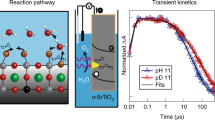
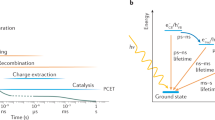
Abstract
THE photoelectrolysis of water using n-type semiconductors such as TiO2, (refs 1–4) SrTiO3, (refs 5–7) and so on is of current interest and importance for the utilisation of solar energy to produce hydrogen gas for fuel. In steady state conditions under illumination and in the absence of H2 and O2 in the electrolyte, the rate of H2 evolution on a cathode is proportional to that of O2 evolution on a semiconductor anode. The O2 evolution process is considered to proceed by electron tunnelling from electrolyte levels to holes in the semiconductor through the Helmholtz layer, which must take place between states of equal energy. If only isoenergetic electron transfer between holes and occupied electrolyte states overlapping in energy the valence band occurs, the hole currents for O2 evolution at n-TiO2 would be expected to be very low, because the equilibrium redox level of the H2O/O2 couple is considerably above (at 1.8eV for pH = 4.7 (ref. 2)) the top of the valence band. Experimentally, however, the current of holes at the TiO2-electrolyte interface is found to be quite high and the efficiency in charge transfer of holes at the interface is almost 100%8. To account for such a high efficiency, a contribution from some surface states has been proposed8,9. A possible importance of surface states in electrochemistry at TiO2 electrodes has been described by Frank and Bard10. Also, a recent electroluminescence measurement made for n-TiO2 indicates that occupied surface states exist near the midgap at the TiO2–electrolyte interface11. At present, however, it is not understood which kind or character of surface states can improve the charge transfer efficiency for holes at the interface. We present here a theoretical treatment of electron tunnelling between the semiconductor and electrolyte by surface states. We apply a theory of surface states (more exactly interface states) in tunnel MOS (metal–oxide–semiconductor) devices of Freeman and Dahlke12 to the semiconductor–electrolyte interface. A quite high efficiency for holes at the interface can be obtained when the electron population of the surface states is controlled by the electrolyte and the surface states are almost completely occupied.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fujishima, A. & Honda, K. Nature 238, 37–38 (1972).
Ohnishi, T., Nakato, Y. & Tsubomura, H. Ber. Bunsenges. phys. Chem. 79, 523–525 (1975).
Nozik, A. J. Nature, 257, 383–386 (1975).
Desplat, J. L. J. appl. Phys. 47, 5102–5104 (1976).
Mavroides, J. G., Kafalas, J. A. & Kolesar, D. F. Appl. Phys. Lett. 28, 241–243 (1976).
Wrighton, M. S. et al. J. Am. chem. Soc. 98, 2774–2779 (1976).
Watanabe, T., Fujishima, A. & Honda, K. Bull. chem. Soc. Jap. 49, 355–358 (1976).
Mavroides, J. G. & Kolesar, D. F. J. Vac. Sci. Technol., 15, 538–541 (1978).
Bockris, J. O'M. & Uosaki, K. Int. J. Hydrogen Energy 2, 123–138 (1977).
Frank, S. N. & Bard, A. J. J. Am. chem. Soc. 97, 7427–7433 (1975).
Noufi, R. N., Kohl, P. A., Frank, S. N. & Bard, A. J. J. electrochem. Soc. 125, 246–252 (1978).
Freeman, L. B. & Dahlke, W. E. Solid-St. Electron. 13, 1483–1503 (1970).
Henrich, V. E., Dresselhaus, G. & Zeiger, H. J. Phys. Rev. Lett. 36, 1335–1339 (1976); Solid State Commun. 24, 623–626 (1977); J. Vac. Sci. Technol. 15, 534–537 (1978).
Lo, W. J., Chung, Y. W. & Somorjai, G. A. Surf. Sci. 71, 199–219 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
NISHIDA, M. Charge transfer by surface states in the photoelectrolysis of water using a semiconductor electrode. Nature 277, 202–203 (1979). https://doi.org/10.1038/277202a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/277202a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.