Abstract
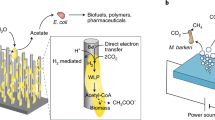
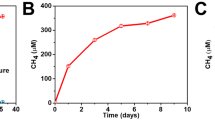
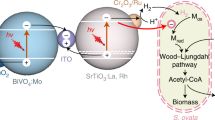
Integrating light-harvesting materials with microbial biochemistry is a viable approach to produce chemicals with high efficiency from air, water and sunlight. Yet it remains unclear whether all photons absorbed in the materials can be transferred through the material–biology interface for solar-to-chemical production and whether the presence of materials beneficially affects microbial metabolism. Here we report a microbe–semiconductor hybrid by interfacing the CO2- and N2-fixing bacterium Xanthobacter autotrophicus with CdTe quantum dots for light-driven CO2 and N2 fixation with internal quantum efficiencies of 47.2% ± 7.3% and 7.1% ± 1.1%, respectively, reaching the biochemical limits of 46.1% and 6.9% imposed by the stoichiometry in biochemical pathways. Photophysical studies suggest fast charge-transfer kinetics at the microbe–semiconductor interfaces, while proteomics and metabolomics indicate a material-induced regulation of microbial metabolism favouring higher quantum efficiencies compared with biological counterparts alone.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The proteomic and metabolomic data generated in this study are available in the Supplementary Data files. The reference proteome of X. autotrophicus was obtained from the UniProt database (UP000305131), and the pathway analyses were performed based on the KEGG database (https://www.genome.jp/kegg/). Other data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.
References
Pan, Y. et al. A large and persistent carbon sink in the world’s forests. Science 333, 988–993 (2011).
Canfield, D. E., Glazer, A. N. & Falkowski, P. G. The evolution and future of Earth’s nitrogen cycle. Science 330, 192–196 (2010).
Blankenship, R. E. et al. Comparing photosynthetic and photovoltaic efficiencies and recognizing the potential for improvement. Science 332, 805–809 (2011).
Cestellos-Blanco, S., Zhang, H., Kim, J. M., Shen, Y. & Yang, P. Photosynthetic semiconductor biohybrids for solar-driven biocatalysis. Nat. Catal. 3, 245–255 (2020).
Chen, H., Dong, F. & Minteer, S. D. The progress and outlook of bioelectrocatalysis for the production of chemicals, fuels and materials. Nat. Catal. 3, 225–244 (2020).
Lu, L. et al. Wastewater treatment for carbon capture and utilization. Nat. Sustain. 1, 750–758 (2018).
Hogewoning, S. W. et al. Photosynthetic quantum yield dynamics: from photosystems to leaves. Plant Cell. 24, 1921–1935 (2012).
Emerson, R. The quantum yield of photosynthesis. Annu. Rev. Plant Physiol. 9, 1–24 (1958).
Allen, M. B. & Arnon, D. I. Studies on nitrogen-fixing blue-green algae. I. Growth and nitrogen fixation by Anabaena cylindrica Lemm. Plant Physiol. 30, 366–372 (1955).
Minchin, F. R. & Witty, J. F., in Advances in Photosynthesis and Respiration (eds Lambers, H. & Ribas-Carbo, M.) 195–205 (Springer, 2006).
Foyer, C. H., Neukermans, J., Queval, G., Noctor, G. & Harbinson, J. Photosynthetic control of electron transport and the regulation of gene expression. J. Exp. Bot. 63, 1637–1661 (2012).
Schwander, T., Schada von Borzyskowski, L., Burgener, S., Cortina Niña, S. & Erb, T. J. A synthetic pathway for the fixation of carbon dioxide in vitro. Science 354, 900–904 (2016).
Salimijazi, F. et al. Constraints on the efficiency of engineered electromicrobial production. Joule 4, 2101–2130 (2020).
Wise, L. et al. Thermodynamic constraints on electromicrobial protein production. Front. Bioeng. Biotechnol. https://doi.org/10.3389/fbioe.2022.820384 (2022).
Claassens, N. J., Sousa, D. Z., dos Santos, V. A. P. M., de Vos, W. M. & van der Oost, J. Harnessing the power of microbial autotrophy. Nat. Rev. Microbiol. 14, 692–706 (2016).
Dogutan, D. K. & Nocera, D. G. Artificial photosynthesis at efficiencies greatly exceeding that of natural photosynthesis. Acc. Chem. Res. 52, 3143–3148 (2019).
Sakimoto, K. K. et al. Physical biology of the materials–microorganism interface. J. Am. Chem. Soc. 140, 1978–1985 (2018).
Zhang, T. More efficient together. Science 350, 738–739 (2015).
Fang, X., Kalathil, S. & Reisner, E. Semi-biological approaches to solar-to-chemical conversion. Chem. Soc. Rev. 49, 4926–4952 (2020).
Sakimoto, K. K., Wong, A. B. & Yang, P. D. Self-photosensitization of nonphotosynthetic bacteria for solar-to-chemical production. Science 351, 74–77 (2016).
Brown, K. A. et al. Light-driven dinitrogen reduction catalyzed by a CdS: nitrogenase MoFe protein biohybrid. Science 352, 448–450 (2016).
Liu, C., Colón, B. C., Ziesack, M., Silver, P. A. & Nocera, D. G. Water splitting—biosynthetic system with CO2 reduction efficiencies exceeding photosynthesis. Science 352, 1210–1213 (2016).
Liu, C., Sakimoto, K. K., Colón, B. C., Silver, P. A. & Nocera, D. G. Ambient nitrogen reduction cycle using a hybrid inorganic–biological system. Proc. Natl Acad. Sci. USA 114, 6450–6455 (2017).
Cao, B. et al. Silver nanoparticles boost charge-extraction efficiency in Shewanella microbial fuel cells. Science 373, 1336–1340 (2021).
Zhang, R. et al. Proteomic and metabolic elucidation of solar-powered biomanufacturing by bio–abiotic hybrid system. Chem 6, 234–249 (2020).
Haro-González, P., Martínez-Maestro, L., Martín, I. R., García-Solé, J. & Jaque, D. High-sensitivity fluorescence-lifetime thermal sensing based on CdTe quantum dots. Small 8, 2652–2658 (2012).
Brown, K. A., Song, Q., Mulder, D. W. & King, P. W. Diameter-dependent electron transfer kinetics in semiconductor–enzyme complexes. ACS Nano 8, 10790–10798 (2014).
Wiegel, J. in The Prokaryotes (eds Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K. H. & Stackebrandt, E.) 290–314 (Springer, 2006).
Zhang, H. et al. Bacteria photosensitized by intracellular gold nanoclusters for solar fuel production. Nat. Nanotechnol. 13, 900–905 (2018).
Wang, Z. et al. Efficiency accreditation and testing protocols for particulate photocatalysts toward solar fuel production. Joule 5, 344–359 (2021).
Hisatomi, T. & Domen, K. Reaction systems for solar hydrogen production via water splitting with particulate semiconductor photocatalysts. Nat. Catal. 2, 387–399 (2019).
Ferguson, S. J. ATP synthase: from sequence to ring size to the P/O ratio. Proc. Natl Acad. Sci. USA 107, 16755 (2010).
Rittmann, B. E. et al. in Environmental Biotechnology: Principles and Applications 128–130 (McGraw-Hill, 2001).
Somers, R. C., Bawendi, M. G. & Nocera, D. G. CdSe nanocrystal based chem-/bio-sensors. Chem. Soc. Rev. 36, 579–591 (2007).
Baskoutas, S. & Terzis, A. F. Size-dependent band gap of colloidal quantum dots. J. Appl. Phys. 99, 013708 (2006).
Wang, B. et al. Biohybrid photoheterotrophic metabolism for significant enhancement of biological nitrogen fixation in pure microbial cultures. Energy Environ. Sci. 12, 2185–2191 (2019).
Jiang, Z. et al. AglnS2/In2S3 heterostructure sensitization of Escherichia coli for sustainable hydrogen production. Nano Energy 46, 234–240 (2018).
Gai, P. P. et al. Solar-powered organic semiconductor-bacteria biohybrids for CO2 reduction into acetic acid. Angew. Chem. Int. Ed. 59, 7224–7229 (2020).
Lakowicz, J. R. in Principles of Fluorescence Spectroscopy 3rd edn (ed. Lakowicz, J. R.) 277–286 (Springer, 2006).
Elsen, S., Colbeau, A., Chabert, K. & Vignais, P. M. The hupTUV operon is involved in negative control of hydrogenase synthesis in Rhodobacter capsulatus. J. Bacteriol. 178, 5174–5181 (1996).
Elsen, S., Swem, L. R., Swem, D. L. & Bauer, C. E. RegB/RegA, a highly conserved redox-responding global two-component regulatory system. Microbiol. Mol. Biol. Rev. 68, 263–279 (2004).
Yates, M. G., Jones, C. W. in Advances in Microbial Physiology (eds Rose, A. H. & Tempest D. W.) 71–135 (Academic Press, 1974).
Grosz, R. & Stephanopoulos, G. Statistical mechanical estimation of the free energy of formation of E. coli biomass for use with macroscopic bioreactor balances. Biotechnol. Bioeng. 25, 2149–2163 (1983).
Stouthamer, A. H. A theoretical study on the amount of ATP required for synthesis of microbial cell material. Antonie Van Leeuwenhoek 39, 545–565 (1973).
Jones, S. W., Karpol, A., Friedman, S., Maru, B. T. & Tracy, B. P. Recent advances in single cell protein use as a feed ingredient in aquaculture. Curr. Opin. Biotechnol. 61, 189–197 (2020).
Leger, D. et al. Photovoltaic-driven microbial protein production can use land and sunlight more efficiently than conventional crops. Proc. Natl Acad. Sci. USA 118, e2015025118 (2021).
Øverland, M., Tauson, A., Shearer, K. & Skrede, A. Evaluation of methane-utilising bacteria products as feed ingredients for monogastric animals. Arch. Anim. Nutr. 64, 171–189 (2010).
Peng, Y. et al. Copper(I) cysteine complexes: efficient Earth-abundant oxidation co-catalysts for visible light-driven photocatalytic H2 production. Chem. Commun. 51, 12556–12559 (2015).
Doyle, A., Weintraub, M. N. & Schimel, J. P. Persulfate digestion and simultaneous colorimetric analysis of carbon and nitrogen in soil extracts. Soil Sci. Soc. Am. J. 68, 669–676 (2004).
Cabrera, M. L. & Beare, M. H. Alkaline persulfate oxidation for determining total nitrogen in microbial biomass extracts. Soil Sci. Soc. Am. J. 57, 1007–1012 (1993).
Sigman, D. M. et al. A bacterial method for the nitrogen isotopic analysis of nitrate in seawater and freshwater. Anal. Chem. 73, 4145–4153 (2001).
Atallah, T. L. et al. Decay-associated Fourier spectroscopy: visible to shortwave infrared time-resolved photoluminescence spectra. J. Phys. Chem. A 123, 6792–6798 (2019).
Lakowicz, J. R. in Principles of Fluorescence Spectroscopy 3rd edn (ed. Lakowicz, J. R.) 142 (Springer, 2006).
Brown, K. A., Dayal, S., Ai, X., Rumbles, G. & King, P. W. Controlled assembly of hydrogenase–CdTe nanocrystal hybrids for solar hydrogen production. J. Am. Chem. Soc. 132, 9672–9680 (2010).
Wiegel, J., Wilke, D., Baumgarten, J., Opitz, R. & Schlegel, H. G. Transfer of the nitrogen-fixing hydrogen bacterium Corynebacterium autotrophicum Baumgarten et al. to Xanthobacter gen. nov. Int. J. Syst. Evol. Microbiol. 28, 573–581 (1978).
Björkman, O. & Demming, B. Photon yield of O2 evolution and chlorophyll fluorescence characteristics at 77 K among vascular plants of diverse origins. Planta 170, 489–504 (1987).
Redfield, A. C., Ketchum, B. H., Richards, F. A. in The Sea (ed. Hill, M. N.) 26–77 (Wiley, 1963).
Lu, S., Guan, X. & Liu, C. Electricity-powered artificial root nodule. Nat. Commun. 11, 1505 (2020).
Srivastava, A. K. & Ambasht, R. S. Biomass, production, decomposition of and N release from root nodules in two Casuarina equisetifolia plantations in Sonbhadra, India. J. Appl. Ecol. 32, 121–127 (1995).
Wang, B., Jiang, Z., Yu, J. C. M., Wang, J. & Wong, P. K. Enhanced CO2 reduction and valuable C2+ chemical production by a CdS-photosynthetic hybrid system. Nanoscale 19, 9296–9301 (2019).
Bertram, J. R., Ding, Y. & Nagpal, P. Gold nanoclusters cause selective light-driven biochemical catalysis in living nano-biohybrid organisms. Nanoscale Adv. 2, 2363–2370 (2020).
Ding, Y. et al. Nanorg microbial factories: light-driven renewable biochemical synthesis using quantum dot–bacteria nanobiohybrids. J. Am. Chem. Soc. 141, 10272–10282 (2019).
Göbbels, L. et al. Cysteine: an overlooked energy and carbon source. Sci. Rep. 11, 2139 (2021).
Wang, Y., Zheng, J. W., Zhang, Z. J., Yuan, C. W. & Fu, D. G. CdTe nanocrystals as luminescent probes for detecting ATP, folic acid and l-cysteine in aqueous solution. Colloids Surf. A 342, 102–106 (2009).
Acknowledgements
We thank S. Tenney for the quantum yield characterization experiment, D. Xiang and B. Natinsky for the gas chromatography and A. Xiao for coding assistance. We thank E. Sletten at UCLA for the use of the fluorometer and the dynamic light scattering instrument. We also thank the UCLA Molecular Instrumentation Center, the UCLA Metabolomics Center, the UCLA Proteome Research Center and the UCLA California NanoSystems Institute for instrumental support. This study was supported by National Institutes of Health grant R35GM138241 (C.L.), the Jeffery and Helo Zink Endowed Professional Development Term Chair (C.L.), National Institutes of Health grant S10OD016387 (J.O.P.), a Hellman Fellowship (J.O.P.) and a UCLA Summer Mentored Research Fellowship (X.G.).
Author information
Authors and Affiliations
Contributions
C.L. supervised the project. C.L. and X.G. designed the experiments and wrote the manuscript. S.E conducted the metabolomic experiments and analysed the data under the supervision of J.O.P. X.H. performed the electron microscopy characterizations under the supervision of Y.Y. T.L.A. led the lifetime measurements under the supervision of J.R.C. Y.X. assisted the proteomics analysis. S.L. conducted the flow-cytometry characterization. B.C. assisted with sample preparation for electron microscopy characterization under the supervision of X.D. and Y.H. J.S. and K.W. assisted in the microbial inoculation and photocatalytic experiments. All the authors discussed the results and assisted during the manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Buz Barstow, Paul King and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Characterizations of CdTe quantum dots (QDs).
a, HAADF-STEM image of aggregated QDs and the corresponding EDS mapping in the same region for (b) cadmium (Cd) and (c) tellurium (Te). d, HRTEM image of individual CdTe QDs. e, Magnified HRTEM image of the highlighted region in d. f, the corresponding Fourier transform results indicate a lattice spacing of 0.21 nm, agree with the (220) plane of the cubic phase. g, Ultraviolet-visible spectrum (black) overlayed with the emission spectrum (yellow) of CdTe QDs. h, The measurement of dynamic light scattering along with the corresponding size distribution. The experiment of electron microscopy has been repeated independently for more than 3 times with similar results. Scale bars are 30 nm in a-c, 10 nm in d, and 1 nm in e.
Extended Data Fig. 2 Viability results of X. autotrophicus measured by flow cytometry.
a, The gate of X. autotrophicus in the forward-scattered light area/side-scattered light area (FSC-A/SSC-A) plot. b, Fluorescent intensity of SYTO 9 stained (live) X. autotrophicus versus its FSC-A. c, Viability percentage of the microbes under different conditions, n = 3 biological replicates with 10,000 particles recorded before gating (error bars present the standard deviation).
Extended Data Fig. 3 Gas composition analysis of reactors’ headspace by gas chromatography.
Gas chromatograms of photoreactors’ headspace for the microbe–semiconductor hybrids before (light blue) and after (dark blue) a 4-day photocatalytic reaction, along with a standard sample (black) containing O2 and H2 (0.5 v/v% for each component in N2).
Extended Data Fig. 4 Electron microscopy images of the microbe–semiconductor hybrids and controls.
a, HRTEM image of the microbe–semiconductor hybrid. b, The magnified image of the red box in a. c, The magnified image of the red box in b. d, The corresponding Fourier transform results of c indicating a lattice spacing of 0.22 nm for the QDs. e, TEM image of the microbe–semiconductor hybrid. f, The magnified image of the red box in e. g, HAADF-STEM image of sectioned microbe–semiconductor hybrid in the absence of cysteine. h, HAADF-STEM image of sectioned pure microbes. The experiment of electron microscopy has been repeated independently for more than 3 times with similar results. Scale bars are 1 μm in a, 20 nm in b, 500 nm in e, 50 nm in f, and 200 nm in g and h.
Extended Data Fig. 5 Absorption and emission spectra of partially aggregated CdTe QDs.
Absorbance (solid lines) and photoluminescence (PL) (dashed lines) spectra of the well-dispersed QD suspension (black) and the partially aggregated CdTe QDs (yellow). The average particle diameters from the dynamic light scattering measurements are 4.02 ± 0.61 and 62.06 ± 0.18 nm for the well-dispersed and partially aggregated QDs (mean + /− standard deviation), respectively.
Extended Data Fig. 6 Interactions between QDs and microbes studied by flow cytometry.
a and b, Dispersion of pure CdTe QDs. c and d, Microbial culture of X. autotrophicus. The microbe–semiconductor hybrids at (e and f) t = 0 hr and (i and j) t = 24 hrs after assembly. The mixture of QDs and microbes at (g and h) t = 0 hr and (k and l) t = 24 hrs after assembly. a, c, e, g, i, and k are the plots of SSC-A versus FSC-A that illustrate the identity of the microbial population. b, d, f, h, j, and l are the plots of emission at 525 ± 50 nm versus FSC-A that illustrate the distribution of microbial population gated in the SSC-A/FSC-A plot attached with emissive QDs. The circles in the plots illustrate the gated areas corresponding to the microbial populations attached with emissive QDs whose temporal trends were shown in (m). Representative results presented above (n = 3 biological replicates with 10,000 particles recorded before gating, error bars present the standard deviation). Additional quantification of the percentage of QDs closely interacting with microbes are available in Supplementary Fig. 4.
Extended Data Fig. 7 Stern–Volmer study for the mixture of CdTe QDs and cysteine.
The ratios of PL emission intensities of CdTe QDs (I0/I) without and with the addition of different equivalents of cysteine (Cys). A linear relationship was observed at low Cys equivalents, demonstrating a dynamic quenching mechanism orientated from the diffusive encounters between the Cys molecules and the emissive QDs. The saturated values of I0/I under high Cys equivalents due to quencher accessibility are consistent with the literature report64.
Extended Data Fig. 8 Schematic illustrations of the discussed pathways and the relevant metabolic regulations.
The regulation of the (a) Calvin–Benson-Bassham cycle, (b) two-component regulatory system, and (c) oxidative phosphorylation in the Hybrid compared to the H2-fed microbes.
Extended Data Fig. 9 The optical absorbance of the CdTe QDs in the hybrids as a function of microbial inoculation.
The optical absorbances of the CdTe QDs in the microbe–semiconductor hybrids (ODQDs), sensitive to the presence of microbial scattering centers, are plotted against the amounts of microbial inoculation presented as OD600. See section “Analytical procedures for the aliquoted samples in the photosynthetic experiments” in the Methods for details.
Supplementary information
Supplementary Information
Supplementary Notes 1–3, Methods, Table 1, Figs. 1–4 and references.
Supplementary Data 1
LC-MS results of the metabolome in the X. autotrophicus–CdTe hybrid system before and after the 15N and 13C isotope-labelling experiment.
Supplementary Data 2
Heatmap data for significantly regulated proteins and metabolites in the microbe–semiconductor hybrids in comparison to the H2-fed microbes.
Supplementary Data 3
LC-MS results of the proteome in X. autotrophicus under different experimental conditions.
Supplementary Data 4
Fold changes of protein expression comparing the microbe–semiconductor hybrids against the H2-fed microbes.
Supplementary Data 5
LC-MS results of the metabolome in X. autotrophicus under different experimental conditions.
Supplementary Data 6
Fold changes of metabolites comparing the microbe–semiconductor hybrids against the H2-fed microbes.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guan, X., Erşan, S., Hu, X. et al. Maximizing light-driven CO2 and N2 fixation efficiency in quantum dot–bacteria hybrids. Nat Catal 5, 1019–1029 (2022). https://doi.org/10.1038/s41929-022-00867-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-022-00867-3
This article is cited by
-
Solar-driven waste-to-chemical conversion by wastewater-derived semiconductor biohybrids
Nature Sustainability (2023)
-
Pushing hybrids to the limits
Nature Catalysis (2022)