Abstract
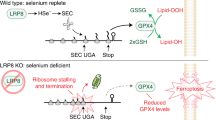
The canonical biological function of selenium is in the production of selenocysteine residues of selenoproteins, and this forms the basis for its role as an essential antioxidant and cytoprotective micronutrient. Here we demonstrate that, via its metabolic intermediate hydrogen selenide, selenium reduces ubiquinone in the mitochondria through catalysis by sulfide quinone oxidoreductase. Through this mechanism, selenium rapidly protects against lipid peroxidation and ferroptosis in a timescale that precedes selenoprotein production, doing so even when selenoprotein production has been eliminated. Our findings identify a regulatory mechanism against ferroptosis that implicates sulfide quinone oxidoreductase and expands our understanding of selenium in biology.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Uncropped immunoblots are provided in Supplementary Information. Additional raw data and materials related to the study are available from the corresponding authors on request. Source Data are provided with this paper.
References
Labunskyy, V. M., Hatfield, D. L. & Gladyshev, V. N. Selenoproteins: molecular pathways and physiological roles. Physiol. Rev. 94, 739–777 (2014).
Burk, R. F. & Hill, K. E. Regulation of selenium metabolism and transport. Annu. Rev. Nutr. 35, 109–134 (2015).
Yang, W. S. et al. Regulation of ferroptotic cancer cell death by GPX4. Cell 156, 317–331 (2014).
Dixon, S. J. et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149, 1060–1072 (2012).
Ingold, I. et al. Selenium utilization by GPX4 is required to prevent hydroperoxide-induced ferroptosis. Cell 172, 409–422 (2018).
Alim, I. et al. Selenium drives a transcriptional adaptive program to block ferroptosis and treat stroke. Cell 177, 1262–1279 (2019).
Karaye, K. M. et al. Selenium supplementation in patients with peripartum cardiomyopathy: a proof-of-concept trial. BMC Cardiovasc. Disord. 20, 457 (2020).
Benstoem, C. et al. Selenium and its supplementation in cardiovascular disease–what do we know? Nutrients 7, 3094–3118 (2015).
Jedrychowski, M. P. et al. Facultative protein selenation regulates redox sensitivity, adipose tissue thermogenesis, and obesity. Proc. Natl Acad. Sci. USA 117, 10789–10796 (2020).
Carlisle, A. E. et al. Selenium detoxification is required for cancer-cell survival. Nat. Metab. 2, 603–611 (2020).
Olm, E. et al. Extracellular thiol-assisted selenium uptake dependent on the x(c)− cystine transporter explains the cancer-specific cytotoxicity of selenite. Proc. Natl Acad. Sci. USA 106, 11400–11405 (2009).
Singha, S. et al. Toward a selective, sensitive, fast-responsive, and biocompatible two-photon probe for hydrogen sulfide in live cells. Anal. Chem. 87, 1188–1195 (2015).
Jiang, X., Stockwell, B. R. & Conrad, M. Ferroptosis: mechanisms, biology and role in disease. Nat. Rev. Mol. Cell Biol. 22, 266–282 (2021).
Stockwell, B. R., Jiang, X. & Gu, W. Emerging mechanisms and disease relevance of ferroptosis. Trends Cell Biol. 30, 478–490 (2020).
Mandal, P. K. et al. System x(c)− and thioredoxin reductase 1 cooperatively rescue glutathione deficiency. J. Biol. Chem. 285, 22244–22253 (2010).
Banjac, A. et al. The cystine/cysteine cycle: a redox cycle regulating susceptibility versus resistance to cell death. Oncogene 27, 1618–1628 (2008).
Mao, C. et al. DHODH-mediated ferroptosis defence is a targetable vulnerability in cancer. Nature 593, 586–590 (2021).
Bersuker, K. et al. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature 575, 688–692 (2019).
Doll, S. et al. FSP1 is a glutathione-independent ferroptosis suppressor. Nature 575, 693–698 (2019).
National Research Council. Mineral Tolerance of Animals: Second Revised Edition. Washington, DC: The National Academies Press https://doi.org/10.17226/11309 (2005)
Combs, G. F. Jr. et al. Bioavailability of selenium accumulated by selenite-reducing bacteria. Biol. Trace Elem. Res. 52, 209–225 (1996).
Mishima, E. et al. A non-canonical vitamin K cycle is a potent ferroptosis suppressor. Nature 608, 778–783 (2022).
Muthusamy, T. et al. Serine restriction alters sphingolipid diversity to constrain tumour growth. Nature 586, 790–795 (2020).
Tawfik, D. S. Enzyme promiscuity and evolution in light of cellular metabolism. FEBS J. 287, 1260–1261 (2020).
Landry, A. P., Ballou, D. P. & Banerjee, R. Hydrogen sulfide oxidation by sulfide quinone oxidoreductase. ChemBioChem 22, 949–960 (2021).
Xia, L. et al. The mammalian cytosolic selenoenzyme thioredoxin reductase reduces ubiquinone. A novel mechanism for defense against oxidative stress. J. Biol. Chem. 278, 2141–2146 (2003).
Nordman, T. et al. Regeneration of the antioxidant ubiquinol by lipoamide dehydrogenase, thioredoxin reductase and glutathione reductase. Biofactors 18, 45–50 (2003).
Magtanong, L. et al. Context-dependent regulation of ferroptosis sensitivity. Cell Chem. Biol. 29, 1568 (2022).
Gao, M. et al. Role of mitochondria in ferroptosis. Mol. Cell 73, 354–363 (2019).
Tan, S., Sagara, Y., Liu, Y., Maher, P. & Schubert, D. The regulation of reactive oxygen species production during programmed cell death. J. Cell Biol. 141, 1423–1432 (1998).
Hakkaart, G. A., Dassa, E. P., Jacobs, H. T. & Rustin, P. Allotopic expression of a mitochondrial alternative oxidase confers cyanide resistance to human cell respiration. EMBO Rep. 7, 341–345 (2006).
Martínez-Reyes, I. et al. Mitochondrial ubiquinol oxidation is necessary for tumour growth. Nature 585, 288–292 (2020).
Spinelli, J. B. et al. Fumarate is a terminal electron acceptor in the mammalian electron transport chain. Science 374, 1227–1237 (2021).
Neitemeier, S. et al. BID links ferroptosis to mitochondrial cell death pathways. Redox Biol. 12, 558–570 (2017).
Tobaben, S. et al. Bid-mediated mitochondrial damage is a key mechanism in glutamate-induced oxidative stress and AIF-dependent cell death in immortalized HT-22 hippocampal neurons. Cell Death Differ. 18, 282–292 (2011).
Ferrante, R. J. et al. Therapeutic effects of coenzyme Q10 and remacemide in transgenic mouse models of Huntington’s disease. J. Neurosci. 22, 1592–1599 (2002).
Beal, M. F. Coenzyme Q10 as a possible treatment for neurodegenerative diseases. Free Radic. Res. 36, 455–460 (2002).
Mantle, D. & Dybring, A. Bioavailability of coenzyme Q(10): an overview of the absorption process and subsequent metabolism. Antioxidants (Basel) 9, 386 (2020).
Pravst, I. et al. Comparative bioavailability of different coenzyme Q10 formulations in healthy elderly individuals. Nutrients 12, 787 (2020).
Sanjana, N. E., Shalem, O. & Zhang, F. Improved vectors and genome-wide libraries for CRISPR screening. Nat. Methods 11, 783–784 (2014).
Lee, N., Carlisle, A. E. & Kim, D. Examining xCT-mediated selenium uptake and selenoprotein production capacity in cells. Methods Enzymol. 662, 1–24 (2022).
Cupp-Sutton, K. A. & Ashby, M. T. Biological chemistry of hydrogen selenide. Antioxidants (Basel) 5, 42 (2016).
Lionella Palego, L. B., Betti, L. & Giannaccini, G. Sulfur metabolism and sulfur-containing amino acids: I- molecular effectors. Biochem. Pharmacol. 4, 3:158 (2015).
Koelmel, J. P. et al. Expanding lipidome coverage using LC-MS/MS data-dependent acquisition with automated exclusion list generation. J. Am. Soc. Mass. Spectrom. 28, 908–917 (2017).
Pang, Z. et al. Using MetaboAnalyst 5.0 for LC-HRMS spectra processing, multi-omics integration and covariate adjustment of global metabolomics data. Nat. Protoc. 17, 1735–1761 (2022).
Sampaio, J. L. et al. Membrane lipidome of an epithelial cell line. Proc. Natl Acad. Sci. USA 108, 1903–1907 (2011).
Ejsing, C. S. et al. Global analysis of the yeast lipidome by quantitative shotgun mass spectrometry. Proc. Natl Acad. Sci. USA 106, 2136–2141 (2009).
Surma, M. A. et al. An automated shotgun lipidomics platform for high throughput, comprehensive, and quantitative analysis of blood plasma intact lipids. Eur. J. Lipid Sci. Technol. 117, 1540–1549 (2015).
Herzog, R. et al. A novel informatics concept for high-throughput shotgun lipidomics based on the molecular fragmentation query language. Genome Biol. 12, R8 (2011).
Herzog, R. et al. LipidXplorer: a software for consensual cross-platform lipidomics. PLoS ONE 7, e29851 (2012).
Spears, M. E. et al. De novo sphingolipid biosynthesis necessitates detoxification in cancer cells. Cell Rep. 40, 111415 (2022).
Possemato, R. et al. Functional genomics reveal that the serine synthesis pathway is essential in breast cancer. Nature 476, 346–350 (2011).
Seashore-Ludlow, B. et al. Harnessing connectivity in a large-scale small-molecule sensitivity dataset. Cancer Discov. 5, 1210–1223 (2015).
Acknowledgements
We thank A. Mercurio, C. Haynes, N. Uma Naresh, S. Kim and O. Ponomarova for advice, assistance and feedback. This work was supported by the Suh Kyungbae Foundation Young Investigator Award and a grant from the National Institutes of Health (no. R01GM148832 to D.I.K.). N.L. and S.J.P. are recipients of a Mogam Science Scholarship. J.B.S. is funded by the Worcester Foundation Grant. This research was also supported by a grant from the National Institutes of Health (no. R01GM112948 to J.A.O.). J.A.O. is a Chan Zuckerberg Biohub investigator. P.L.G. is supported by fellowships from the Searle Scholars Program and the Rita Allen Foundation and by grant no. DP2 OD027719-01 from the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
N.L. and D.I.K. conceived the project and designed the research. N.L. and S.J.P. performed most of the experiments with assistance from M.L., T.T., M.B.D., Y.S., P.L.G. and J.B.S. Data analyses assistance and advice was provided by T.Y.K., D.I.K., Y.S., M.L., J.A.O. and J.B.S. Cell viability-related experiments and ROS and other biochemical measurement experiments were carried out by N.L. and S.J.P. In vitro reactivity experiments were performed by N.L. Immunocytochemistry was performed by M.B.D. The lipid-profiling experiment was performed by M.L., N.L. and J.A.O. Polar metabolite profiling was performed by J.B.S. and N.L. Ubiquinone and ubiquinol analysis was performed by J.B.S., T.T., N.L., S.J.P. and Y.S. Data mining, pathway analyses and interpretation of metabolomics data were carried by M.L., J.A.O., T.T., J.B.S., N.L., S.J.P., T.Y.K. and D.I.K. N.L., S.J.P. and D.K. wrote the manuscript, with consultation provided by all authors.
Corresponding authors
Ethics declarations
Competing interests
J.A.O. is a member of the scientific advisory board for Vicinitas Therapeutics and has ferroptosis-related patent applications. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Metabolism thanks Rajiv Ratan, Fabien Pierrel and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alfredo Giménez-Cassina, in collaboration with the Nature Metabolism team
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Selenium has an antiferroptotic effect that is independent of selenoprotein production, supplemental data 1.
(a) Detailed schematic diagram of canonical selenocysteine synthesis metabolic pathway and selenoprotein production. Created with BioRender.com. (b) Quantification of mRNA levels of selenoproteins in SK-Hep1 and U251 cells after 3 µM selenite treatment for 24 hr. (c) Immunoblots of GPX4 and organelle marker proteins in fractionated SK-Hep1 and U251 cells after vehicle or 3 µM selenite treatment for 24 hr. (d) Quantification of mRNA levels of selenoproteins in SK-Hep1 and U251 cells after 6 µM selenite treatment for 2 hr. (e) Immunoblots of GPX4 and organelle marker proteins in fractionated SK-Hep1 and U251 cells after vehicle or 6 µM selenite treatment for 2 hr. N, M, C, STE, and LTE represent nucleus, mitochondria, cytoplasm, short-term exposure, and long-term exposure, respectively. (f, g) Immunoblots of GPX4 or other selenoproteins in 6 µM selenite treated Hela (f, left panel), NCIH838 (f, right panel), and SK-Hep1 (g) cells. (h) Representative flow cytometry data showing gating strategy for measurement of lipid peroxidation, iron, mitochondrial mass, and ROS. FSC-Area and SSC-Area gating strategy was used to eliminate cell debris. FSC-Height and FSC-Area subgating strategy was used to identify single cell population. The histogram of the single cell population was used for quantifying lipid peroxidation, iron, mitochondrial mass, or ROS. Data are mean ± S.D. from biological replicates (n = 3 for a,b) and were analyzed by two-tailed Student’s t-test. (n.s., not significant).
Extended Data Fig. 2 Selenium has an antiferroptotic effect that is independent of selenoprotein production, supplemental data 2.
(a) Measurement of lipid peroxidation in SK-Hep1 cells treated with 1µ M RSL3, 10 µM FIN56, 1 µM ML210, or 5 µM JKE1675 with/without 6 µM selenite treatment for 2 hr. (b) Quantification of lipid peroxidation in 2 µM RSL3 or 5 µM ML210 treated Hela cells with/without 6µM selenite for indicated times. (c) Quantification of lipid peroxidation in 0.5 µM RSL3 or 1 µM ML210 treated NCIH838 cells with/without 6 µM selenite for 2 hr. (d) Viability of SK-Hep1 cells after treating with vehicle, 100 nM RSL3, or 100 nM ML210 with/without 10 µM Ferrostatin for 24 hr. (e) Viability of SK-Hep1 cells after treating with vehicle or 6 µM erastin with/without 3 µM selenide or 3 µM selenite+6 µM L-GSH for 72 hr. (f) Fluorescence image of PI/Hoechst double stained SK-Hep1 cells treated with 100nM RSL3 and/or 3 µM selenite for 24 h. Scale bar represents 100 µm. (g) ML210, JKE1675, and FIN56 dose response curve for vehicle or 3 µM selenite treated SK-Hep1 cells. Cell viability was measured at 24 hr after treatment with the GPX4 inhibitors. (h) ML210 dose response curve for vehicle or 3 µM selenite treated NCIH838, SNU449, Hela, A498, DU145, and LN229 cells. Cell viability was measured at 24 hr after treatment with the ML210. Data are mean ± S.D. from biological replicates (n = 3 for a-e,g,h) and were analyzed by two-tailed Student’s t-test.
Extended Data Fig. 3 xCT mediated thiol formation induces selenite to selenide reduction.
(a) Lead acetate embedded paper-based colorimetric selenide detection of different doses of selenide. Selenide results in brown coloration due to reaction with lead acetate. (b) Chemical structure of P3 probe originally developed for hydrogen sulfide detection12. Created with BioRender.com. (c) P3 probe-based selenide detection of different doses of selenide. Selenide results in increase of fluorescent intensity due to reaction with P3 probe. (d) Measurement of selenide levels using fluorescence based P3 probe after mixing with solutions of 0.5 mM selenite and/or 5 mM different metabolites as indicated. The selenite was added immediately after mixing P3 probe with thiol-containing metabolites (L-glutathione (L-GSH), L-Cysteine, β-mercaptoethanol (β-Mer), N-acetylcysteine), with negative controls of sulfur non thiol metabolites (Cystine, Methionine, Sulfite), or non-sulfur, non-thiol metabolite Glutamate. Values are relative to fluorescent intensity of P3 probe mixed with each metabolite without selenite (=1.0). (e) RNA expressions from Cancer Cell Line Encyclopedia of the xCT subunits SLC7A11 and SLC3A2, used to designate xCT high and low cells. (f) Doubling time of high and low xCT cell lines represented in e. Doubling time information was collected from https://www.cellosaurus.org/. (g) Immunoblot of xCT subunits, SLC7A11 and SLC3A2 in three high xCT and low xCT cell lines. (h) Total thiol measurement of conditioned media from high xCT and low xCT cell lines conditioned for 24 hr. Each value is relative to that of the unconditioned medium (UCM), set to 1. (i) Wells containing Ellman’s solution for total thiol quantification in the conditioned media in Fig. 2c, f. (j) Viability of high xCT cell lines treated with 3 µM erastin for 24 hr. (k) Immunoblot of GSS and GCLM from CTRL and GSS/GCLM KO SK-Hep1 cells. (l) Immunoblot of GPX4 in SK-Hep1 cells treated with/without 6 µM erastin and/or 1 µM selenite for 4 hr. (m) Immunoblots of GPX4 in CTRL, PSTK KO, and SEPH2 KO SK-Hep1 cells at 4 hr after the treatment of the indicated dosages of selenite. Values were normalized to that of vehicle treated cells (=1.0). Data are mean ± S.D. from biological replicates (n = 3 for c,d,h,j; n=13 for high xCT and n=11 for low xCT of f and were analyzed by two-tailed Student’s t-test. (n.s., not significant).
Extended Data Fig. 4 The selenium metabolite selenide reduces ubiquinone to ubiquinol via SQOR enzyme.
(a) Time progression images of 96 well plate containing 200 µM ubiquinone/nol solutions before and after selenide gas exposure. Timepoints post gas exposure are indicated at the right bottom side of each image. (b) Brightfield images of solution containing 1 µg/µl selenium powder (Se). 100X magnification. (c) UV-vis spectrophotometer analysis of varying doses of ubiquinone and ubiquinol solutions. (d) Ubiquinol/none ratio in ubiquinone solution mixed with vehicle, 10 mM sulfide or 10 mM selenide solution. (e) Left, list of enzymes known to reduce ubiquinone to ubiquinol, and their known substrate. Middle, proposed chemical reaction of hydrogen sulfide by SQOR. Right, a list of enzymes that process sulfur-containing metabolites51,43. (f) Immunoblot of SQOR in control and SQOR KO SK-Hep1 single clonal cells. (g) Schematic diagram of the experiment for testing the effect of selenite on ubiquinol (CoQ8) production in control and human SQOR-induced BL21 bacteria. Created with BioRender.com. (h) Immunoblot of SQOR protein in the hSQOR protein-induced bacteria. (i) Immunoblots of GPX4 or other selenoproteins in 6 µM selenite treated SK-Hep1 cells. (j) Relative ubiquinol/none (CoQ10) ratio in mitochondria from TXNRD1 KO SK-Hep1 cells treated with vehicle or 6 µM selenite for 2 hr. (k) Immunoblot of TXNRD1 in control and TXNRD1 KO cells. Data are mean ± S.D. from biological replicates (n = 3 for d) and were analyzed by two-tailed Student’s t-test. (n.s., not significant).
Extended Data Fig. 5 SQOR protects against ferroptosis.
(a) Measurement of lipid peroxidation in CTRL and SQOR KO SK-Hep1 cells treated with vehicle or 0.5 µM RSL3 for 2 hr. Bracketed bar indicates the gating for peroxidized lipids. (b) Dose response curves for the GPX4 inhibitors RSL3 in LN229 glioma cells overexpressing blank vector or SQOR. Cell viability was measured at 24 hr after treatment with the GPX4 inhibitors. (c–e) Left, viability of hydrogen sulfide production impaired CTRL and SQOR KO SK-Hep1 cells after 31 nM RSL3 treatment for 24 hr. Sulfide-producing enzyme genes, CBS (c), CTH (d), or MPST (e) were ablated by CRISPR-Cas9 in CTRL and SQOR KO SK-Hep1 cells. (f) 50 µM AOAA, an inhibitor for sulfide-producing enzymes was treated in CTRL and SQOR KO SK-Hep1 cells with vehicle or 31 nM RSL3 for 24 hr. (c-e) Right, immunoblot of (c) CBS, (d) CTH, and (e) MPST of CTRL and SQOR KO SK-Hep1 cells, which were subjected to additional KO with CTRL or CBS/CTH/MPST. (g) Viability of CTRL and SQOR KO SK-Hep1 cells treated with 100nM RSL3 and/or 40 µM Ferrostatin-1 (Fer-1) for 24 hr. (h) Viability of CTRL and SQOR KO SK-Hep1 cells treated with 100nM RSL3 and/or 20 µM Mitotempo for 24 hr. (i) Measurement of mitochondrial lipid peroxidation in CTRL and SQOR KO SK-Hep1 cells treated with vehicle or 0.5 µM RSL3 for 2 hr. Bracketed bar indicates the gating for quantifying mitochondrial lipid peroxidation. (j) Quantification of peroxidized lipids from panel i. Data are mean ± S.D. from biological replicates (n = 3 for c-g,h-j) and were analysed by two-tailed Student’s t-test.
Extended Data Fig. 6 SQOR protects against ferroptosis, supplemental data.
(a) BODIPY dye measurement of lipid peroxidation in 1 µM RSL3 or/and 6 µM selenite-treated SK-Hep1 cells with/without 50 µM Antimycin, concomitantly for 2 hr. Bracketed bar indicates the gating for lipid peroxidation. (b) BODIPY dye measurement of lipid peroxidation levels of SK-Hep1 cells with/without induced AOX overexpression in two independent clonal cell lines, as indicated in panel d, treated with/without 0.5 µM RSL3 and/or 6 µM selenite for 2 hr. (c) Quantification of lipid peroxidation level from AOX clone 8 in panel b. (d) Immunoblot of flag-tagged AOX protein. 10 single clones obtained after viral infection containing AOX expression construct were treated with doxycycline for 48h and collected samples for confirming the expression of AOX protein. Clone 1 and 8 were used for panels b and c. (e) Schematic diagram of enzymes involved in ubiquinone reduction to ubiquinol. The substrate and product of the enzymes are depicted. Created with BioRender.com. (f) Immunoblots of components of the electron transport chain and CoQ10 oxidoreductases, which affect the levels of ubiquinol, and key ferroptosis regulators in SQOR KO SK-Hep1 cells and SQOR OE/SOQR mut OE LN229 cells. ATP5A, ATP synthase lipid-binding protein; UQCRC2, Ubiquinol-cytochrome c reductase Core Protein 2; SDHB, Succinate dehydrogenase complex iron sulfur subunit B; COXII, Cytochrome c oxidase subunit 2; NDUFB8, NADH:Ubiquinone oxidoreductase subunit B8; NDUFS4, NADH:Ubiquinone oxidoreductase subunit S4; PRODH, proline dehydrogenase; G3PDH, Glyceraldehyde-3-phosphate dehydrogenase; ETFA, Electron transfer flavoprotein subunit alpha; DHODH, Dihydroorotate dehydrogenase; FSP1, Ferroptosis Suppressor Protein 1; GPX4, Glutathione peroxidase 4. (g) Mitotracker dye quantification of mitochondria in CTRL or SQOR KO SK-Hep1 cells and Vector, SQOR OE, or SQOR mut OE LN229 cells. Bracketed bar indicates the gating for increased mitochondria population. Cells incubated with 1% serum for 36 hr were used as a control. (h) Quantification of high mitochondria population from in panel g. (i) BODIPY dye measurement of lipid peroxidation in CTRL and DHODH KO SK-Hep1 cells treated with 0.2 µM RSL3 and/or 6 µM selenite for 2 hr. SK-Hep1 cells at 5 days after infection of virus containing guide RNA targeting DHODH were used. Bracketed bar indicates the gating for lipid peroxidation. (j) Quantification of lipid peroxidation from panel i. (k) Immunoblot of DHODH in CTRL and DHODH KO SK-Hep1 cells. Data are mean ± S.D. from biological replicates (n = 3 for c,h,j) and were analyzed by two-tailed Student’s t-test. (n.s., not significant).
Extended Data Fig. 7 Selenide protects hippocampal neurons from glutamate induced ROS, lipid peroxidation and membrane depolarization.
(a) Viability of mouse hippocampal neuronal HT29 cells after 5 mM glutamate treatment for 24 hr with/without 1 µM selenide, relative to vehicle treated cells (=1.0). (b) BODIPY dye measurement of lipid peroxidation in HT29 cells treated with 5 or 25 mM glutamate and/or 3 µM selenide for 12 hr. Bracketed bar indicates the gating for peroxidized lipids. (c) Quantification of lipid peroxidation from panel b. (d) DCFDA dye measurement of ROS in HT29 cells treated with 5 mM glutamate and/or 3 µM selenide for 12 hr. Bracketed bar indicates the gating for high ROS population. (e) Quantification of high ROS population from panel d. (f) TMRE dye measurement of mitochondrial membrane potential in HT29 cells treated with 50 µM FCCP or 5 mM glutamate and/or 3 µM selenide for 12 hr. The uncoupler FCCP was used as a control. Bracketed bar indicates the gating for the depolarized population. (g) Quantification of the depolarized population from panel f. (h) Immunocytochemistry of Bid protein, mitochondria, and nucleus in HT29 cells treated with 5 mM glutamate and/or 3 µM selenide for 12 hr. Scale bar indicates 20 µm. (i) Quantification of Bid protein localized to mitochondria from panel h. (j) Quantification of Bid protein expression from panel h. Data are mean ± S.D. from biological replicates (n = 3 for a,c,e,g; n = 10 for i,j) and were analyzed by two-tailed Student’s t-test. (n.s., not significant).
Supplementary information
Supplementary Information
Supplementary discussion, references and Tables 1–4.
Supplementary Tables 1–3
Table 1. List of materials and primer/guide sequence information used in the study. Table 2. List of 165 metabolites of SK-Hep1 cells treated with vehicle or selenite. Table 3. List of lipid profiles of SK-Hep1 cells treated with vehicle or selenite.
Source data
Source Data Fig. 1
Statistical Source Data.
Source Data Fig. 2
Statistical Source Data.
Source Data Fig. 3
Statistical Source Data.
Source Data Fig. 4
Statistical Source Data.
Source Data Fig. 5
Statistical Source Data.
Source Data Fig. 6
Statistical Source Data.
Source Data Extended Data Fig. 1
Statistical Source Data.
Source Data Extended Data Fig. 2
Statistical Source Data.
Source Data Extended Data Fig. 3
Statistical Source Data.
Source Data Extended Data Fig. 4
Statistical Source Data.
Source Data Extended Data Fig. 5
Statistical Source Data.
Source Data Extended Data Fig. 6
Statistical Source Data.
Source Data Extended Data Fig. 7
Statistical Source Data.
Source Data Fig. 1 and Extended Data Figs. 1 and 3–6
Source data supporting blots.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, N., Park, S.J., Lange, M. et al. Selenium reduction of ubiquinone via SQOR suppresses ferroptosis. Nat Metab 6, 343–358 (2024). https://doi.org/10.1038/s42255-024-00974-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-024-00974-4
This article is cited by
-
Selenium abandons selenoproteins to inhibit ferroptosis rapidly
Nature Metabolism (2024)