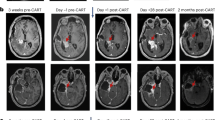
Abstract
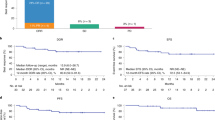
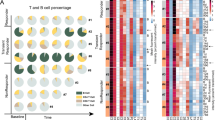
We previously showed that chimeric antigen receptor (CAR) T-cell therapy targeting epidermal growth factor receptor variant III (EGFRvIII) produces upregulation of programmed death-ligand 1 (PD-L1) in the tumor microenvironment (TME). Here we conducted a phase 1 trial (NCT03726515) of CAR T-EGFRvIII cells administered concomitantly with the anti-PD1 (aPD1) monoclonal antibody pembrolizumab in patients with newly diagnosed, EGFRvIII+ glioblastoma (GBM) (n = 7). The primary outcome was safety, and no dose-limiting toxicity was observed. Secondary outcomes included median progression-free survival (5.2 months; 90% confidence interval (CI), 2.9–6.0 months) and median overall survival (11.8 months; 90% CI, 9.2–14.2 months). In exploratory analyses, comparison of the TME in tumors harvested before versus after CAR + aPD1 administration demonstrated substantial evolution of the infiltrating myeloid and T cells, with more exhausted, regulatory, and interferon (IFN)-stimulated T cells at relapse. Our study suggests that the combination of CAR T cells and PD-1 inhibition in GBM is safe and biologically active but, given the lack of efficacy, also indicates a need to consider alternative strategies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
scRNAseq and TCRseq data that support the findings of this study have been deposited in the Gene Expression Omnibus under accession code GSE242790. Source data for Figs. 1–5 and Extended Data Fig. 1 have been provided as source data files. All other data supporting the findings of this study including de-identified individual participant clinical data are available from the corresponding author upon reasonable request. Source data are provided with this paper.
Code availability
The scRNAseq and TCRseq analyses presented in the paper were performed with open-source algorithms as described in Methods. Further details will be made available by the authors on request. No custom code was generated in the course of this study.
References
van de Donk, N., Usmani, S. Z. & Yong, K. CAR T-cell therapy for multiple myeloma: state of the art and prospects. Lancet Haematol. 8, e446–e461 (2021).
June, C. H. & Sadelain, M. Chimeric antigen receptor therapy. N. Engl. J. Med. 379, 64–73 (2018).
Bagley, S. J. & O’Rourke, D. M. Clinical investigation of CAR T cells for solid tumors: Lessons learned and future directions. Pharmacol. Ther. 205, 107419 (2020).
Hou, A. J. et al. Navigating CAR-T cells through the solid-tumour microenvironment.Nat. Rev. Drug Discov. 20, 531–550 (2021).
Bagley, S. J. et al. CAR T-cell therapy for glioblastoma: recent clinical advances and future challenges. Neuro. Oncol. 20, 1429–1438 (2018).
Ostrom, Q. T. et al. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2013-2017. Neuro. Onco.l 22, iv1–iv96 (2020).
Stupp, R. et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 352, 987–996 (2005).
Wen, P. Y. et al. Glioblastoma in adults: a Society for Neuro-Oncology (SNO) and European Society of Neuro-Oncology (EANO) consensus review on current management and future directions. Neuro. Oncol. 22, 1073–1113 (2020).
O’Rourke, D. M. et al. A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci. Transl. Med. 9, eaaa0984 (2017).
Ahmed, N. et al. HER2-specific chimeric antigen receptor-modified virus-specific T cells for progressive glioblastoma: a phase 1 dose-escalation trial. JAMA Oncol. 3, 1094–1101 (2017).
Brown, C. E. et al. Bioactivity and safety of IL13Rα2-redirected chimeric antigen receptor CD8+ T cells in patients with recurrent glioblastoma. Clin. Cancer Res. 21, 4062–4072 (2015).
Brown, C. E. et al. Regression of glioblastoma after chimeric antigen receptor T-cell therapy. N. Engl. J. Med. 375, 2561–2569 (2016).
Durgin, J. S. et al. Case Report: Prolonged survival following EGFRvIII CAR T cell treatment for recurrent glioblastoma. Front. Oncol. 11, 669071 (2021).
Felsberg, J. et al. Epidermal growth factor receptor variant III (EGFRvIII) positivity in EGFR-amplified glioblastomas: prognostic role and comparison between primary and recurrent tumors. Clin. Cancer Res. 23, 6846–6855 (2017).
Li, G. & Wong, A. J. EGF receptor variant III as a target antigen for tumor immunotherapy. Expert Rev. Vaccines 7, 977–985 (2008).
Koga, T. et al. Mapping of genomic EGFRvIII deletions in glioblastoma: insight into rearrangement mechanisms and biomarker development. Neuro. Oncol. 20, 1310–1320 (2018).
Tang, O. Y. et al. PD1 expression in EGFRvIII-directed CAR T cell infusion product for glioblastoma is associated with clinical response. Front. Immunol. 13, 872756 (2022).
Weller, M. et al. Rindopepimut with temozolomide for patients with newly diagnosed, EGFRvIII-expressing glioblastoma (ACT IV): a randomised, double-blind, international phase 3 trial. Lancet Oncol. 18, 1373–1385 (2017).
Louis, D. N. et al. The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro. Oncol. 23, 1231–1251 (2021).
Fraietta, J. A. et al. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat. Med. 24, 563–571 (2018).
Giles, J. R. et al. Human epigenetic and transcriptional T cell differentiation atlas for identifying functional T cell-specific enhancers. Immunity 55, 557–574 (2022).
Alanio, C. et al. Immunologic features in de novo and recurrent glioblastoma are associated with survival outcomes. Cancer Immunol. Res. 10, 800–810 (2022).
Chong, E. A. et al. Pembrolizumab for B-cell lymphomas relapsing after or refractory to CD19-directed CAR T-cell therapy. Blood 139, 1026–1038 (2022).
Adusumilli, P. S. et al. A phase I trial of regional mesothelin-targeted CAR T-cell therapy in patients with malignant pleural disease, in combination with the anti-PD-1 agent pembrolizumab. Cancer Discov. 11, 2748–2763 (2021).
Heczey, A. et al. CAR T cells administered in combination with lymphodepletion and PD-1 inhibition to patients with neuroblastoma. Mol. Ther. 25, 2214–2224 (2017).
Amini, L. et al. Preparing for CAR T cell therapy: patient selection, bridging therapies and lymphodepletion. Nat. Rev. Clin. Oncol. 19, 342–355 (2022).
Huang, J. et al. Clinical and dosimetric predictors of acute severe lymphopenia during radiation therapy and concurrent temozolomide for high-grade glioma. Int. J. Radiat. Oncol. Biol. Phys. 92, 1000–1007 (2015).
Eskilsson, E. et al. EGFR heterogeneity and implications for therapeutic intervention in glioblastoma. Neuro Oncol 20, 743–752 (2018).
Leahy, A. B. et al. CD19-targeted chimeric antigen receptor T-cell therapy for CNS relapsed or refractory acute lymphocytic leukaemia: a post-hoc analysis of pooled data from five clinical trials. Lancet Haematol. 8, e711–e722 (2021).
Liau, L. M. et al. Association of autologous tumor lysate-loaded dendritic cell vaccination with extension of survival among patients with newly diagnosed and recurrent glioblastoma: a phase 3 prospective externally controlled cohort trial. JAMA Oncol. 9, 112–121 (2023).
Nerreter, T. et al. Super-resolution microscopy reveals ultra-low CD19 expression on myeloma cells that triggers elimination by CD19 CAR-T. Nat. Commun. 10, 3137 (2019).
Grosser, R. et al. Combination immunotherapy with CAR T cells and checkpoint blockade for the treatment of solid tumors. Cancer Cell 36, 471–482 (2019).
Kalinin, R. S. et al. Engineered removal of PD-1 from the surface of CD19 CAR-T cells results in increased activation and diminished survival. Front. Mol. Biosci. 8, 745286 (2021).
Wei, J. et al. PD-1 silencing impairs the anti-tumor function of chimeric antigen receptor modified T cells by inhibiting proliferation activity. J. Immunother. Cancer 7, 209 (2019).
Ellingson, B. M., Wen, P. Y. & Cloughesy, T. F. Modified criteria for radiographic response assessment in glioblastoma cinical trials. Neurotherapeutics 14, 307–320 (2017).
Garfall, A. L. et al. Anti-BCMA/CD19 CAR T cells with early immunomodulatory maintenance for multiple myeloma responding to initial or later-line therapy.Blood Cancer Discov. 4, 118–133 (2023).
Butler, A. et al. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
Hafemeister, C. & Satija, R. Normalization and variance stabilization of single-cell RNA-seq data using regularized negative binomial regression. Genome Biol. 20, 296 (2019).
Korsunsky, I. et al. Fast, sensitive and accurate integration of single-cell data with Harmony. Nat. Methods 16, 1289–1296 (2019).
Tran, H. T. N. et al. A benchmark of batch-effect correction methods for single-cell RNA sequencing data. Genome Biol. 21, 12 (2020).
Hanley, J. A. & Lippman-Hand, A. If nothing goes wrong, is everything all right? Interpreting zero numerators. JAMA 249, 1743–1745 (1983).
Eypasch, E. et al. Probability of adverse events that have not yet occurred: a statistical reminder. Brit. Med. J. 311, 619–620 (1995).
Kaplan, E. L. & Meier, P. Nonparametric estimation from incomplete observations. J. Am. Stat. Assoc. 53, 457–481 (1958).
Acknowledgements
We thank the patients who participated in this study and their families for their dedication to furthering GBM treatment. We also thank the Neurosurgery Clinical Research Division, J. Fraietta and the Translational and Correlative Sciences Laboratory and A. Brennan and the Clinical Cell and Vaccine Production Facility at the University of Pennsylvania Perelman School of Medicine for all of their clinical trial contributions and support. This work was funded by Novartis, the Abramson Cancer Center Glioblastoma Translational Center of Excellence, The Templeton Family Initiative in Neuro-Oncology, the Maria and Gabriele Troiano Brain Cancer Immunotherapy Fund, the Parker Institute for Cancer Immunotherapy (PICI Award Number C-01919), a public grant overseen by the French National Research Agency (Agence Nationale de la Recherche (ANR)) as part of the Investment Programme France 2030 under grant agreement ANR-21-RHUS-0016, and the French National Cancer Institute, the Ministry of Health and Prevention and Interm. High-throughput sequencing was performed by the ICGex NGS platform of the Institut Curie supported by the grants ANR-10-EQPX-03 (Equipex) and ANR-10-INBS-09-08 (France Génomique Consortium) from the ANR (“Investissements d’Avenir” program), by the ITMO-Cancer Aviesan (Plan Cancer III) and by the SiRIC-Curie program (SiRIC Grant INCa-DGOS-465 and INCa-DGOS- Inserm_12554). We also acknowledge ANR-10-IDEX-0001-02 PSL, CIC IGR-Curie 1428, INCa-DGOS-Inserm-ITMO cancer_18000 and LabEx DCBIOL. We thank the Parker Institute for Cancer Immunotherapy members for all of their constructive feedback, as well as Clinical Immunology laboratory and U932 at the Institut Curie (Paris, France) for insightful discussions, O. Lantz for critical feedback on the ancillary work, N. Amzallag for her support and the genomic and cytometry platforms of Institut Curie for their help.
Author information
Authors and Affiliations
Contributions
S.J.B., A.S.D., E.M., A.M., J.L.B., E.H. and D.M.O. designed the study. S.J.B., A.S.D., E.M., S.B., R.L., G.K., M.A.B., S.M., E.H. and D.M.O. recruited and treated patients in the study. L.L., C.A., Z.A.B., M.P.N., S.K., W.T.H., O.Y.T., M.L., M.B., K.M. and D.D. generated, curated and analyzed data. C.A.,P.E.B., C.G., W.R., E.M., D.M., E.P. and J.J.W. performed bioinformatic analyses. S.J.B., Z.A.B.,L.L. and C.A. wrote the original paper, and S.J.B., Z.A.B., L.L., M.P.N., S.B., S.M., S.K., E.J.W, W.T.H., O.Y.T.,E.P,.J.J.W., S.A.,D.M., G.L.B., J.L.B., C.A. and D.M.O. reviewed and edited the paper. S.J.B., Z.A.B., G.L.B., E.J.W., D.M., C.A. and D.M.O. supervised the study. Funding support was provided by C.A. and J.L.B. C.A. and D.M.O. contributed equally to this study.
Corresponding authors
Ethics declarations
Competing interests
S.J.B. has received consulting fees from Kiyatec, Novocure and Bayer and has received research funding from Incyte, Tmunity, Novocure, GSK and Eli Lilly, all outside of the submitted work. S.J.B., A.S.D. and D.M.O. have inventorship interest in intellectual property owned by Novartis and the University of Pennsylvania: U.S. patent 62/809,245: “Combination therapies of EGFRvIII CARs and PD-1 inhibitors”. Z.A.B. has inventorship interest in intellectual property owned by the University of Pennsylvania and has received royalties related to CAR T-cell therapy in solid tumors. S.M. has received consulting fees from AI Integrated Radiological Solutions Medial and Qynapse SAS and has received research funding from Novocure and Galileo CDS, all outside of the submitted work. G.L.B. has received consulting fees from Seattle Genetics (now Seagen), Adicet Bio, Aduro Biotech, AstraZeneca, BiolineRx, BioMarin Pharmaceuticals, Bristol-Myers Squibb, Cantargia, Cour Pharmaceuticals, Boehinger Ingelheim, Genmab, Hibercell, HotSpot Therapeutics, Incyte Corporation, Janssen, Merck, Molecular Partners, NanoGhost, Pancreatic Cancer Action Network, Shattuck Labs and Verastem and reports receiving research funding from Incyte Corporation, Bristol-Myers Squibb, Verastem, Halozyme, Biothera, Newlink, Novartis, Arcus Biosciences and Janssen, all outside of the submitted work. E.J.W. receives honoraria and/or research support from BMS, Merck, MedImmune, Surface Oncology, Takeda and KyMab. E.J.W. has a patent licensing agreement for the PD-1 pathway. J.L.B. reports employment with, is a current holder of stock options and holds patents with Novartis Institutes for Biomedical Research. D.M. is inventor of patents in the field of cell and gene therapy filed by the University of Pennsylvania and the University of Geneva, is scientific co-founder of Cellula Therapeutics and is a member of the scientific advisory board of MPC, Limula and Cellula Therapeutics. D.M.O. has received consulting fees from Celldex Therapeutics, Prescient Therapeutics and Century Therapeutics; has received research funding from Celldex Therapeutics, Novartis and Tmunity Therapeutics; and is an inventor of intellectual property (U.S. patent numbers 7,625,558 and 6,417,168 and related families) and has received royalties related to targeted ErbB therapy in solid cancers previously licensed by the University of Pennsylvania. C.A. is a consultant for Biotherapy Partners. All other authors declare no competing interests.
Peer review
Peer review information
Nature Cancer thanks Jon Amund Kyte, Wendy London, Julie Park and Mario Suva for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 CAR T cells in the infusion products are activated and exhausted effector T cells, with comparable features across patients.
(a) Representative example of EGFRvIII CAR T cells staining in the infusion product. (b) Scatterplot shows proportion of EGFRvIII CAR T cells staining in the infusion products of the patients. (c) Representative example of CD4/CD8 staining in EGFRvIII CAR T cells staining in the infusion product. (d) Scatterplot shows proportion of CD4 T cells within EGFRvIII CAR T cells in the infusion products of the patients. (e) Representative example of CD127/CD25 regulatory T cells staining in the infusion product. (f) Representative example of Foxp3 expression in the different CD4 T-cell subsets in the infusion product. (g) Expression of CD4, CD40LG, Foxp3 in the infusion product as detected by RNA in scRNAseq data projected on a UMAP. (h) Representative example of CD45RA/CD127 staining in CD4 CAR T cells in the infusion product (P1, left; P4, right). (i) Representative example of CD45RA/CD127 staining in CD8 CAR T cells in the infusion product (P1, left; P4, right). (j) Representative example of PD1, CD39, Ki67 staining in CD8 T cells in the infusion product (P1, top; P4, bottom). (k) Heatmap shows expression in CD8 T cells in the infusion product of P1 and P4 as compared to a normal donor.
Extended Data Fig. 2 Bioinformatic analysis.
(a) Example of our strategy for detecting CAR T cells by RNA on scRNAseq data, here applied to one healthy donor where we spiked small amount of CAR T cells. (b) Clonal space homeostasis indicating percentage of clones in distinct proportions in the six paired samples. (c) Clonal Proportion where clonotypes are ranked by copy or frequency of occurrence in the six paired samples. (d) UMAP shows the overall cell space for the TME of three paired patients as in Fig. 4c, here split by 3 Pre (left) and 3 Post (right). (e) UMAP shows the overall cell space for the TME of three paired patients as in Fig. 4c, here split by sample.
Extended Data Fig. 3 Increased myeloid inflammation with therapy.
(a) UMAP shows the overall cell space for the TME of three paired patients (3 Pre and Post, total six samples) after filtering myeloid cells from the overall UMAP as in Fig. 4c. (b) Top 10 genes for each cluster as in A. (c) Gene expression on top of each cluster as in A. GSEA of cluster 0 (d), 2 (e), and 3 (f) as defined in A. GSEA employs a permutation-based test usng Kolmogorov-Smirnov; Benjamini-Hochberg procedure was used to adjust for multiple comparisons. (g) UMAP shows the overall cell space for the myeloid TME of three paired patients as in A, here split by 3 Pre (left) and 3 Post (right).
Extended Data Fig. 4 Flow cytometry analysis of PD-1 expression.
(a) Flow cytometry gating schema for quantification of CD4+/CAR+ PD-1 expression in infusion product. (b) Exemplary flow cytometry plots taken from 6 total samples.
Supplementary information
Supplementary Information
Study protocol, statistical analysis plan, and CONSORT diagram.
Supplementary Tables
Supplementary Table 1: Markers for Cytek panel. Supplementary Table 2: Antibodies and staining reagents used for flow cytometry.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Extended Data Fig./Table 1
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bagley, S.J., Binder, Z.A., Lamrani, L. et al. Repeated peripheral infusions of anti-EGFRvIII CAR T cells in combination with pembrolizumab show no efficacy in glioblastoma: a phase 1 trial. Nat Cancer 5, 517–531 (2024). https://doi.org/10.1038/s43018-023-00709-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43018-023-00709-6
This article is cited by
-
Mechanistic insights and the clinical prospects of targeted therapies for glioblastoma: a comprehensive review
Experimental Hematology & Oncology (2024)
-
Immunotherapy for Brain Tumors: Where We Have Been, and Where Do We Go From Here?
Current Treatment Options in Oncology (2024)