Abstract
CD8+ T cells eliminate intracellular infections through two contact-dependent effector functions: cytolysis and secretion of antiviral cytokines. Here we identify the following additional function for memory CD8+ T cells that persist at front-line sites of microbial exposure: to serve as local sensors of previously encountered antigens that precipitate innate-like alarm signals and draw circulating memory CD8+ T cells into the tissue. When memory CD8+ T cells residing in the female mouse reproductive tract encountered cognate antigen, they expressed interferon-γ (IFN-γ), potentiated robust local expression of inflammatory chemokines and induced rapid recruitment of circulating memory CD8+ T cells. Anamnestic responses in front-line tissues are thus an integrated collaboration between front-line and circulating populations of memory CD8+ T cells, and vaccines should establish both populations to maximize rapid responses.
Similar content being viewed by others
Main
Most infections are initiated at vulnerable body surfaces, such as the mucosae. CD8+ T cells, which are endowed with potent effector functions, are typically required for the elimination of viral infections. These effector functions (cytotoxic elimination of infected host cells and the secretion of cytokines that interfere with viral replication or promote inflammation) operate locally and require direct interaction with cells bearing viral antigen1,2,3. Thus, CD8+ T cells must be present in sufficient quantity at sites of infection to examine each host cell for the presence of viral antigens. The critical challenge is that before a primary infection, pathogen-specific CD8+ T cells are exceedingly rare and patrol only the secondary lymphoid organs (SLOs)2,4. Thus, in naive hosts, the nonspecific innate immune system, which is constitutively distributed throughout the host, must hold pathogen replication in check until more-potent CD8+ T cells can proliferate and migrate to infected tissues5. This temporal lag in CD8+ T cell responses provides a critical window of opportunity for pathogen replication.
In the event that the pathogen is cleared, hosts retain expanded populations of pathogen-specific memory CD8+ T cells that patrol SLOs6,7 and discrete populations that patrol nonlymphoid tissues2. The memory T cells that patrol SLOs (central memory T cells) routinely recirculate among the blood, lymph and various SLOs1,2. Nonlymphoid memory T cells (effector memory T cells) constitutively recirculate among blood, lymph and nonlymphoid tissues1,2. That model has now been amended. Memory T cells do not undergo demonstrable recirculation through many nonlymphoid anatomical compartments, including the epithelium of the small intestine, skin epidermis, lungs, salivary gland and central nervous system9,10,11,12,13,14,16,17,18. Instead, T cells are able to migrate into many nonlymphoid compartments for only a brief time after antigen stimulation and then differentiate into organ-specific resident non-recirculating memory T cells in situ11,12,15,18. These sessile front-line populations have been called 'resident memory T cells'.
Present models suggest that memory T cells positioned in nonlymphoid tissues or SLOs make anatomically and temporally discrete contributions to secondary responses, becoming functionally active only when their respective tissues encounter cognate antigen. CD8+ T cells that populate SLOs recognize antigen draining from the site of infection before proliferating, differentiating into effector cells that respond to inflammatory chemokines and migrating to the front-line infected tissue. The contribution of this reserve force of the response, although potent, is delayed by several days. In contrast, resident memory CD8+ T cells that are positioned at sites of infection at the time of a pathogen-reexposure event are anatomically poised for immediate recognition and response14,15,17,19,20,21. Their role in protective immunity has been likened to that of an occupying force that operates at front-line sites of infection independently of recirculating memory T cells21. It has been demonstrated that mice that have recirculating, but not resident, memory CD8+ T cell populations (rather than having both populations) show substantially delayed control of viral rechallenges at front-line tissues9,13,22,23.
Here we define an additional function for resident memory CD8+ T cells, as sentinels of pathogen-derived antigens. Local recognition by the adaptive immune system was far more potent and sensitive than that of the innate immune system in inducing local chemokine expression. Notably, when resident memory T cells in the female mouse reproductive tract recognized cognate antigen in situ, they precipitated rapid and abundant recruitment of unstimulated memory T cells to this tissue, which is not under routine immunosurveillance by recirculating memory T cells. Thus, resident memory T cells are more than just an occupying force, as they orchestrate migration and thus serve to recruit reinforcements to fight infections.
Results
Rapid local CD8+ T cell recall response
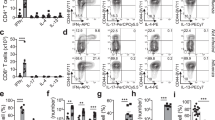
To visualize virus-specific CD8+ T cells in the female mouse reproductive tract, we transferred Thy-1.1+ naive P14 CD8+ T cells (which recognize the gp33 epitope of lymphocytic choriomeningitis virus (LCMV)) into naive C57BL/6J mice. We then infected those recipients intraperitoneally with 2 × 105 plaque-forming units of LCMV, Armstrong strain (to generate 'P14 immune chimeras'), and identified LCMV-specific memory CD8+ T cells 2 months later by staining for Thy-1.1. We determined the ratio of P14 CD8+ T cells to nucleated cells by computerized image analysis of sections stained for Thy-1.1 and with the DNA-intercalating dye DAPI (Supplementary Fig. 1). To assess the dynamics of the CD8+ T cell recall response after local reexposure to antigen, we challenged P14 immune chimeras transcervically with a recombinant vaccinia virus expressing gp33 (VV-gp33) by depositing the virus directly into the cervical canal and uterine lumen23,24. Local recall infection elicited a rapid increase in the number of P14 T cells in the reproductive tract within 2 d (Fig. 1a and Supplementary Fig. 1). Infection with vaccinia virus expressing the irrelevant antigen chicken ovalbumin (VV-OVA) elicited little accumulation of memory P14 T cells in the tissue, which indicated that recognition of cognate antigen by memory CD8+ T cells was required. To determine if infection-induced inflammation was necessary for the accumulation of antigen-specific T cells in the female reproductive tract, we injected cognate gp33 peptide transcervically into mice. We found that gp33 peptide itself, without infection, was sufficient to recapitulate the greater abundance of P14 T cells in the tissue noted after local rechallenge with VV-gp33 (Fig. 1). Control SIINFEKL peptide (derived from chicken OVA), which is not recognized by P14 T cells, had little effect. These data showed that recognition of cognate antigen by memory CD8+ T cells precipitated the rapid accumulation of memory CD8+ T cells in the reproductive tract. We recapitulated this response without the use of transgenic T cells (Supplementary Fig. 2).
(a) P14 cells in the female reproductive tract of mice (n = 6 per group) with widely disseminated Thy-1.1+ memory P14 CD8+ T cells established after infection LCMV, assessed 48 h after no treatment (Memory) or transcervical challenge with 4 × 105 plaque-forming units of VV-gp33 or VV-OVA or 50 μg gp33 or SIINFEKL peptide; results are presented relative to total nucleated cells. NS, not significant; *P < 0.05 and **P < 0.001 (unpaired two-tailed t-test). (b) Microscopy of tissue from the female reproductive tract of an untreated P14 immune chimera (Memory) or a P14 immune chimera challenged with gp33, stained for Thy-1.1 (red; P14) and with DAPI (blue). Scale bars, 100 μm. Data are from two experiments with a total of six mice (pooled), representative of four independent experiments with similar results (a; mean and s.e.m.), or are from one experiment representative of four experiments with three mice per group in each (12 mice total per group; b).
Antigen recognition by T cells induces chemokine expression
Chemokines are necessary for the extravasation of T cells from blood to tissues24. For example, the chemokine CXCL9 induces the migration of effector T cells to the female reproductive tract18. To determine if the reactivation of memory CD8+ T cells induced chemokine expression in the female reproductive tract, we rechallenged P14 immune chimeras transcervically with VV-gp33. Within 12 h, VV-gp33 induced 5.5-fold higher CXCL9 expression on the CD31+ vasculature (Fig. 2a and Supplementary Fig. 3). In contrast, transcervical challenge with VV-OVA induced little induction of CXCL9 expression (Fig. 2), which demonstrated that the recognition of cognate antigen by T cells potentiated almost immediate local chemokine expression. Antigen recognition without the context of infection was sufficient to drive this chemokine gradient, as transcervical inoculation of gp33 peptide recapitulated the CXCL9 expression on CD31+ cells (Fig. 2a,b). Further analysis showed that reactivation of memory T cells induced a general inflammatory chemokine response. For example, CCL2 and CXCL9 were expressed by tissue CD11c+MHCII+ dendritic cells, CCL3 and CCL4 were expressed by memory P14 CD8+ T cells, and CXCL10 and CX3CL1 were present on CD31+ vasculature within 12 h (Fig. 2c and Supplementary Fig. 4). These data demonstrated that the recognition of cognate antigen by memory CD8+ T cells rapidly induced tissue chemokine expression, at much higher concentrations at this early time point than those achieved by innate recognition of infection with vaccinia virus.
(a) Mean fluorescence intensity (MFI) of CXCL9 expression on CD31+ vessels in mice with widely disseminated Thy-1.1+ memory P14 CD8+ T cells established after infection with LCMV, assessed 12 h after no treatment or transcervical challenge with 4 × 105 plaque-forming units of VV-gp33 or VV-OVA or 50 μg gp33 or SIINFEKL peptide. (b) Microscopy of tissue from the female reproductive tract of an untreated P14 immune chimera (Memory) or a P14 immune chimera mouse challenged with gp33 as in a, stained for CXCL9 (red) and CD31 (blue). Scale bars, 100 μm. (c) Frequency of CCL2+ cells (left) or CXCL9+ cells (middle) among CD11c+MHCII+ cells and of CCL3+ and/or CCL4+ cells among memory P14 CD8+ T cells (right) from mice as in a, 12 h after no treatment or transcervical challenge with gp33, assessed by flow cytometry. *P < 0.05 and **P <0.001 (unpaired two-tailed t-test). Data are from two experiments with a total of six mice per group (pooled), representative of nine experiments (a,c; mean and s.e.m.), or one experiment representative of three experiments with four mice per group each (12 mice total per group; b).
Reactivated memory T cells recruit resting memory T cells
We next investigated whether the accumulation of memory CD8+ T cells was dependent on the in situ proliferation of local memory CD8+ T cells or, alternatively, on the migration of either antigen-reactivated, canonical effector CD8+ T cells or resting memory CD8+ T cells from outside the tissue. To assess this, we transferred naive OT-I CD45.1+ CD8+ T cells (specific for the SIINFEKL epitope of OVA) into P14 immune chimeras, which we then infected with recombinant vesicular stomatitis virus expressing ovalbumin (VSV-OVA). This experimental design permitted the visualization of two memory CD8+ T cell populations with distinct specificities: Thy-1.1+ gp33-specific P14 cells and CD45.1+ SIINFEKL-specific OT-I cells. We either left those mice untreated or challenged them transcervically with gp33 peptide to reactivate the P14 memory CD8+ T cell population (Fig. 3a), then counted OT-I CD8+ T cells 2 d later. Transcervical administration of gp33 peptide resulted in 450% more memory OT-I cells in the female reproductive tract, which was coupled with 58% fewer memory OT-I cells in the spleen and 81% fewer memory OT-I cells in blood (Fig. 3b). Notably, the number of memory OT-I CD8+ T cells did not change in draining and nondraining lymph nodes. These data demonstrated that bystander memory CD8+ T cells of irrelevant specificities accumulated in the female reproductive tract in response to reactivation of memory CD8+ T cells, which indicated that local amassment of CD8+ T cells was not dependent on in situ proliferation or antigen-mediated effector differentiation but instead was inversely correlated with the dispersion of resting memory CD8+ T cells from spleen and blood.
(a) Protocol for the establishment of two populations of memory CD8+ T cells with different specificities: Thy-1.1+ P14 (gp33-specific) cells and CD45.1+ OT-I (SIINFEKL-specific) cells. t.c., transcervical. (b) OT-I CD8+ T cells in the female reproductive tract (FRT), spleen, blood, draining lymph nodes (DLN) and nondraining lymph nodes (NDLN) of mice generated as in a, assessed 48 h after no challenge (Memory) or challenge with gp33. *P < 0.05 and **P < 0.001 (unpaired two-tailed t-test). Data are pooled from two experiments with six mice per tissue (all tissues except blood) or five mice (blood; mean and s.e.m.).
Local memory CD8+ T cells orchestrate recruitment
We next sought to determine whether antigen-dependent reactivation of memory CD8+ T cells in prototypical inductive sites of immune responses, such as lymph nodes and spleen, was needed to precipitate the migration of resting memory CD8+ T cells to the female reproductive tract. For this, we developed a model with which we could remove memory P14 cells from SLOs but preserve them in the female reproductive tract by injecting P14 immune chimeras with 3 μg of complement-fixing Thy-1.1-specific antibody (Fig. 4a). We then injected the depleted mice intravenously with memory OT-I CD8+ T splenocytes and challenged them with gp33 transcervically, then counted OT-I CD8+ T cells 2 d later. This showed that front-line memory CD8+ T cells, in the absence of SLO and blood memory, were able to recruit memory CD8+ T cells from outside the tissue (Fig. 4b). However, naive OT-I CD8+ T cells were excluded from the female reproductive tract, which indicated that only antigen-experienced T cells migrated to local sites of T cell reactivation (Fig. 4b).
(a) P14 CD8+ T cells in the spleen, lymph nodes (LN) and female reproductive tract of P14 immune chimeras 72 h after no treatment (Memory) or depletion of Thy-1.1+ memory P14 cells from recirculating compartments (with preservation in the female reproductive tract) through administration of complement-fixing antibody to Thy-1.1 (+ α-Thy-1.1). (b) Quantification of transferred OT-I cells in naive mice (N), P14 immune chimeras (P14) and P14 immune chimeras depleted of circulating P14 cells as in a (P14 (dep)), each given intravenous transfer of no cells (−) or memory (Mem OT-I) or naive (Naive OT-I) OT-I CD8+ T cells (+), then, 24 h later, challenged transcervically with gp33, assessed 48 h later in coronal sections (7 μm in thickness) of the entire female reproductive tract. *P < 0.01 and **P < 0.001 (unpaired two-tailed t-test). Data are representative of three independent experiments with three mice per group in each (a; mean and s.e.m.) or three (Mem OT-I) or two (Naive OT-I) independent experiments with three mice per group in each (b; mean and s.e.m.).
Recruitment requires resident memory T cell–derived IFN-γ
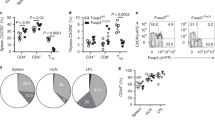
It remained unclear how local memory CD8+ T cells communicated an antigen-recognition event that precipitated the recruitment of peripheral T cells. IFN-γ is a secreted cytokine that signals 'danger' or microbial infection to the host. Antigen-specific memory T cells in the female reproductive tract rapidly expressed IFN-γ in vivo after local antigen rechallenge (Fig. 5a). To determine whether IFN-γ expression was important for inducing rapid recruitment of CD8+ T cells to the female reproductive tract, we generated C57BL/6 mice that had either wild-type or IFN-γ-deficient memory OT-I CD8+ T cells after immunization with VV-OVA. We then challenged the mice transcervically with SIINFEKL peptide to reactivate memory OT-I cells. Quantification of CD8β+ T cells and CXCL9 expression showed that the alarm function of sensitized memory CD8+ T cells was dependent on their ability to express IFN-γ (Fig. 5b and Supplementary Fig. 5).
(a) Immunofluorescence microscopy of P14 cells and IFN-γ in P14 immune chimeras 12 h after no treatment (Memory) or transcervical challenge with gp33 or SIINFEKL. Scale bars, 20 μm. (b) CD8β+ T cells in the female reproductive tracts of IFN-γ-sufficient (Ifng+/+) and IFN-γ-deficient (Ifng−/−) 'OT-I immune chimeras' (C57BL/6 mice with OT-I memory CD8+ T cells after immunization with VV-OVA) at 48 h after transcervical challenge with SIINFEKL. (c) Quantification of memory P14 cells in the spleen, lymph nodes and female reproductive tract (right) of conjoined parabionts consisting of naive recipients of intravenous (i.v.) transfer of OT-I CD45.1+ memory cells (Naive) conjoined via parabiosis (far left) to Thy-1.1+ P14 immune chimeras (Memory), assessed 14–16 d after surgery. (d) OT-I CD8+ T cells in coronal sections (7 μm in thickness) through the entire female reproductive tract of parabionts as in c, 48 h after no challenge (Naive or Memory) or transcervical challenge with gp33 (Naive + gp33 or Memory + gp33). *P < 0.001 (unpaired two-tailed t-test). Data are pooled from two independent experiments with five to six mice per group (mean and s.e.m. in b–d).
In short-term homing assays, effector CD8+ T cells migrate to the intestinal epithelium, salivary glands or female reproductive tract, but memory CD8+ T cells do not11,12,18. Such data support a model in which effector T cells seed mucosal tissues shortly after antigen stimulation, then differentiate into resident memory T cells that persist in situ without recirculating9. To further investigate that hypothesis, we conjoined P14 immune chimeras by parabiosis surgery with naive C57BL/6J mice that had received intravenous transfer of OT-I memory CD8+ T cells. We found that, 14–16 d later, P14 memory CD8+ T cells had equilibrated between the SLOs, but not the female reproductive tracts, of both parabionts (Fig. 5c), which indicated that memory T cells in the female reproductive tract were not recirculating during this interval. When we challenged both parabionts with gp33 transcervically, we found that rapid accumulation of OT-I cells occurred only in mice that had P14 cells in the female reproductive tract (Fig. 5d). These data demonstrated that resident memory T cells at the site of antigen exposure were required for inciting rapid local amassment of unstimulated memory T cells and thus defined an additional function for cell-mediated immunity positioned at initial sites of infection.
Discussion
After primary infection, the innate immune system responds to microbe-derived 'danger' signals, which results in the expression of inflammatory chemokines and recruitment of leukocytes to the affected tissue5,26. Although this response is often deemed 'immediate' because the innate immune system is pre-positioned in tissues, clearance of most pathogens requires the specificity and more potent effector mechanisms of the adaptive immune system. However, primary adaptive responses are slow to develop because rare pathogen-specific lymphocytes must proliferate and migrate from SLOs to sites of infection, which provides a temporal window of opportunity for continued pathogen replication and disease.
Our study has defined an additional function for nonlymphoid tissue–resident memory CD8+ T cells. We found that these cells use the highly sensitive lymphocyte antigen receptor to precipitate much faster and more-robust tissue chemokine expression than that accomplished by the innate immune system, and they were able to initiate the rapid recruitment of preformed memory CD8+ T cells to sites of antigen recognition. This feature of the adaptive immune response allows numerical amassment of antigen-experienced T cells to sites of reinfection before the protracted process of SLO-dependent reactivation and subsequent migration.
Our results do not exclude the possibility of other roles for front-line memory CD8+ T cells during anamnestic responses. Published reports indicate that resident memory CD8+ T cells can initiate proliferation in situ in response to local antigen stimulation27,28. It has also been shown that reactivation of CD4+ T cells in the lungs potentiates IFN-γ-independent, inflammatory, innate-like cytokine production29. Perhaps this process could facilitate the recruitment of various leukocyte subsets, including monocytes, neutrophils and natural killer cells. A published study has shown that topical vaginal application of CXCL9 and CXCL10 enhances the migration of effector T cells to the female reproductive tract but does not recruit resting memory T cells18,23. We found that the reactivation of local resident memory, which was established during a prior effector response, was sufficient to drive the recruitment of unstimulated memory T cells from outside the tissue. Thus, the exclusion of memory T cells from recirculation through restricted nonlymphoid compartments is context dependent and can be overcome when local memory T cells 'sound the alarm' in response to antigen recognition in the tissue. This process provides a means for recruiting pathogen-experienced T cells to sites of reinfection more rapidly, by bypassing the delay inherent in waiting for the dissemination of antigen from infected sites, followed by recognition by the T cell antigen receptor and effector differentiation of the cells. These observations highlight the importance of establishing memory CD8+ T cells at front-line sites of pathogen exposure to provide sensitive detection of antigens previously recognized in a pathogenic context. However, in addition, optimal rapid responses in tissues will require memory CD8+ T cell populations that can be recruited from outside the affected tissue.
Methods
Mice and infection.
All mice were used in accordance with the guidelines of the Institutional Animal Care and Use Committees at the University of Minnesota. C57BL/6J mice were from The Jackson Laboratory. Thy-1.1+ P14, CD45.1+ OT-I and CD45.1+ mice were fully backcrossed to C57BL/6J mice and maintained in our animal colony. OT-I IFN-γ-deficient mice (provided by M. Mescher) were generated as follows: IFN-γ-deficient B6.129S7-Ifngtm1Ts/J mice (The Jackson Laboratory) were bred with OT-I mice. P14 immune chimeras were generated by transfer of 5 × 104 naive Thy-1.1+ P14 T cells into naive C57BL/6J mice and infection of the recipient mice with 2 × 105 plaque-forming units (PFU) of LCMV (Armstrong strain) the next day. Memory OT-I cells were generated by transfer of 5 × 104 naive CD45.1+ OT-I T cells into naive C57BL/6J mice or into memory P14 chimeras. The next day, recipient mice were infected intravenously with either 1 × 106 PFU VSV-OVA or 2 × 106 PFU VV-OVA For local rechallenge experiments, 50 μg of the appropriate peptide or 4 × 105 PFU VV-gp33 or VV-OVA was delivered transcervically as described23 in a volume of 35 μl delivered by modified gel-loading pipet. For depletion of circulating memory P14 CD8+ T cells, mice were injected in the peritoneal cavity with 3 μg antibody to Thy-1.1 (HIS51; BD Biosciences). Depletion was confirmed by staining with antibody to Thy-1.1 (OX-7; BioLegend) and with a tetramer of major histocompatibility complex class I H-2Db and gp33.
Parabiosis surgery.
Parabiosis surgery was done as described13,30. Each mouse of the pair was shaved along the opposite lateral flank. The skin was then wiped clean of fur with alcohol prep pads and was further cleaned with Betadine solution and 70% alcohol. A identical incision was made on the lateral aspect of each mouse, and 5-0 Vicryl suture was used to sew the skin to conjoin the mice. Additional sutures were placed through the olecranon and knee joints to secure the legs. Conjoined mice were then allowed to rest for 14–16 d before experiments.
Immunofluorescence microscopy and flow cytometry.
For immunofluorescence microscopy, tissues were frozen in 2-methylbutane surrounded by dry ice. Frozen blocks were cut into sections 7 μm in thickness and fixed in acetone; nonspecific binding was blocked for 1 h with a solution of 5% bovine serum albumin in PBS, followed by staining with DAPI (4,6-diamidino-2-phenylindole; Invitrogen) and anti-Thy-1.1 (OX-7), anti-CD31 (390) and anti-CD8β (YTS156.7.7; all three from BioLegend); anti-IFN-γ (XMG1.2) and anti-CD45.1 (A20; both from eBioscience); goat polyclonal anti-CXCL9 (AF-492-NA) and goat polyclonal anti-CXCL11 (AF572; both from R&D Systems); and rabbit polyclonal anti-CX3CL1 (NBP1-49539; Novus Biologicals). Staining of IFN-γ–phycoerythrin was amplified with rabbit anti-phycoerythrin (NB120-7011; Novus Biologicals). Jackson Immunoresearch secondary antibodies conjugated to various fluorochromes were used for staining of unconjugated antibodies: goat antibodies were amplified with Alexa Fluor 647–conjugated bovine anti-goat (805-605-180), and rabbit antibodies were amplified with indocarbocyanine-conjugated donkey anti-rabbit (711-165-152). 'Tiled' images were acquired with an automated Leica DM5500B microscope and analysis of coronal sections was done with ImageJ software and Adobe Photoshop. Cell isolation and flow cytometry were done as described30. Female reproductive tracts, including the vagina, cervix, uterine horns and ovaries, were excised and chopped into small pieces. Tissue pieces were then digested in 500 mg/l of collagenase IV (Sigma) with stirring at 300 r.p.m. for 1 h at 37 °C with a magnetic stir bar 1 inch in length. Tissues were then homogenized with a gentleMACS dissociator (Miltenyi) and filtered. For staining of chemokines by flow cytometry, cells were stained with anti-CD45.2 (104; eBioscience), anti-CD11c (N418; eBioscience), anti-CD3ɛ (145-2C11; eBioscience) and anti-Thy-1.1 (HIS51; BD). Chemokines were stained as described31.
Quantification of microscopy images.
Individual images of staining for Thy-1.1 or with DAPI were loaded into ImageJ64 software as JPEG files. Color images were then converted into binary format, which transforms images into black pixels (either DAPI or Thy-1.1) and white pixels. Black pixels were then quantified with the 'analyze particle' function. Computer counting was confirmed by periodic manual counting. Analysis of fluorescence intensity was done on grayscale TIFF files. Images of staining of CD31 and chemokines were loaded into stacks. Polygonal 'gates' were placed around CD31+ vessels and then those gates were applied to the matching images of chemokine staining. Mean gray values were then measured with the 'measure' function.
Statistical analysis.
P values were determined by a two-tailed, unpaired Student's t-test. Differences between groups were considered significant for P values of 0.05 or less.
Change history
08 April 2013
In the version of this article initially published, a label in Figure 2b and the scale size for Figure 5a were incorrect. The left label under the left image in Figure 2b should be CXCL9 (in red); the legend for Figure 5a should end "Scale bars, 20 μm." The errors have been corrected in the HTML and PDF versions of this article.
References
Sallusto, F., Geginat, J. & Lanzavecchia, A. Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu. Rev. Immunol. 22, 745–763 (2004).
von Andrian, U.H. & Mackay, C.R. T-cell function and migration. Two sides of the same coin. N. Engl. J. Med. 343, 1020–1034 (2000).
Seder, R.A. & Ahmed, R. Similarities and differences in CD4+ and CD8+ effector and memory T cell generation. Nat. Immunol. 4, 835–842 (2003).
Obar, J.J., Khanna, K.M. & Lefrançois, L. Endogenous naive CD8+ T cell precursor frequency regulates primary and memory responses to infection. Immunity 28, 859–869 (2008).
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006).
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Wherry, E.J. et al. Lineage relationship and protective immunity of memory CD8 T cell subsets. Nat. Immunol. 4, 225–234 (2003).
Masopust, D., Vezys, V., Marzo, A.L. & Lefrançois, L. Preferential localization of effector memory cells in nonlymphoid tissue. Science 291, 2413–2417 (2001).
Gebhardt, T. et al. Memory T cells in nonlymphoid tissue that provide enhanced local immunity during infection with herpes simplex virus. Nat. Immunol. 10, 524–530 (2009).
Wakim, L.M., Woodward-Davis, A. & Bevan, M.J. Memory T cells persisting within the brain after local infection show functional adaptations to their tissue of residence. Proc. Natl. Acad. Sci. USA 107, 17872–17879 (2010).
Hofmann, M. & Pircher, H. E-cadherin promotes accumulation of a unique memory CD8 T-cell population in murine salivary glands. Proc. Natl. Acad. Sci. USA 108, 16741–16746 (2011).
Masopust, D. et al. Dynamic T cell migration program provides resident memory within intestinal epithelium. J. Exp. Med. 207, 553–564 (2010).
Jiang, X. et al. Skin infection generates non-migratory memory CD8+ TRM cells providing global skin immunity. Nature 483, 227–231 (2012).
Gebhardt, T., Mueller, S.N., Heath, W.R. & Carbone, F.R. Peripheral tissue surveillance and residency by memory T cells. Trends Immunol. 34, 27–32 (2013).
Masopust, D. & Picker, L.J. Hidden memories: frontline memory T cells and early pathogen interception. J. Immunol. 188, 5811–5817 (2012).
Teijaro, J.R. et al. Cutting edge: tissue-retentive lung memory CD4 T cells mediate optimal protection to respiratory virus infection. J. Immunol. 187, 5510–5514 (2011).
Cauley, L.S. & Lefrançois, L. Guarding the perimeter: protection of the mucosa by tissue-resident memory T cells. Mucosal Immunol. 6, 14–23 (2013).
Shin, H. & Iwasaki, A. A vaccine strategy that protects against genital herpes by establishing local memory T cells. Nature 491, 463–467 (2012).
Wakim, L.M., Waithman, J., van Rooijen, N., Heath, W.R. & Carbone, F.R. Dendritic cell-induced memory T cell activation in nonlymphoid tissues. Science 319, 198–202 (2008).
Ariotti, S. et al. Tissue-resident memory CD8+ T cells continuously patrol skin epithelia to quickly recognize local antigen. Proc. Natl. Acad. Sci. USA 109, 19739–19744 (2012).
Bevan, M.J. Memory T cells as an occupying force. Eur. J. Immunol. 41, 1192–1195 (2011).
Mackay, L.K. et al. Long-lived epithelial immunity by tissue-resident memory T (TRM) cells in the absence of persisting local antigen presentation. Proc. Natl. Acad. Sci. USA 109, 7037–7042 (2012).
Collins, M.K., Tay, C.-S. & Erlebacher, A. Dendritic cell entrapment within the pregnant uterus inhibits immune surveillance of the maternal/fetal interface in mice. J. Clin. Invest. 119, 2062–2073 (2009).
Bromley, S.K., Mempel, T.R. & Luster, A.D. Orchestrating the orchestrators: chemokines in control of T cell traffic. Nat. Immunol. 9, 970–980 (2008).
Nakanishi, Y., Lu, B., Gerard, C. & Iwasaki, A. CD8+ T lymphocyte mobilization to virus-infected tissue requires CD4+ T-cell help. Nature 462, 510–513 (2009).
Luster, A.D. The role of chemokines in linking innate and adaptive immunity. Curr. Opin. Immunol. 14, 129–135 (2002).
Wakim, L.M., Gebhardt, T., Heath, W.R. & Carbone, F.R. Cutting edge: local recall responses by memory T cells newly recruited to peripheral nonlymphoid tissues. J. Immunol. 181, 5837–5841 (2008).
Çuburu, N. et al. Intravaginal immunization with HPV vectors induces tissue-resident CD8+ T cell responses. J. Clin. Invest. 122, 4606–4620 (2012).
Strutt, T.M. et al. Memory CD4+ T cells induce innate responses independently of pathogen. Nat. Med. 16, 558–564 (2010).
Casey, K.A. et al. Antigen-independent differentiation and maintenance of effector-like resident memory T cells in tissues. J. Immunol. 188, 4866–4875 (2012).
Eberlein, J. et al. Comprehensive assessment of chemokine expression profiles by flow cytometry. J. Clin. Invest. 120, 907–923 (2010).
Acknowledgements
We thank M. Mescher (University of Minnesota) for IFN-γ-deficient OT-I mice, and S. Jameson for discussions. Supported by the US National Institutes of Health (R01AI084913-01 to D.M., DP2OD006467 (by the Office of The Director) to D.M., and T32AI007313 to J.M.S.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
J.M.S., V.V. and D.M. designed the experiments; J.M.S. and K.A.F. did the experiments; and J.M.S. and D.M. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 1048 kb)
Rights and permissions
About this article
Cite this article
Schenkel, J., Fraser, K., Vezys, V. et al. Sensing and alarm function of resident memory CD8+ T cells. Nat Immunol 14, 509–513 (2013). https://doi.org/10.1038/ni.2568
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2568
This article is cited by
-
Prevention of respiratory virus transmission by resident memory CD8+ T cells
Nature (2024)
-
Unique properties of tissue-resident memory T cells in the lungs: implications for COVID-19 and other respiratory diseases
Nature Reviews Immunology (2023)
-
IKK2/NFkB signaling controls lung resident CD8+ T cell memory during influenza infection
Nature Communications (2023)
-
Local γδ T cells: translating promise to practice in cancer immunotherapy
British Journal of Cancer (2023)
-
Tumor-specific memory CD8+ T cells are strictly resident in draining lymph nodes during tumorigenesis
Cellular & Molecular Immunology (2023)