Abstract
Breast cancer (BC), the most common type of malignant tumor, is the leading cause of death, having the highest incidence rate among women. The lack of early diagnostic tools is one of the clinical obstacles for BC treatment. The current study was designed to evaluate a panel of long non-coding RNAs (lncRNAs) BC040587, HOTAIR, MALAT1, CCAT1, CCAT2, PVT1, UCA1, SPRY4-IT1, PANDAR, and AK058003—and two mRNAs (SNCG, BDNF) as novel prognostic biomarkers for BC. This study was ethically approved by the Institutional Review Board of the National Cancer Institute, Cairo University. Our study included 75 women recently diagnosed with BC and 25 healthy women as normal controls. Patients were divided into three groups: 24 with benign breast diseases, 28 with metastatic breast cancer (MBC, stage IV), and 23 with non-metastatic breast cancer (NMBC, stage III). LncRNA and mRNA expression levels were measured in patient plasma using quantitative real-time PCR. We found that 10 lncRNAs (BCO40587, HOTAIR, PVT1, CCAT2, PANDAR, CCAT1, UCA1, SPRY4-IT1, AK058003, and MALAT1) and both mRNAs demonstrated at least a 2-fold change in expression with a more than 95% probability of significance. BCO40587 and SNCG were significantly up-regulated in MBC and NMBC patients (3.2- and 4-fold, respectively) compared with normal controls. The expression of UCA1 was repressed by 1.78-fold in MBC and NMBC patients compared with those with benign diseases. SPRY4-IT1 was down-regulated by 1.45-fold in MBC patients compared with NMBC and benign disease patients. Up-regulation of lncRNAs plays an important role in BC development. SNCG and BCO40587 may be potential prognostic markers for BC.
The organization number is IORG0003381 (IRB No: IRB00004025).
Similar content being viewed by others
Introduction
Breast cancer (BC) is a malignant tumor that has a high mortality rate and mostly occurs in women1. Although treatment strategies for molecular subtypes of BC have significantly progressed, the therapeutic response is often unsatisfactory1. Histopathological examination of tumor biopsies, which is an invasive procedure, remains the gold standard for diagnosing BC2,3. Other screening methods like mammography and breast examination were less effective in women with early breast tumors, aggressive tumors, and dense breast tissues2,3. BC metastasis is the leading cause of death, accounting for more than 90% of cancer-related deaths. Hence, new effective diagnostic and prognostic biomarkers are urgently needed to improve the survival rate and treatment efficiency of patients with BC4.
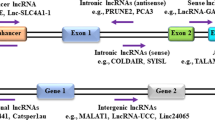
Genome-wide transcriptomics has revealed that protein-coding genes constitute only 2% of the human genome5. About 78% of the genome is composed of non-coding RNA (ncRNA) transcripts5. ncRNAs are divided into infrastructural and regulatory ncRNAs6,7. Infrastructural ncRNAs include ribosomal, transfer, small nuclear, and small nucleolar RNAs6,7. Regulatory ncRNAs include long ncRNAs (lncRNAs), microRNAs, piwi-interacting RNAs, and circular RNAs6,7.
LncRNAs are the longest members of this group, with sequences exceeding 200 nucleotides in length8,9. Most lncRNAs do not have open reading frames and coding potential, but they are transcribed by RNA polymerase II and undergo posttranscriptional modifications like coding RNAs8,9. LncRNAs are widespread in humans and are essential for regulating gene expression in physiological and pathological processes10. Advanced RNA sequencing and microarray technologies have shown that several lncRNAs play critical roles in BC progression by enhancing cancer cell proliferation, migration, and metastatic potential11,12. Thus, these molecules can be considered as promising diagnostic and therapeutic biomarkers for various cancers13,14. For example, overexpression of the lncRNAHox transcript antisense RNA (HOTAIR) was associated with an increased tumor aggressiveness and poor prognosis in both primary and metastatic breast tumors15.
The oncogenic lncRNA metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) promoted triple-negative breast cancer proliferation and metastasis by regulating the expression of cell cycle genes16. Suppressing MALAT1 suppressed cancer cell proliferation by promoting cell cycle arrest in the G2/M phase and apoptosis17. Colon cancer associated transcript 2 (CCAT2) also served as a prognostic biomarker to predict the outcome of BC patients18.
LncRNAs affected the expression levels of some mRNAs such as The γ-synuclein gene (SNCG) and Brain-derived neurotrophic factor (BDNF). SNCG is a small naturally unfolded protein belonging to the synuclein family19. Normally, SNCG is expressed in peripheral neurons, ocular tissue, and adipose20. The expression of SNCG has been closely associated with tumor invasion and metastasis in a number of cancer types in a stage specific manner21. The expression of Ak058003 promotes the breast cancer proliferation, invasion and metastasis by regulating SNCG expression22.
BDNF is a member of the neurotrophin super-family which has been indicated in the pathophysiology of the nervous system as it is widely considered as a neuronal transcriptional factor regulating various aspects of neurogenesis and neuron growth in the human nervous system23. Over-expression of BDNF and TrkB has been demonstrated to act as a predictor of a poor clinical outcome and a worse survival when patients are suffering from human bladder cancer, neuroblastoma and breast carcinoma 24,25,26. Thus, assessing the expression of multiple lncRNAs as well as related genes, such as SNCG and BDNF, is imperative to better understand their roles in BC progression.
In this study, we investigated the expression profiles of a panel of plasma lncRNAs—BCO40584, AK58003, HOTAIR, CCAT1, CCAT2, MALAT1, PVT1, promoter of CDKN1A antisense DNA damage activated RNA (PANDAR), urothelial cancer associated 1 (UCA1), and sprouty RTK signaling antagonist 4-intronic transcript 1 (SPRY4-IT1) and the genes SNCG and BDNF in metastatic and non-metastatic BC cases compared with their expressions in benign breast diseases and healthy controls to identify the lncRNAs specifically associated with BC. We then explored whether the differentially expressed lncRNAs affected the expression of SNCG and BDNF. Furthermore, we examined the association between abnormal lncRNA expression and clinical pathology parameters of BC patients. We also calculate the correlation between LncRNAs & mRNAs expression levels and survival analysis of MBC & NMBC patients.
Methods and materials
Study subjects
This study included 51 BC patients diagnosed and treated at the National Cancer Institute, Cairo University, Egypt in the period from December 2017 to May 2020. Of them, 28 were metastatic cases (stage IV) and 23 were non-metastatic (stage III). The mean age of patients at the time of diagnosis was 49.8 ± 9.4 years for metastatic BC (MBC) and 52.6 ± 11.3 years for non-metastatic BC (NMBC). We also included 24 age-matched individuals with benign lesions and 25 apparently healthy normal controls. The mean ages of patients with benign lesions and healthy controls were 45.4 ± 8.4 and 47.96 ± 10.4 years, respectively. The clinical records of each patient were checked for characteristics including age, tumor size, pathologic staging, grading, lymph node status, and the expression of immunohistochemically confirmed hormone receptor markers: estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2), as shown in Table 1. MBC patients were significantly associated with large tumor size (P = 0.003) and positive lymph node metastasis (P < 0.001). The study was approved by the National Cancer Institute’s Institutional Review Board and conducted in accordance with the 2013 Declaration of Helsinki. Written informed consent was obtained from each patient.
Sample collection
By direct venous puncture, 5 ml of blood samples were collected from subjects in tubes containing ethylenediaminetetraacetic acid and centrifuged at 1000 g for 5 min. The plasma samples were carefully transferred into RNase-free tubes for extraction of RNA, aliquoted, and stored at − 80 °C.
RNA extraction and reverse transcription
RNA was extracted using the RNeasy Mini Kit for total RNA (Qiagen, GmBH, Cat. No. 74104), according to the manufacturer’s protocol. The concentration and quality of the extracted RNA were assessed using the NanoDrop 2000 spectrophotometer (Thermo Scientific, USA). The integrity of RNA was verified by the Agilent Bioanalyzer 2100 (Agilent Technologies, Inc.). Reverse transcription of 2 µg of the extracted RNA was carried out using the High-Capacity c-DNA Reverse Transcription Kit (Applied Biosystem, ThermoFisher Scientific, USA, Cat. No. k1691), according to the manufacturer’s protocol. Reverse transcription was performed in a 20 µl reaction volume at 25 °C for 10 min, 37 °C for 120 min, and finally, heat inactivation at 85 °C for 5 min.
Quantitative real-time polymerase chain reaction (qRT-PCR)
qRT-PCR was performed in a 20 µl reaction volume using Maxima SYBR Green RT-PCR Kit (Thermo Scientific, USA, Cat. No. k0221) and the ABI 7900HT Fast Real-Time PCR system software. Each reaction included 20 ng of cDNA and 10 pmol of primers for HOTAIR, MALAT1, CCAT1, CCAT2, UCA1, PVT1, BC040587, SPRY4-IT1, PANDAR, AK058003, SNCG, BDNF, β-actin, and 18S rRNA. β-actin and 18S rRNA were used as housekeeping genes for normalization, as their expression levels are relatively stable in the plasma. The primers of lncRNAs, mRNAs and housekeeping genes were mentioned in Table 2. The thermal cycling protocol was as follows: an initial activation step for 10 min at 95 °C followed by 40 cycles of denaturation at 94 °C for 15 s, annealing at individually determined temperatures for 30 s, and DNA synthesis at 72 °C for 30 s. All reactions were performed in duplicates and melting curves were analyzed. Fold changes in mRNA levels were calculated using the 2−ΔΔCt method. The ΔCts were obtained from the Cts normalized to those of the housekeeping genes.
Statistical analysis
All statistical tests were performed using Studio software (version 3.6). The Shapiro–Wilk test was used to investigate the normality of the data. Different studied groups were compared with an individual’s clinicopathological characteristics using Pearson’s chi-squared test. Differential expression of lncRNAs among the investigated groups was compared with patients’ clinicopathological characteristics using the Kruskal–Wallis test with post-hoc Dunn’s test and Mann–Whitney U test. Hierarchical clustering was used to classify lncRNAs according to differential expression. The lncRNA expression correlation matrices were calculated using Spearman correlation. Overall survival (OS) analysis was performed using Kaplan–Meier curves and verified with the log-rank test estimator. The Cox regression hazard proportional model was applied for univariate and multivariate survival analysis. Statistical significance was indicated with a two-tailed P value of ≤ 0.05.
Ethics approval and consent to participate
The study was ethically approved by the Institutional Review Boards (IRB) of the National Cancer Institute, Cairo University. Organization No·IORG0003381 (IRB NO·IRB00004025).
Results
Expression of lncRNAs and mRNAs
We investigated the fold change (FC) in expression of ten lncRNAs(BCO40587, HOTAIR, PVT1, CCAT2, PANDAR, CCAT1, UCA1, SPRY4-IT1, AK058003, and MALAT1) and two mRNAs (SNCG and BDNF) in patients with MBC, NMBC, benign disease, and normal controls (NC, Fig. 1, Supplementary 1). The median FC in BCO40587 expression was elevated in MBC (9.32), NMBC (9.51) and benign diseases (11.9) compared with NC (1.23) (P = 0.05, P = 0.03, and P = 0.0016, respectively), which indicates that BC040587 is a good marker for all stages of BC. The median FC in UCA1 levels was higher for benign diseases (4.8) than for MBC (0.73), NMBC (1.3), and NC (1.24) (P = 0.0016, P = 0.0153, and P = 0.0078, respectively), which indicates that UCA1 is a good marker for BC. The median FC in SPRY4-IT1 expression was lower in MBC (1.09) compared with benign diseases (1.59) and NC (1.44) (P = 0.0031 and P = 0.013, respectively) with no difference compared with NMBC (median = 1.32, P = 0.406 and P = 0.81, respectively). These results indicate that SPRY4-IT1 is a good marker for MBC. The median FC in AK058003 expression was higher in benign breast diseases (8.71) than in MBC (2.4) and NC (2.0) (P = 0.034 and P = 0.042) with no difference when compared with NMBC (median = 2.9, P = 0.106). These results indicate that AK058003 is a good marker for MBC and benign breast diseases. The expressions of the other lncRNAs did not significantly differ between the different groups. The median FC in SNCG levels was higher in MBC (6.43) and NMBC (21.86) than in NC (0.26) (P = 0.0048 and P = 0.00049, respectively). However, no difference was found between SNCG expression in BC cases and patients with benign diseases, which indicate that SNCG is a good marker for MBC and NMBC.
Associations between mRNAs and/or lncRNAs expression and clinicopathological features of MBC and NMBC patients
We investigated the associations between the expressions of the studied lncRNAs and mRNAs and the clinicopathological characteristics of MBC and NMBC patients (Supplementary 2, Table 1). Reduced SNCG expression was significantly associated with tumor size (P = 0.024), HER2 overexpression (P = 0.016), and BC-related mortality in MBC patients (P = 0.027). However, SNCG expression was not related significantly with the clinicopathological features of NMBC patients. BDNF expression was significantly associated with negative ER expression in NMBC patients (P = 0.03). Left breast tumors in MBC patients were significantly associated with UCA1 and SPRY4-IT1 down-regulation (P = 0.025 and P = 0.027, respectively). The median FC in SPRY4-IT1 expression was significantly reduced with HER2 overexpression in MBC patients (P = 0.025). The expression of AK058003 was significantly higher in MBC patients with tumor size of < 2.5 cm than in those with tumors ≥ 2.5 cm (P = 0.017). In NMBC patients, an up-regulation of AK058003 was significantly associated with young age (P = 0.005), premenopausal status (P = 0.024), and invasive ductal carcinoma compared with invasive lobular carcinoma (P = 0.048). Similarly, CCAT1 expression was significantly higher in MBC patients with ductal than lobular carcinomas (P = 0.02) (Fig. 2). These results indicate that SNCG, SPRY4IT1, AK058003 & CCAT1 expressions were associated with clinicopathological features of MBC patients. BDNF & AK058003 expressions were associated with clinicopathological features of NMBC patients.
Clinicopathological features were significantly associated with lncRNAs/ mRNAs (a–c) SNCG with tumor size, HER2 overexpression and live status, (d) CCAT1 with tumor type, (e) UCA1with laterality, (f, g) SPRY4T1 with laterality and HER2 overexpression, (h–k) AK058003 with age, menopause, tumor size and type, (l) BDNF with ER expression and (m) MALAT1 with tumor type.
Correlations between expression of the studied lncRNAs&mRNAs in different patient cohorts
Using Spearman’s correlation, we evaluated the correlations between the expression levels of the studied lncRNAs (Fig. 3). In the overall BC cohort, BCO40587 expression was positively correlated with PVT1 expression (r = 0.40, P = 0.0036). UCA1 expression was positively correlated with the expressions of BDNF (r = 0.54, P < 0.0001), HOTAIR (r = 0.50, P = 0.00019), MALAT1 (r = 0.31, P = 0.025), and PANDAR (r = 0.29, P = 0.036). The expression of MALAT1 was negatively correlated with that of SPRY4-IT1 (r = 0.4, P = 0.0040) but positively correlated with that of PANDAR (r = 0.41, P = 0.0027). In MBC patients, UCA1 expression was positively correlated with BDNF (r = 0.53, P = 0.0036) and HOTAIR (r = 0.57, P = 0.0016) expression. The expression of PANDAR was positively correlated with HOTAIR (r = 0.44, P = 0.019) and PVT1 (r = 0.38, P = 0.043) expression. By contrast, the expressions of SPRY4-IT1 and MALAT1 were significantly inversely correlated (r = –0.43, P = 0.02). In NMBC patients, a moderate positive correlation was found between the expressions of BCO40587 and PVT1 (r = 0.54, P = 0.0076). UCA1 expression moderately positively correlated with BDNF (r = 0.594, P = 0.003) and MALAT1 (r = 0.42, P = 0.047) expression. A strong positive correlation was found between the expressions of MALAT1 and PANDAR (r = 0.72, P = 0.00012). By contrast, CCAT1 expression was moderately inversely correlated with the expressions of CCAT2 (r = –0.50, P = 0.014) and HOTAIR (r = –0.48, P = 0.019). In patients with benign breast disease, BCO40587 expression was positively correlated with BDNF (r = 0.67, P = 0.0003), HOTAIR (r = 0.6, P = 0.0019), CCAT2 (r = 0.52, P = 0.009), and PVT1 (r = 0.42, P = 0.44) expressions. Moreover, UCA1 expression was positively correlated with BCO40587 (r = 0.58, P = 0.0029), PVT1 (r = 0.43, P = 0.037), and CCAT2 (r = 0.41, P = 0.044) expressions. A positive correlation was found between the expression of AK058003 and BDNF (r = 0.41, P = 0.048) as well as between HOTAIR expression and those of BDNF (r = 0.66, P = 0.0004) and CCAT2 (r = 0.56, P = 0.004). SNCG expression was inversely correlated with BDNF (r = − 0.59, P = 0.0025), BCO40587 (r = − 0.7, P = 0.00015), and PVT1 (r = − 0.42, P = 0.039) expressions.
Survival analysis
The median OS was 25.2 months (range, 0.6–115.2 months). Kaplan–Meier survival analysis showed that the OS rate did not differ between MBC and NMBC patients (P = 0.22, log-rank) (Fig. 1). Based on the median FC in expression, patients were categorized into two groups: those with high expression (≥ median FC) and those with low expression (< median FC). Then, Kaplan–Meier survival analysis examined the survival outcomes of MBC and NMBC patients associated with their lncRNA expressions. In MBC patients, a low expression of SNCG was associated with reduced OS (P = 0.049, log-rank) (Fig. 4). A low expression of CCAT2 was associated with decreased OS (P = 0.023, log-rank) in NMBC patients (Fig. 5). No relation was found between expressions of the other lncRNAs and patients’ outcomes. Univariate Cox regression analysis revealed that large tumor size, high tumor grade, HER2 overexpression, and a low expression of SNCG were associated with an increased risk of poor survival outcomes in MBC patients. HER2 overexpression and down-regulation of CCAT2 were associated with decreased OS in NMBC patients (Table 3). Multivariate survival analysis revealed that high tumor grade and low expression of SNCG were predictors of poorer survival outcomes in MBC patients, while low expression of CCAT2 was the only predictor of poor prognosis in NMBC patients.
Discussion
BC is the second most common type of cancer with high morbidity and mortality rates27.LncRNAs can regulate different aspects of gene expression, including chromatin organization, transcriptional regulation, and posttranscriptional control28. Dysregulation of lncRNA expression has been linked to cancer and other human disorders28. Many lncRNAs play oncogenic roles in BC, such as HOTAIR, MALAT1, UCA1, PANDAR, CCAT1, CCAT2, SPRY4-IT1, and AK05800329. Some lncRNAs act as tumor suppressors in breast cancer, such as BC040587 and growth arrest specific 529.
In the current study, MALAT1 was down-regulated but SNCG and HOTAIR were significantly up-regulated in patients with benign breast diseases. These results agree with previous studies that have suggested that MALAT1 acts as a tumor suppressor gene30, and that HOTAIR was significantly up-regulated in breast tumors31. In contrast, Wu et al. found that 81% of stage III/IV BCs were positive for SNCG expression, but this proportion was only 15% for stage I/II BCs and SNCG was undetectable in benign breast lesions32.
In the present study, SNCG, BC040587, CCAT2, UCA1, AK058003, MALAT1, and PANDAR were up-regulated in NMBC patients. These results corroborate previous studies that have reported the up-regulation of all these lncRNAs in BC tissues33,34,35,36,37,38,39,40,41,42. In contrast, Chi et al.43, found that the expression of BC040587 declined in breast cancer tissues compared with normal tissues. Recent studies have reported a suppressive role for MALAT-1 in BC30,44,45,46.
In the present study, BDNF, HOTAIR, PVT1, CCAT1, and SPRY4-IT1 were down-regulated in NMBC patients. Huth et al. have reported the suppressive properties of BDNF in BC development47. However, our results contrast those of previous studies, wherein the levels of all these lncRNAs have been shown to be elevated in BC tissues48,49,50,51,52,53.
In patients with MBC, UCA1 was found to be down-regulated, which is contrary to Li et al. who reported that the expression of AC026904.1 and UCA1 was higher in MBC than in NMBC35. Also, we found that SNCG expression was lower in MBC patients than in NMBC patients, but Wu et al. have reported that the relative SNCG mRNA levels were higher in the MBC group in their study32. In our study, AK058003 levels were lower in MBC compared with NMBC patients, while a previous study has demonstrated the opposite trend33. Our findings about CCAT2 expression corroborate those of Wu et al., CCAT2 was up-regulated in MBC compared with NMBC patients34.
The present study showed that the expressions of BC040587, HOTAIR, PVT1, and CCAT2 were not correlated with clinicopathological features in MBC and NMBC patients. These results agree with Abd El-Fattah et al., who reported that PVT1 was not significantly associated with other clinicopathological features49. Another study demonstrated that BC040587 was significantly correlated with menopausal status but its expression in breast cancer was not associated with other parameters such as age, tumor location, tumor size, ER status, PR status, P53 status, Ki67 status, lymph node status, and TNM staging43. HOTAIR expression has been shown to be associated with lymph node metastases but not with tumor size or grading28. Tan et al. reported that high CCAT2 expression was significantly associated with cancer growth and metastasis, including tumor size, clinical stage, and TNM classification but other factors like age, gender, and histological differentiation were not significant46. In contrast to our results, studies have reported a significant association between serum HOTAIR expression, tumor size, and tumor stage, over-expression of HOTAIR in HER2-positive samples than in negative ones, and a significant association between PVT1 expression, tumor size, and TNM stage of breast cancer49,55,56.
The current study showed that CCAT1 expression was significantly associated with tumor type in MBC patients, but was not correlated with any clinicopathological features in NMBC patients. However, a previous study has reported CCAT1 expression to be significantly correlated with differentiation grade, TNM stage, and lymph node metastases in BC patients, but not with other factors, such as age, tumor size, ER, PR, and HER2 status57.
We showed that PANDAR expression was significantly associated with family history in MBC patients, but no other correlations were established with clinicopathological features of NMBC patients. Previously, PANDAR expression was shown to be positively associated with lymph node metastasis and advanced clinical stage in patients42.
In the present study, UCA1 expression was significantly associated with laterality in MBC patients, while this lncRNA has been shown to be significantly associated with tumor size and stage of BC, previously35.
SPRY4-IT1 expression was significantly associated with laterality and HER2 in MBC patients. A previous study showed that an up-regulation of SPRY4-IT1 was associated with larger tumor size and later stage of tumor development in BC patients53.
We found MALAT1 expression to be significantly associated with tumor type in MBC patients. Recent studies have showed that high MALAT1 expression was associated with increased tumor stage, recurrence, decreased survival, lymph node size, ER expression, and histological grade41,58.
In our study, AK058003 expression was related to age, menopausal status, and tumor type in NMBC patients, while it was associated with tumor size in MBC patients. A previous study has also shown AK058003 expression to be associated with the extent of lymph node metastasis33.
We found that BDNF expression was associated with ER status in NMBC but it did not correlate with any clinicopathological features in MBC patients. Higher BDNF levels have been shown to be associated with unfavorable pathological parameters, including nodal positivity, and adverse clinical outcomes, including local recurrence, death, poor prognosis, and reduced disease-free survival and OS. The expression of BDNF did not increase with the TNM stage48.
In the present study, SNCG expression was significantly associated with tumor type, HER2, and live status in MBC patients. However, a previous study reported that SNCG expression in the primary tumor was also significantly associated with lymph node involvement and metastasis32. No significant correlation was established between SNCG gene expression, age, menstruation, and the status of ER, PR, proliferating cell nuclear antigen, and HER2.
We demonstrated that AK058003 was positively correlated with BDNF expression, while this lncRNA was positively correlated with SNCG expression in a previous study33.
In our study, low SNCG expression was significantly associated with reduced OS in MBC patients (p = 0.049, log-rank), while Wu et al. reported that SNCG-positive patients showed a significantly poorer prognosis than SNCG-negative patients32. Low CCAT2 expression was significantly associated with decreased OS (p = 0.023, log-rank) in NMBC patients, while Tan et al. found that CCAT2 was significantly correlated with OS and progression-free survival54.
Conclusion
Our study clearly demonstrates that the identification of reliable lncRNA biomarkers for BC demands multiple validation studies in independent patient cohorts with large sample sizes that take into account tumor stages and molecular subtypes as well. In addition, lncRNAs might warrant investigation as components of biomarker panels for BC.
Data availability
The datasets used and analysed during the current study available from the corresponding author on reasonable request.
Abbreviations
- BC:
-
Breast cancer
- NC:
-
Healthy women as normal control
- BB:
-
Benign breast tumor
- MBC:
-
Metastatic breast cancer
- NMBC:
-
Non-metastatic breast cancer
- OS:
-
Overall survival
References
Jin, H. et al. lncRNA and breast cancer: progress from identifying mechanisms to challenges and opportunities of clinical treatment. Mol. Ther. Nucleic Acids 25, 613–637 (2021).
Boyd, N. F. et al. Mammographic density and the risk and detection of breast cancer. N. Engl. J. Med. 356, 227–236 (2007).
Tang, J., Rangayyan, R. M., Xu, J., El Naqa, I. & Yang, Y. Computer-aided detection and diagnosis of breast cancer with mammography: recent advances. IEEE Trans. Inf Technol. Biomed. 13, 236–251 (2009).
Gobbini, E. et al. Time trends of overall survival among metastatic breast cancer patients in the real-life ESME cohort. Eur. J. Cancer 96, 17–24 (2018).
Mattick, J. S. & Rinn, J. L. Discovery and annotation of long noncoding RNAs. Nat. Struct. Mol. Biol. 22, 5–7 (2015).
Wright, M. W. & Bruford, E. A. Naming’junk. Hum. Genomics 5, 90–98 (2011).
Sinkovics, J. G. RNA/DNA and Cancer (Springer, 2016).
Jarroux, J., Morillon, A. & Pinskaya, M. History. In discovery, and classification of lncRNAs in Long Non Coding (ed. Biology, R. N. A.) 1–5 (Springer, 2017).
Dhanoa, J. K., Sethi, R. S., Verma, R., Arora, J. S. & Mukhopadhyay, C. S. Long non-coding RNA: its evolutionary relics and biological implications in mammals: a review. J. Anim. Sci. Technol. 60, 25 (2018).
Mei, J. et al. Systematic characterization of non-coding RNAs in triple-negative breast cancer. Cell Prolif. 53, e12801 (2020).
Spizzo, R., Almeida, M. I. e., Colombatti, A. & Calin, G. A. Long non-coding RNAs and cancer: A new frontier of translational research?. Oncogene 31, 4577–4587 (2012).
Prensner, J. R. & Chinnaiyan, A. M. The emergence of lncRNAs in cancer biology. Cancer Discov. 1, 391–407 (2011).
Silva, A., Bullock, M. & Calin, G. The clinical relevance of long non-coding RNAs in cancer. Cancers 7, 2169–2182 (2015).
Shi, T., Gao, G. & Cao, Y. Long noncoding RNAs as novel biomarkers have a promising future in cancer diagnostics. Dis. Markers 2016, 9085195 (2016).
Gupta, R. A. et al. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature 464, 1071–1076 (2010).
Jin, C., Yan, B., Lu, Q., Lin, Y. & Ma, L. Reciprocal regulation of Hsa-miR-1 and long noncoding RNA MALAT1 promotes triple-negative breast cancer development. Tumour Biol. 37, 7383–7394 (2016).
Zhao, Z., Chen, C., Liu, Y. & Wu, C. 17β-estradiol treatment inhibits breast cell proliferation, migration and invasion by decreasing MALAT-1 RNA level. Biochem. Biophys. Res. Commun. 445, 388–393 (2014).
Redis, R. S. et al. CCAT2, a novel long non-coding RNA in breast cancer: expression study and clinical correlations. Oncotarget 4, 1748–1762 (2013).
Surguchov, A. Intracellular dynamics of synucleins: Here, there and everywhere. Int. Rev. Cell Mol. Biol. 320, 103–169 (2015).
Surguchov, A., Palazzo, R. E. & Surgucheva, I. Gamma synuclein: subcellular localization in neuronal and non-neuronal cells and effect on signal transduction. Cell MotilCytoskelet. 49, 218–228 (2001).
Shao, T. et al. Gamma synuclein is a novel Twist1 target that promotes TGF-β-induced cancer cell migration and invasion. Cell Death Dis. 9, 625 (2018).
Kai, H. E. & Wang, P. Unregulating long non-coding RNA- AK058003 promotes proliferation, invasion and metastasis of breast cancer by regulating the expression of the γ-synuclein gene. Exp. Ther. Med. 9, 1727–1732 (2015).
Pruunsild, P., Kazantseva, A. & Aid, T. Dissecting the human BDNF locus: bidirectional transcription, complex splicing, and multiple promoters. Genomics 90, 397–406 (2007).
Lai, P. C., Chiu, T. H. & Huang, Y. T. Overexpression of BDNF and TrkB in human bladder cancer specimens. Oncol. Rep. 24, 1265–1270 (2010).
Hua, Z. et al. PI3K and MAPK pathways mediate the BDNF/TrkB-increased metastasis in neuroblastoma. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 37(12), 16227–16236 (2016).
Tajbakhsh, A. et al. Therapeutic potentials of BDNF/ TrkB in breast cancer; current status and perspectives. J Cell Biochem 118(9), 2502–2515 (2017).
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Collina, F. et al. LncRNA HOTAIR up-regulation is strongly related with lymph nodes metastasis and LAR subtype of Triple Negative Breast Cancer. J. Cancer 10, 2018–2024 (2019).
Cerk, S. et al. Current status of long non-coding RNAs in human breast cancer. Int. J. Mol. Sci. 17, 1485 (2016).
Kim, J. et al. Long noncoding RNA MALAT1 suppresses breast cancer metastasis. Nat. Genet. 50, 1705–1715 (2018).
Kong, X., Liu, W. & Kong, Y. Roles and expression profiles of long noncoding RNAs in triple-negative breast cancers. J. Cell Mol. Med. 22, 390–394 (2018).
Wu, K. et al. Expression of neuronal protein synuclein gamma gene as a novel marker for breast cancer prognosis. Breast Cancer Res. Treat. 101, 259–267 (2007).
He, K. & Wang, P. Unregulated long non-coding RNA-AK058003 promotes the proliferation, invasion and metastasis of breast cancer by regulating the expression levels of the γ-synuclein gene. Exp. Ther. Med. 9, 1727–1732 (2015).
Wu, Z. J. et al. Long non-coding RNA CCAT2 promotes the breast cancer growth and metastasis by regulating TGF-beta signaling pathway. Eur. Rev. Med. Pharmacol. Sci. 21, 706–714 (2017).
Li, G. Y. et al. Long non-coding RNAs AC026904.1 and UCA1: A “one-two punch” for TGF-beta-induced SNAI2 activation and epithelial-mesenchymal transition in breast cancer. Theranostics 8, 2846–2861 (2018).
Huang, X. J. et al. MALAT1 promotes angiogenesis of breast cancer. Oncol. Rep. 40, 2683–2689 (2018).
Wang, Y. et al. Elevated long noncoding RNA MALAT-1 expression is predictive of poor prognosis in patients with breast cancer: A meta-analysis. Biosci. Rep. 40, BSR20200215 (2020).
Fattahi Dolatabadi, N. et al. The interaction between MALAT1 target, miR-143-3p, and RALGAPA2 is affected by functional SNP rs3827693 in breast cancer. Hum. Cell 33, 1229–1239 (2020).
Stone, J. K. et al. Hypoxia induces cancer cell-specific chromatin interactions and increases MALAT1 expression in breast cancer cells. J. Biol. Chem. 294, 11213–11224 (2019).
Zheng, L. et al. Long non-coding RNA MALAT1 regulates BLCAP mRNA expression through binding to miR-339-5p and promotes poor prognosis in breast cancer. Biosci. Rep. 39, BSR2018284 (2019).
Zidan, H. E., Karam, R. A., El-Seifi, O. S. & Abd Elrahman, T. M. Circulating long non-coding RNA MALAT1 expression as molecular biomarker in Egyptian patients with breast cancer. Cancer Genet. 220, 32–37 (2018).
Li, Y., Su, X. & Pan, H. Inhibition of lncRNA PANDAR reduces cell proliferation, cell invasion and suppresses EMT pathway in breast cancer. Cancer Biomark. Sect. A Dis. Mark. 25, 185–192 (2019).
Chi, Y. et al. Role of BC040587 as a predictor of poor outcome in breast cancer. Cancer Cell Int. 14, 123 (2014).
Yang, Z. et al. Down-regulation of long non-coding RNA MALAT1 induces tumor progression of human breast cancer through regulating CCND1 expression. Open Life Sci. 11, 232–236 (2016).
Kwok, Z. H., Roche, V., Chew, X. H., Fadieieva, A. & Tay, Y. A non-canonical tumor suppressive role for the long non-coding RNA MALAT1 in colon and breast cancers. Int. J. Cancer 143, 668–678 (2018).
Eastlack, S. C., Dong, S., Mo, Y. Y. & Alahari, S. K. Expression of long noncoding RNA MALAT1 correlates with increased levels of Nischarin and inhibits oncogenic cell functions in breast cancer. PLoS ONE 13, e0198945 (2018).
Huth, L. et al. BDNF is associated with SFRP1 expression in luminal and basal-like breast cancer cell lines and primary breast cancer tissues: A novel role in tumor suppression?. PLoS ONE 9, e102558 (2014).
Patani, N., Jiang, W. G. & Mokbel, K. Brain-derived neurotrophic factor expression predicts adverse pathological & clinical outcomes in human breast cancer. Cancer Cell Int. 11, 23 (2011).
El-Fattah, A. A. A., Sadik, N. A. H., Shaker, O. G., Mohamed Kamal, A. & Shahin, N. N. Serum long non-coding RNAs PVT1, HOTAIR, and NEAT1 as potential biomarkers in Egyptian women with breast cancer. Biomolecules 11, 1–19 (2021).
Arshi, A. et al. A comparative study of HOTAIR expression in breast cancer patient tissues and cell lines. Cell J. 22, 178–184 (2020).
Guo, G. et al. Long non-coding RNA PVT1 facilitates cell proliferation by epigenetically regulating FOXF1 in breast cancer. RSC Adv. 8, 2740–2750 (2018).
Tang, T. et al. LncCCAT1 promotes breast cancer stem cell function through activating WNT/β-catenin signaling. Theranostics 9, 7384–7402 (2019).
Shi, Y. et al. The long noncoding RNA SPRY4-IT1 increases the proliferation of human breast cancer cells by upregulating ZNF703 expression. Mol. Cancer 14, 51 (2015).
Tan, J. et al. Long noncoding RNA CCAT2 as a potential novel biomarker to predict the clinical outcome of cancer patients: A meta-analysis. J. Cancer 8, 1498–1506 (2017).
Avazpour, N., Hajjari, M. & Tahmasebi Birgani, M. T. HOTAIR: A promising long non-coding RNA with potential role in breast invasive carcinoma. Front. Genet. 8, 170 (2017).
Zou, B. et al. Prognostic value of long non-coding RNA plasmacytoma variant translocation1 in human solid tumors: A meta-analysis. Medicine (Baltimore) 98, e16087 (2019).
Zhang, Z. et al. Long non-coding RNA CCAT1 as a diagnostic and prognostic molecular marker in various cancers: A meta-analysis. Oncotarget 9, 23695–23703 (2018).
Arun, G. & Spector, D. L. MALAT1 long non-coding RNA and breast cancer. RNA Biol. 16, 860–863 (2019).
Acknowledgements
We would like to thank National Cancer Institute in Egypt for helping us collecting samples.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). This work was supported by National Cancer Institute in Egypt.
Author information
Authors and Affiliations
Contributions
A.Z. and O.A. designed the experiments, while B.E.L., M.E. and O.A. performed all experiments. A.Z., B.E.L., M.E., M.M., O.A. and S.F. carried out interpretation of the results. A.Z. and O.A. conceived of the experiments. All authors participated in coordination and helped to draft the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El-Helkan, B., Emam, M., Mohanad, M. et al. Long non-coding RNAs as novel prognostic biomarkers for breast cancer in Egyptian women. Sci Rep 12, 19498 (2022). https://doi.org/10.1038/s41598-022-23938-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-23938-8
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.