Abstract
This study examined a pair of neighbouring small watersheds with contrasting vegetations: artificial forestland and natural grassland. Since 1954, afforestation which mainly planted with black locust has been conducted in one of these watersheds and natural revegetation in the other. The differences in soil total N, nitrate, ammonium, foliar litterfall δ15N and dual stable isotopes of δ15N and δ18O in soil nitrate were investigated in the two ecosystems. Results showed that there was no significant difference in soil total N storage between the two ecosystems, but the black locust forestland presented higher soil nitrate than the grassland. Moreover, the foliar litterfall N content and δ15N of the forestland were significant higher than the grassland. These results indicate that 60 years of watershed black locust afforestation have increased soil N availability. The higher nitrate in the forestland was attributed to the biological N fixation of black locust and difference in ecosystem hydrology. The dual stable isotopes of δ15N and δ18O revealed that the two ecosystems had different sources of soil nitrate. The soil nitrate in the forestland was likely derived from soil N nitrification, while the soil nitrate in the grassland was probably derived from the legacy of NO3− fertiliser.
Similar content being viewed by others
Introduction
Afforestation, which is the conversion of historically treeless areas into forests, is the most important forestry practice contributing to vegetation restoration and ecosystem rehabilitation. In humid regions, afforestation is thought to be appropriate for promoting ecosystem restoration and carbon sequestration. However, in arid and semiarid regions, this method has raised grave concerns1,2,3. Numerous studies have demonstrated that dryland afforestation can exacerbate water shortages and lead to the deterioration of soil ecosystems in many cases4,5,6. In addition to water depletion, dryland afforestation could also consume large amounts of soil nutrients, especially nitrogen (N)7,8. Previous studies have demonstrated that afforestation or forest regrowth can deplete soil N pools9,10 and/or decrease soil N availability11,12,13. In drylands, available soil N is low14,15; thus, progressive N limitation would be more serious as a result of afforestation. However, the extent to which soil N availability will change during drylands afforestation remains poorly understood. A better understanding of these processes will assist in assessing the potential of soil N limitation in forest ecosystem construction.
An alternative to afforestation that also promotes ecosystem restoration and carbon sequestration is the protection and facilitation of natural revegetation on abandoned lands16,17. In contrast to afforestation, natural revegetation represents a natural process of recovering ecosystem function and can therefore be used as a control for the study of ecosystem biogeochemical cycles. Exploring the differences in soil N storage and availability between natural and artificial measures can provide information that is valuable for the prediction of changes in soil N cycling. Studies by Zhang et al.18,19 demonstrated that natural revegetation in humid areas improves soil N nutrient levels to a greater extent than tree plantation. Moreover, Li et al.13 found that cropland afforestation in humid areas could lead to progressive N limitation. Drylands occupy 47 percent of the surface of the earth20. However, information on the effects of the natural and artificial management practices on soil N in drylands is still limited. Moreover, few studies have used the dual stable isotopes of δ15N and δ18O to characterise the changes in soil N cycling during drylands afforestation or natural revegetation.
Recently, several studies have demonstrated that foliar δ15N is a useful indicator of ecosystem N availability21,22,23. The basic premise is that δ15N measured in organic material characterises the fractionation processes24,25. When the N supply is high relative to biotic demand, N is lost through fractionating pathways; the remaining ecosystem N is enriched in 15N26. Therefore, ecosystems with high N availability typically exhibit high δ15N values in plant tissues27. Moreover, dual stable isotopes (δ15N and δ18O) of nitrate have been used successfully to identify the sources and transformation processes of nitrate in water and terrestrial ecosystems28,29,30,31. In many cases, the dual stable isotopes offer a direct means of source identification because different sources of nitrate often have different isotopic compositions32. For example, nitrate and ammonium fertilisers, animal and human wastes, and soils have distinct δ15N and δ18O levels, which can be used to distinguish the sources of nitrate32.
The Loess Plateau of China is a unique geographical area that is characterised by an extensive loess distribution, severe soil erosion and low vegetation coverage. Since the 1950s, the Chinese government has made great efforts to control soil erosion and restore vegetation, including implementing large-scale tree plantation in the 1970s, integrated soil erosion control in the 1980s and 1990s and the Grain for Green Project in the 2000s33,34. The most recent study by Lü et al.35 demonstrated that a total of 8.69 × 105 ha of cropland was converted to forestland on the Loess Plateau between 2000 and 2008. This extensive afforestation has been reported to decrease regional water yield and deplete soil water resources35,36. However, it is unknown whether several decades of afforestation will lead to more serious N limitation along with soil water deficits. The resulting information will greatly assist in understanding the effects of dryland afforestation on soil N cycling.
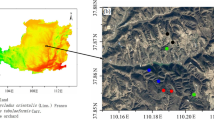
In this study, a pair of neighbouring small watersheds with similar topographical and geological backgrounds on the Loess Plateau were selected and used to compare the effects of artificial affestatation and natural revegetation on soil N storage and availability (Fig. 1). Since 1954, afforestation which mainly planted with black locust has been conducted in one of these watersheds and natural revegetation in the other. The two watersheds have formed completely different ecosystems: black locust forestland and natural grassland. In a previous study, we found that the two ecosystems have different patterns of soil carbon cycling and the grassland is more beneficial to soil surface organic and inorganic carbon sequestration than the forestland37. The objectives of this study were (1) to examine the difference in soil N storage and availability between the two ecosystems, and (2) to use the dual stable isotopes of δ15N and δ18O to identify the soil nitrate source.
Map showing the location of the study area in Xifeng District, Qingyang city, Gansu province, including the watersheds of forestland and grassland (Chinese map in the figure was created by ArcGIS 9.3 software, http://www.arcgis.com/features/; satellite image in the figure derives from Google Earth and copyright of the satellite image belongs to Google Earth and CNES/Astrium).
Results
Soil physical and chemical properties in the two ecosystems
Significant differences were identified in the physical and chemical properties of the soils in the two ecosystems (Table 1). The black locust forestland had higher soil bulk density and lower soil organic carbon (SOC), soil moisture and C/N compared with the natural grassland. However, no significant difference between the two ecosystems could be identified with respect to the soil pH and total N concentrations (STN).
Soil total N, nitrate and ammonium content in the two ecosystems
The soil total N content exhibited no significant difference between the forestland and grassland ecosystems (Fig. 2a,b, P = 0.702). The storages of soil total N down to a depth of 1 m were 6.93 and 7.65 Mg ha−1 for the black locust forestland and natural grassland, respectively. The concentrations of soil nitrate were significantly different between the two ecosystems, with the content of soil nitrate being significantly higher in the black locust forestland than in the natural grassland (Fig. 2d, P = 0.003), with mean values at a depth of 1 m of 3.95 and 0.79 mg kg−1, respectively. Moreover, the soil ammonium content exhibited no significant difference between the forestland and grassland ecosystems (Fig. 2c, P = 0.499).
Foliar N content and natural abundance of 15N in the two ecosystems
In this study, the black locust forestland exhibited a higher foliar litterfall N content and δ15N than the natural grassland (Fig. 3, P < 0.0001). The mean foliar N content and δ15N in the black locust forestland were 3.31% and -1.14‰, respectively, and 0.84% and −6.20‰, respectively, for the natural grassland.
Natural abundance of soil nitrate 15N and 18O in the two ecosystems
In this study, soil nitrate δ15N and δ18O also exhibited a large difference between the forestland and grassland ecosystems (Fig. 4). The nitrate δ15N level ranged from −1.37‰ to 2.57‰ in the black locust forestland and from −2.44‰ to 1.96‰ in the natural grassland. The δ18O level ranged from 3.05‰ to 13.16‰ in the black locust forestland and from 10.80‰ to 32.61‰ in the natural grassland.
Discussion
In this study, the artificial forestland and natural grassland provides a unique opportunity to examine the effects of long-term dryland afforestation and natural revegetation on soil N storage and availability. Our results showed that the two ecosystems exhibited no significant difference in soil total N storage (Fig. 2b), while the content of soil nitrate in the black locust forestland was significantly higher than in the natural grassland (Fig. 2d). Davidson et al.21,38,39 demonstrated that higher nitrate in ecosystems typically indicates excess available N relative to plant demand and higher soil N availability. Rather than decreasing soil N availability, the higher nitrate content of the black locust forestland investigated in this study indicates that 60 years of watershed black locust afforestation have increased soil N availability. Moreover, the results of the foliar litterfall δ15N analysis demonstrated that the black locust forestland had a higher foliar N content and δ15N level than the natural grassland (Fig. 3), which further proved that the forestland had higher soil N availability than the grassland.
In a previous study, Jiao et al.6 assessed the ecological success of restoration by afforestation on the northern Loess Plateau and found that afforestation with black locust offered few additional advantages when compared with natural recovery sites. Moreover, Wei et al.40 showed that the conversion of grasslands to pine woodlands on the northern Loess Plateau reduced soil N availability. However, Liu et al.41 evaluated the ecological functions of black locust on the Loess Plateau and found that black locust had the potential to improve soil N availability. Besides, Qiu et al.42 demonstrated that afforestation with black locust in loessial gully region of the Loess Plateau obviously improved soil N levels and the improvements were greater in long-term than middle-term black locust stands. In this study, we conclude that two factors have led to the increased soil N availability in the forestland. One factor is the excess N input due to biological N fixation of black locust. In the forestland, the main planted species is black locust, which is well known to be a symbiotic N fixer. Previous studies have demonstrated that black locust has high rates of biological N fixation43,44. Lopez et al.45 showed that approximately 10 years after black locust establishment, soil N had already been enriched by black locust N. In this study, the plantation age is approximately 60 years; thus, the forestland N has probably been enriched by black locust N. Therefore, the biological N fixation by black locust could be the dominant factor contributing to the increased soil N availability in the forestland. The second factor is the difference in ecosystem hydrology. Many studies have demonstrated that the two ecosystems exhibit a large difference in hydrology46,47. The annual runoff in the forestland was observed to be significantly lower than in the grassland37,48,49. Moreover, the soil water content of the forestland was lower than in the grassland, and soil desiccation had clearly occurred in the forestland due to higher forest transpiration and canopy interception37,46,47. The poor soil water conditions and low runoff have probably decreased the soil nitrate loss and improved the nitrate accumulation in the forestland.
Kendall et al.32 reviewed that different sources of N had different ranges of δ15NNO3 and δ18ONO3 values and therefore could be isotopically distinguishable. Regarding atmospheric N, the δ15N values of atmospheric NO3− and NH4+ are usually in the range of −15 to 15‰32. Moreover, the δ18ONO3 values of precipitation surveyed in the late 1990s range from 14 to 75‰50, while the δ18ONO3 values in precipitation across the USA reported more recently ranged from +63 to +94‰, with a mean value of +76.3‰51. About inorganic fertilisers, the element of N derives from atmospheric N2 and therefore the δ15N values are usually low, generally in the range of −4 to +4‰32. The synthetic fertilisers, where the O is mostly derived from atmospheric O2 (ca. +23.5‰), have δ18O values ranging from +17 to +25‰32 Moreover, nitrate derived from nitrification of ammonium fertilisers has lower δ18O values, usually in the range of −5 to +15‰32. About soils, the δ15N of total soil N ranges from about −10 to +15‰32. Soluble dissolved inorganic nitrogen (mainly NO3−) represents a very small pool while it is more important than the larger organic pool to maintain ecosystem N availability. The δ15N of soil nitrate ranges from about −10 to +15‰, with most soils having δ15NNO3 values in the range of +2 to +5‰32,50.
In this study, the δ15N and δ18O levels in soil nitrate suggested that the two ecosystems were influenced by different sources of nitrate. The nitrate δ15N levels in the forestland and grassland were nearly zero. However, the soil nitrate was more enriched with heavy oxygen isotopes in the grassland than in the forestland (Fig. 4). In comparison with the typical nitrate δ15N and δ18O levels derived or nitrified from various N sources32, we found that the soil nitrate of the grassland was more influenced by NO3− fertilisers because δ18ONO3 values of the grassland falls in the range of δ18O values of NO3− fertiliser (Fig. 5). However, the soil nitrate of the forestland was more influenced by soil N transformation and nitrification (Fig. 5). In the forestland, most of the excessive N derives from the biological N fixation of black locust; thus, the nitrate δ15N and δ18O signals represent the characteristics of soil N transformation and nitrification; while in the grassland, the dual stable isotopes are indicative of NO3− fertiliser, suggesting that NO3− fertilisers had been applied to the grassland or the former farmland.
Materials and Methods
Study site
This study was conducted in the Nanxiaohe Basin, which is located in the Xifeng District of Qingyang city, Gansu province. The region has a semi-arid continental climate; the mean annual temperature and precipitation are 9.3 °C and 556.5 mm, respectively. Approximately 67.3% of the annual precipitation occurs from June to September. The area has a loess gully landscape with elevations varying from 1,050 m to 1,423 m. The soil layer is approximately 250 m thick, and the soil is silt-loamy52.
In the basin, a pair of neighbouring small watersheds with similar topographical and geological backgrounds, the Dongzhuanggou (DZG) watershed and Yangjiagou (YJG) watershed, were selected for this study. DZG is 1.6 km long and covers an area of 1.15 km2. Since 1954, DZG has been subject to natural revegetation measures and currently supports grassland vegetation; the principal grass species are Arundinella hirta, Agropyron cristatum and Artemisia argyi. YJG is 1.5 km long and covers an area of 0.87 km2. The principal afforestation activities in YJG occurred from 1954 to 1958; the current timber volume is 4,000 m3 52. The principal planted species is black locust (Robinia pseudoacacia L.). Following 60 years of vegetation restoration, the two small watersheds have formed completely different vegetation landscapes (YJG: artificial forestland; DZG: natural grassland).
Soil sampling and laboratory analysis
Soil sampling was performed in May and September of 2013. To determine the average content and vertical distribution of soil total N (STN), 14 sampling sites were established in the forestland watershed and 14 in the grassland watershed. In the two watersheds, the area of gully slopes occupies more than 65% of the total area of the watersheds49 and the measures of black locust afforestation and natural revegetation have been mainly conducted in the area. Therefore, the sampling sites were randomly distributed on the gully slopes, which could represent the pure ecosystems of black locust forestland and natural grassland. In the study, soil samples were collected to a depth of 1 m. The soils were sampled at intervals of 10 cm using a hand-held auger (6 cm in diameter), and 10 soil samples were obtained at each site. Accordingly, 140 soil samples were collected in the forestland watershed and 140 in the grassland watershed. To determine the soil nitrate and ammonium contents, bulk density and pH, three soil profiles at a depth of 0–100 cm were established in each of the forestland and grassland watersheds. For the soil bulk density analysis, three replicate samples were collected at intervals of 10 cm for each profile using a soil corer (a stainless steel cylinder with a volume of 100 cm3). Soil samples were collected at the same distance intervals and used to analyse the nitrate and ammonium contents, pH and the dual stable isotopes of nitrate δ15N and δ18O.
All of the collected soil samples were air-dried in the laboratory; gravel and roots were carefully removed from the soil. The air-dried soil samples were ground in an agate mortar and passed through a 0.15 mm sieve. Soil total N contents were measured through micro-Kjeldahl digestion, followed by distillation and titration53. Moreover, SOC was determined using soil samples digested in K2Cr2O7-H2SO4 solution using a heated oil bath, and the organic carbon concentration was subsequently determined via titration53. Soil nitrate and ammonium were extracted with a 2 M KCl solution (soil:solution, 1:5) and filtered through a 0.45 μm filter13. A portion of each solution was prepared to determine the nitrate and ammonium concentrations, while the other portion was prepared to determine the nitrate δ15N and δ18O levels. The nitrate and ammonium concentrations were analysed using a continuous flow analyser (Skalar San++ System, Skalar Analytical B.V., Netherlands). The soil samples used for the bulk density analysis were dried at 115 °C for 24 h. STN storage (Mg ha−1) values were calculated as follows:

where Di, BDi and TNi represent the soil thickness (cm), bulk density (g cm−3) and soil total N content (g kg−1), respectively, for the ith level of the soil profile.
Foliar δ15N and nitrate δ15N and δ18O analyses
The fresh foliar litterfall was sampled in the forestland and grassland in September 5–14, 2013 and returned to the laboratory. Dust and soil were carefully removed from the surfaces of the foliar samples. The samples were air-dried and ground to a powder. The foliar N content and δ15N were analysed using a Vario PYRO cube element analyser and an EA-IsoPrime100 stable isotope ratio mass spectrometer (Isoprime Ltd, U.K). The soil nitrate δ15N and δ18O levels were prepared via quantitative bacterial reduction of nitrate to nitrous oxide. Nitrous oxide was extracted and purified using a trace gas pre-concentrator unit; the product was analysed using an EA-IsoPrime100 stable isotope ratio mass spectrometer (Yue et al., 2014). Three international materials (USGS-32, USGS-34 and USGS-35) were used to calibrate the measured sample data. Each sample was measured in duplicate, and the standard error was 0.3% for nitrate δ15N and 0.4% for nitrate δ18O.
The isotope ratios (δ15N and δ18O) are expressed in δ notation as parts per thousand deviations (‰):

where R = 15 N/14N or 18O/16O. The 15N/14N reference is N2 in air, whereas the 18O/16O reference is Vienna standard mean ocean water (VSMOW).
Statistical analysis
An independent-sample t-test was performed to test the significance of the soil property differences, soil N storage and availability at an alpha level of 0.05 (a = 0.05) between the forestland and grassland. All statistical analyses were performed with the Statistical Program for Social Sciences (SPSS 11.0, SPSS Inc., 2001).
Additional Information
How to cite this article: Jin, Z. et al. Comparing watershed black locust afforestation and natural revegetation impacts on soil nitrogen on the Loess Plateau of China. Sci. Rep. 6, 25048; doi: 10.1038/srep25048 (2016).
References
Cao, S., Wang, G. & Chen, L. Questionable value of planting thirsty trees in dry regions. Nature 465, 31 (2010).
Cao, S. et al. Excessive reliance on afforestation in China’s arid and semi-arid regions: lessons in ecological restoration. Earth Sci. Rev. 104, 240–245 (2011).
Gao, Y. et al. Water use efficiency threshold for terrestrial ecosystem carbon sequestration under afforestation in China. Agric. For. Meteorol. 195–196, 32–37 (2014).
Farley, K. A., Jobbágy, E. G. & Jackson, R. B. Effects of afforestation on water yield: a global synthesis with implications for policy. Glob. Change Biol 11, 1565–1576 (2005).
Cao, S., Chen, L. & Yu, X. Impact of China’s Grain for Green Project on the landscape of vulnerable arid and semi-arid agricultural regions: a case study in northern Shaanxi Province. J. Appl. Ecol. 46, 536–543 (2009).
Jiao, J., Zhang, Z., Bai, W., Jia, Y. & Wang, N. Assessing the ecological success of restoration by afforestation on the Chinese Loess Plateau. Restor. Ecol. 20, 240–249 (2012).
van Groenigen, K. J. et al. Element interactions limit soil carbon storage. Proc. Natl Acad. Sci. 103, 6571–6574 (2006).
LeBauer, D. S. & Treseder, K. K. Nitrogen limitation of net primary productivity in terrestrial ecosystems is globally distributed. Ecology 89, 371–379 (2008).
Binkley, D. & Resh, S. C. Rapid changes in soils following eucalyptus afforestation in Hawaii. Soil Sci. Soc. Am. J. 63, 222–225 (1999).
Berthrong, S. T. Ph. D. Thesis: the effect of afforestation on soil microbes and biogeochemistry across multiple scales (Duke University, Durham, North Carolina, USA, 2009).
Richter, D. D. et al. Legacies of agriculture and forest regrowth in the nitrogen of old-field soils. Forest Ecol. Manage. 138, 233–248 (2000).
McLauchlan, K. K. et al. Changes in nitrogen cycling during the past century in a northern hardwood forest. Proc. Natl Acad. Sci. 104, 7466–7470 (2007).
Li, M., Zhou, X., Zhang, Q. & Cheng, X. Consequences of afforestation for soil nitrogen dynamics in central China. Agr. Ecosyst. Environ. 183, 40–46 (2014).
Krueger-Mangold, J. et al. Identification of the limiting resource within a semi-arid plant association. J. Arid Environ. 58, 309–320 (2004).
Yahdjian, L., Gherardi, L. & Sala, O. E. Nitrogen limitation in arid-subhumid ecosystems: a meta-analysis of fertilization studies. J. Arid Environ. 75, 675–680 (2011).
Lamb, D., Erskine, P. D. & Parrotta, J. A. Restoration of degraded tropical forest landscapes. Science 310, 1628–1632 (2005).
Cramera, V. A., Hobbs, R. J. & Standisha, R. J. What’s new about old fields? Land abandonment and ecosystem assembly. Trends Ecol. Evol. 23, 104–112 (2008).
Zhang, K., Cheng, X., Dang, H., Ye, C. & Zhang, Q. Soil nitrogen and denitrification potential as affected by land use and stand age following agricultural abandonment in a headwater catchment. Soil Use Manage. 28, 361–369 (2012).
Zhang, K., Dang, H., Tan, S., Wang, Z. & Zhang, Q. Vegetation community and soil characteristics of abandoned agricultural land and pine plantation in the Qinling Mountains, China. Forest Ecol. Manage. 259, 2036–2047 (2010).
Food and Agriculture Organization of the United Nations (FAO). World Soil Resources Reports 102: Carbon Sequestration in Dryland Soils (Food and Agriculture Organization, Rome, 2004).
Davidson, E. A. et al. Recuperation of nitrogen cycling in Amazonian forests following agricultural abandonment. Nature 447, 995–998 (2007).
Norby, R. J., Warren, J. M., Iversen, C. M., Medlyn, B. E. & McMurtrie, R. E. CO2 enhancement of forest productivity constrained by limited nitrogen availability. Proc. Natl Acad. Sci. 107, 19368–19373 (2010).
Garten, C. T., Iversen, C. M. & Norby, R. J. Litterfall 15N abundance indicates declining soil nitrogen availability in a free-air CO2 enrichment experiment. Ecology 92, 133–139 (2011).
West, J. B., Bowen, G. J., Cerling, T. E. & Ehleringer, J. R. Stable isotopes as one of nature’s ecological recorders. Trends Ecol. Evol. 21, 408–414 (2006).
Houlton, B. Z. & Bai, E. Imprint of denitrifying bacteria on the global terrestrial biosphere. Proc. Natl Acad. Sci. 106, 21713–21716 (2009).
McLauchlan, K. K., Williams, J. J., Craine, J. M. & Jeffers, E. S. Changes in global nitrogen cycling during the Holocene epoch. Nature 495, 352–355 (2013).
Högberg, P. 15N natural abundance in soil-plant systems. New Phytol. 137, 179–203 (1997).
Pardo, L. H., Kendall, C., Pett-Ridge, J. & Chang, C. C. Y. Evaluating the source of streamwater nitrate using δ15N and δ18O in nitrate in two watersheds in New Hampshire, USA. Hydrol. Process. 18, 2699–2712 (2004).
Liu, C., Li, S., Lang, Y. & Xiao, H. Using δ15N and δ18O-values to identify nitrate sources in karst ground water, Guiyang, Southwest China. Environ. Sci. Technol. 40, 6928–6933 (2006).
Wexler, S. K., Hiscock, K. M. & Dennis, P. F. Catchment-scale quantification of hyporheic denitrification using an isotopic and solute flux approach. Environ. Sci. Technol. 45, 3967–3973 (2011).
Yue, F., Li, S., Liu, C., Lang, Y. & Ding, H. Sources and transport of nitrate constrained by the isotopic technique in a karst catchment: an example from Southwest China. Hydrol. Process. 29, 1883–1893 (2015).
Kendall, C., Elliott, E. M. & Wankel, S. D. Tracing anthropogenic inputs of nitrogen to ecosystems In Stable Isotopes in Ecology and Environmental Science 2nd Edn (ed. Lajtha, K. & Michener, R. ) 375–449 (Blackwell Publishing House, 2007).
Chen, L. et al. Effect of land use conversion on soil organic carbon sequestration in the loess hilly area, loess plateau of China. Ecol. Res. 22, 641–648 (2007).
Zhou, P., Wen, A., Zhang, X. & He, X. Soil conservation and sustainable eco-environment in the Loess Plateau of China. Environ. Earth Sci. 68, 633–639 (2013).
Lü, Y. H. et al. A policy-driven large scale ecological restoration: quantifying ecosystem services changes in the Loess Plateau of China. Plos ONE 7, e31782 (2012). doi: 10.1371/journal.pone.0031782.
Wang, Y., Shao, M., Zhu, Y. & Liu, Z. Impacts of land use and plant characteristics on dried soil layers in different climatic regions on the Loess Plateau of China. Agric. For. Meteorol. 151, 437–448 (2011).
Jin, Z. et al. Natural vegetation restoration is more beneficial to soil surface organic and inorganic carbon sequestration than tree plantation on the Loess Plateau of China. Sci. Total Environ. 485–486, 615–623 (2014).
Davidson, E. A., Keller, M., Erickson, H. E., Verchot, L. V. & Veldkamp, E. Testing a conceptual model of soil emissions of nitrous and nitric oxides. BioScience 50, 667–680 (2000).
Davidson, E. A. & Martinelli, L. A. Nutrient limitations to secondary forest regrowth in Amazonia and Global Change (ed. Keller, M., Bustamante, M., Gash, J., Dias, S. P. ) 299–310 (American Geophysical Union, Washington, DC, 2009).
Wei, X., Shao, M., Fu, X., Ågren G. I. & Yin, X. The effects of land use on soil N mineralization during the growing season on the northern Loess Plateau of China. Geoderma 160, 590–598 (2011).
Liu, J. H. Ph.D. Thesis: Study on growth traits of Robinia Pseudoacacia plantation and its naturalization process on the Loess Plateau (Graduate University of Chinese Academy of Sciences, Beijing, China, 2008).
Qiu, L., Zhang, X., Cheng, J. & Yin, X. Effects of black locust (Robinia pseudoacacia) on soil properties in the loessial gully region of the Loess Plateau, China. Plant Soil 332, 207–217 (2010).
Danso, S. K. A., Zapata, F. & Awonaike, K. O. Measurement of biological N2 fixation in field-grown Robinia pseudoacacia L. Soil Biol. Biochem. 27, 415–419 (1995).
Boring, L. R. & Swank, W. T. Symbiotic nitrogen fixation in regenerating black locust (Robinia pseudoacacia L.) stands. Forest Sci. 30, 528–537 (1984).
Lopez, C. M. L., Mizota, C., Nobori, Y., Sasaki, T. & Yamanaka, T. Temporal changes in nitrogen acquisition of Japanese black pine (Pinus thunbergii) associated with black locust (Robinia pseudoacacia). J. Forest. Res. 25, 585–589 (2014).
Huang, M. B., Kang, S. Z. & Li, Y. S. A comparison of hydrological behaviors of forest and grassland watersheds in gully region of the Loess Plateau. J. Nat. Resour. 14, 226–231 (1999).
Wang, H. S., Huang, M. B. & Zhang, L. Impacts of re-vegetation on water cycle in a small watershed of the Loess Plateau. J. Nat. Resour. 19, 344–350 (2004).
Bian, Y., Lyv, H. & Zhao, P. Hydrological process responses to land use change in Nanxiaohegou. Yellow River 37(9), 88–91 (2015).
Liu, B., Chang, W. & Xu X. Effects of soil and water conservation measures on runoff and sediment load in the Yangjiagou watershed. Yellow River 30(11), 77–81 (2008).
Kendall, C. Tracing nitrogen sources and cycling in catchments in Isotope Tracers In Catchment Hydrology (ed. Kendall, C. & McDonnell, J. J. ) 519–576 (Elsevier, Amsterdam,1998).
Elliott, E. M. et al. Nitrate isotopes in precipitation to distinguish NOx sources, atmospheric processes, and source areas in the United States. Eos (Transactions American Geophysical Union) 87, 36 (2006).
Li, M. Master Thesis: the Influence of vegetation change on hydrologic factors in Nanxiaohe river basin (Xi’an University of Technology, Xi’an, China, 2006).
Bao, S. T. Chemical Analysis for Agricultural Soil (China Agriculture Press, Beijing, 1999).
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (41573067, 41301100), the Key Research Program of the Chinese Academy of Sciences (Grant KZZD-EW-04) and the Youth Innovation Promotion Association CAS (2014370).
Author information
Authors and Affiliations
Contributions
Z.J., K.W. and Y.W. conducted the field soil sampling; Z.J. conducted the data analysis. B.C. prepared the map of the study area. The manuscript was written and commented by Z.J., Y.W. and X.L. The project was planned by Z.J.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Jin, Z., Li, X., Wang, Y. et al. Comparing watershed black locust afforestation and natural revegetation impacts on soil nitrogen on the Loess Plateau of China. Sci Rep 6, 25048 (2016). https://doi.org/10.1038/srep25048
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep25048
This article is cited by
-
Soil erosion differences in paired grassland and forestland catchments on the Chinese Loess Plateau
Journal of Mountain Science (2023)
-
Changes in diversity, composition and assembly processes of soil microbial communities during Robinia pseudoacacia L. restoration on the Loess Plateau, China
Journal of Arid Land (2022)
-
A global meta-analysis shows soil nitrogen pool increases after revegetation of riparian zones
Journal of Soils and Sediments (2021)
-
Spatial variation and soil nitrogen potential hotspots in a mixed land cover catchment on the Chinese Loess Plateau
Journal of Mountain Science (2019)
-
Comparing watershed afforestation and natural revegetation impacts on soil moisture in the semiarid Loess Plateau of China
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.