of Pathology); Jin Hyun Park, MD, MS (Department of Internal Medicine); Miyeon Seo, RN (Department of Surgery); Bumjo Oh, MD (Department of Family Medicine); So Won Oh, MD, PhD (Department of Nuclear Medicine); Sohee Oh, PhD (Medical Research Collaborating Center); Jong Yoon Lee, MD, MS (Department of Radiology); Ji Hyun Chang, MD, PhD (Department of Radiation Oncology); Se Hee Jung, MD, PhD (Department of Rehabilitation Medicine); Young Jun Chai, MD, MS (Department of Surgery); In Sil Choi, MD, PhD (Department of Internal Medicine); A Jung Chu, MD (Department of Radiology); Kyu Ri Hwang, MD, PhD (Department of Obstetrics and Gynecology). All BBS members are from Seoul Metropolitan Government Seoul National University Boramae Medical Center (39, Boramae-Gil, Dongjak-gu, Seoul, 156-707, Republic of Korea).
Author information
Authors and Affiliations
Contributions
K.T.H. conceived, designed, and supervised the study. K.T.H., Y.A.K., J.K., H.J.O., and J.H.P. collected and assembled data. K.T.H., Y.A.K., I.S.C., J.H.P., S.O., A.C., J.Y.L. and K.R.H. analyzed data and interpreted the results. K.T.H. and Y.A.K. wrote the manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hwang, KT., Kim, Y.A., Kim, J. et al. Prognostic influences of BCL1 and BCL2 expression on disease-free survival in breast cancer. Sci Rep 11, 11942 (2021). https://doi.org/10.1038/s41598-021-90506-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-90506-x
This article is cited by
-
Detecting the expression of HRs and BCL2 via IHC can help identify luminal A-like subtypes of triple-positive breast cancers
Clinical and Translational Oncology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.