Abstract
Background
Physical activity (PA) is essential for children throughout their growth and maturation. It improves physiological and psychological health and limits the risk of developing metabolic disorders. However, some chronic physiological and metabolic diseases may lead to decreased PA. The diversity of outcomes in the literature offers no consensus for physical activity and sedentary levels in children with juvenile idiopathic arthritis (JIA) or inflammatory bowel disease (IBD).
Methods
A literature review and a meta-analysis were carried out with original studies from a Medline database search. Only high-quality studies (STROBE checklist) written in English comparing PA level or sedentary behavior (SB) between children with the disorders and their healthy peers were considered. The aim was to examine PA and SB in children with JIA or IBD compared to their healthy peers.
Results
The literature review and meta-analysis identified decreased PA and increased time spent in SB in these populations, which may exacerbate both their lower physical fitness and the symptoms of their health disorders.
Conclusion
Results nevertheless show discrepancies due to the different materials and methods used and the variables measured. Further studies are needed to establish a gold standard method for assessing PA level in these populations.
Similar content being viewed by others
Introduction
Juvenile idiopathic arthritis (JIA) and inflammatory bowel disease (IBD) such as Crohn’s disease (CD) or ulcerative colitis (UC) are among the most common immune-mediated inflammatory diseases affecting the pediatric population. JIA encompasses seven rheumatoid diseases that appear before the age of 16 years, characterized by an overproduction of inflammatory cytokines at the joint level, leading to a chronic inflammatory state and regular painful crises. The prevalence of JIA was 44.7 in 100,000 children in the USA in 20091 and 32.6 in 100,000 children in Europe in 2010.2 IBD is characterized by chronic inflammation located in the gastrointestinal tract, leading to painful injury (obstructions, fistulae, abscesses, etc.). Epidemiological data show that juvenile onset of IBD occurs in 25% of patients worldwide, with 18% of cases appearing before age 10 years.3,4 Epidemiological data also point to a continual increase in the incidence of IBD in young people.5,6
JIA and pediatric IBD profoundly affect the physical, mental, social, and functional aspects of life, resulting in drastically decreased quality of life and physical fitness, and a negative impact on growth.7,8,9 With the onset of the disease, children and adolescents are faced with a challenging new way of living, often with a cluster of disabling symptoms referred to as “sickness behavior,” including pain, fatigue, and disturbed sleep. For instance, the study of Rashid et al. found that 50.2% of children with JIA reported consistently low pain, and 17.9% reported consistently high pain.10 It has also been shown that, in young people with IBD, recurrent bouts of spasmodic abdominal pain and bloody diarrhea are very common.11,12 Fatigue is also widespread in cases of both JIA and IBD. For instance, almost 75% of children with JIA complain of fatigue,13 with one third of them describing moderate to severe levels,14,15 and adolescents with IBD exhibit more fatigue than their healthy counterparts.16 To cope with these challenging pathological conditions, some healthcare practitioners, family, and children themselves have tended to preclude physical activity (PA), fearing that it will worsen the symptoms of pain and fatigue.
PA is defined as any bodily movements produced by the skeletal muscle that result in energy expenditure.17 The beneficial effect of PA in subjects with IBD and JIA has been praised for the potential regulatory effect of exercise on the balance between pro- and anti-inflammatory responses.18,19 However, differences among studies in design and sample composition, disease activity, treatments used, and methods of assessment (objective or subjective) lead to divergent results on PA levels in children with these diseases.
We analyzed PA level in children and adolescents with JIA or IBD, paying special attention to methodological weaknesses that should be avoided in future studies in a review and meta-analysis. Our meta-analysis examined the variations in measurement methods. We hypothesized that children with JIA or IBD would be less active and more sedentary than their healthy peers.
Materials and methods
Inclusion and exclusion criteria
Participants
Participants included in the studies were children or teenagers diagnosed with JIA or IBD and healthy age- and sex-matched children. All participants were aged <18 years. Studies assessing adults or elderly people were excluded from the meta-analysis.
Type of studies and outcome measurement
Case–control, cross-sectional, and cohort studies that analyzed the difference in PA level and sedentary behaviors between children with JIA or IBD and healthy peers between January 1995 and September 2018 were included in this study.
Studies included the presentation of mean and standard deviations (SDs) of PA level and sedentary behaviors. Studies using subjective methods (diaries, questionnaires, self-reported time spent in moderate or vigorous physical activity (MVPA), or sedentary behaviors) and an objective method (accelerometry) were included. Literature reviews and meta-analysis were excluded.
The delta of mean data was calculated to make forest plots.
Search strategy to identify relevant studies
All the studies originated from MedLine (PubMed database), following the PRISMA statement (Preferred Reporting Items for Systematic Reviews and Meta-Analyses)20 (Fig. 1).
Study selection
Relevant keywords were selected according to the population (1), pathology (2), and the variables measured for PA level (3). Studies were found with combined keywords containing “children,” “child,” “teen,” or “youth” (1); “juvenile idiopathic arthritis,” “juvenile arthritis,” “inflammatory bowel disease,” “Crohn’s disease,” or “ulcerative colitis” (2); and “physical activity,” “physical activity level,” “accelerometry,” or “sedentary” (3). These were screened to select relevant studies according to their title. These in turn were screened based on their abstracts read by two independent reviewers. Finally, studies used in the selected articles were screened using the same method.
Risk of bias assessment
To avoid the risk of bias, all studies were submitted to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement.21
Data extraction
Data from the selected studies were extracted using a standardized eligibility criteria sheet. Any discrepancy at this final stage was discussed among the reviewers, with an independent reviewer being consulted if a consensus could not be reached. Authors of papers were contacted where data were incomplete or needed further clarification. Data relating to general study information (authors, year of publication, sample size), study design (cross-sectional or cohort studies), participant characteristics (age, pathology), and results (mean and SD of PA level or sedentary parameters) were collected in a table (Table 1). For the studies that included a range of values, SDs were calculated.
When values were missing, the corresponding author was asked for clarification. In the absence of a response, incomplete studies were excluded.
Statistical analysis
After extraction, the data were compiled using the software designed specifically for meta-analyses (Comprehensive Meta-Analysis, version 2; Biostat, Englewood, NJ). For descriptive analyses, data were presented as mean and SD or median and interquartile range, according to statistical distribution. The Hedges’ standardized mean differences were calculated using a random-effects model (DerSimonian and Laird approach) that accounts for true variation in effects occurring from study to study and for random errors within one study.
In other words, to address the non-independence of data due to study effects, a random-effects model was used in preference to a fixed-effect one, because certain experimental variables showed wide variation. The I2 index was used to measure heterogeneity, with 25, 50, and 75% indicating low, moderate, and high heterogeneity, respectively. Finally, an Egger test and funnel plots were used to assess publication bias. In the absence of bias, studies were distributed evenly around the mean effect size because of random sampling error. A p value of <0.05 was considered statistically significant.
Results
Identification and selection of relevant studies
Study data that met our search criteria obtained before reduction steps were carried out are listed in Table 1. Of the 15 studies presented in the literature review, only 11 were included in the meta-analysis. Data were not available for two studies, and two studies did not compare the data obtained in JIA and IBD groups with healthy controls.
For this meta-analysis, the mean score of the quality of studies (STROBE checklist) was 19.00±1.33/22. The quality of studies selected for the meta-analysis was good, and all these studies were included in the meta-analysis.
Systematic review of the PA level and sedentary behaviors of children with JIA or IBD
The level of PA was lower in sick children than in their healthy counterparts. In the studies presented in Table 1, both subjective and objective methods show a trend toward decreased PA in children with JIA or IBD. Questionnaires and diaries show that children with JIA or IBD report less time spent in PA regardless of its intensity (low, moderate, or vigorous) with a particular decrease in MVPA, together with an increased time spent in sedentary behaviors (such as watching television or using a computer).22 These results support the assertion that only 23% of the pathological pediatric population meet the public health recommendation in terms of PA (at least 1 h of MVPA per day) compared to 66% of healthy children.23 However, most of the studies presented in this review assessed PA level in children with JIA, and fewer studies assessed it in children with IBD. Despite scant studies, similar results were found for both JIA and IBD.24 Studies that assessed levels of PA relative to disease activity found that children with active disease had a lower PA level than children in remission,4,25 suggesting that the activity of the disease was the main deterrent to PA for these children. All the studies showed similar results for PA level. As regards sedentary behaviors, consensus in the data is less clear, although levels of sedentary behaviors seem to be increased in children with JIA or IBD.
Meta-analysis comparing PA level and sedentary behavior between children with JIA or IBD and healthy children
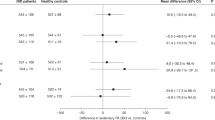
Different methods are used to assess PA level, but no gold standard exists. Although objective methods help to eliminate the methodological bias associated with patient subjectivity, they cannot provide information about the nature of the PA. The meta-analysis showed that children with JIA or IBD are less active than their healthy peers. Results obtained by both objective and subjective methods (Fig. 2) present a decreased PA level in children with JIA or IBD. However, different results were obtained by Mählmann et al.4 (studies 3ba and 3bb) and showed more time spent in vigorous PA. Results obtained by subjective methods (from study 6a to 10b) present weaker significance than those obtained by objective methods.
Results obtained by objective methods (from study 1 to 2cb) show a longer time spent in sedentary behavior in children with JIA and IBD compared to their healthy peers (Fig. 3). By contrast, sedentary behaviors present lower SDs when assessed with subjective methods (from Figs. 3 and 4b) than with objective methods. The p value is also lower for results obtained by subjective methods.
Discussion
The aim of this literature review and meta-analysis was to analyze PA level in children and adolescents with JIA and IBD compared to their healthy peers. The results yield evidence that level of PA is lower and sedentary behaviors are higher in children with JIA or IBD compared to healthy children. However, the difference between means of pathological and control groups is not constant across studies, particularly according to the assessment method (i.e., subjective or objective). The meta-analysis enabled us to isolate subjective and objective methods. Results showed that a lower PA level was observed in children with JIA or IBD only when assessed with an objective method. The distribution and potential bias of the studies will now be discussed.
Results suggest that objective methods highlight greater differences in PA level between children with JIA or IBD and their healthy peers than do subjective methods. For a similar sample size, results obtained by objective methods highlight greater differences between pathological and control groups.4,26 However, different sample sizes between studies and different SD values may explain the differences in results between studies. The heterogeneity of the results warrants discussion. The objective methods included in this meta-analysis used different models of accelerometer (Caltrac, ActiGraph, Fitbit, or SenseWear Pro), with measured variables expressed in quantitative or qualitative units, such as counts per minute (cpm), steps per day, daily energy expenditure (in kcal or MET), or time spent in different types of activity. Data has also been recorded for different periods of time (from 1 to 7 days). Studies include different numbers of subjects and sample sizes, contributing to the heterogeneity of results. Results obtained through the meta-analysis show that children with JIA or IBD are more sedentary than their healthy peers. For the assessment of sedentary behaviors, subjective methods seem to highlight larger differences, although overestimation or underestimation of children’s PA participation must be taken into account.
Our study tells us nothing about mechanisms or reasons for the lower PA level reported in children with JIA or IBD. However, it has been suggested that an impaired immune profile, with a chronic inflammatory state, leads to pain and metabolic disorders. Chronic inflammation is known to promote muscle wasting and induce dysregulations in energy supply (such as difficulties with lipid oxidation). All these consequences tend to increase fatigue and decrease exercise tolerance. These mechanisms lock children with JIA or IBD into a vicious cycle that contributes to decreased PA27 and is confirmed in our meta-analysis (Fig. 4). Inactivity is known for its negative effects on physiological and psychological health. These conditions may thus worsen symptoms and degrade physical fitness,27 resulting in an increase in comorbidities, which may in turn have a long-term negative effect.
Mechanisms and consequences of pain
Liemenis et al.28 stated that the activity of the disease is related to exacerbated pain or fatigue, which may contribute to reduced participation in PA. Fifty percent of children with rheumatic diseases suffer from musculoskeletal pain in several locations, sometimes accompanied by joint pain,29 and 30–50% of children with IBDs experience accompanying abdominal pain.30,31 However, pain is often defined as a subjective sensation that depends on age, fatigue, or type and duration of disease and that may itself be affected by the disease,32 which may explain the divergent results in the literature.
The effects of pain on children with JIA are numerous and involve the whole body. Biological factors have direct effects on pain by impairing mobility or by the action of pro-inflammatory entities on nociceptors. Unlike adults, children have a lower pain threshold, particularly due to a lack of pain experience and to the slow maturation of pain control mechanisms.33 Pain is exacerbated in children with JIA by an acute inflammation produced in the joints.34 Inflammatory entities produced in synovial joints adversely affect nervous afferent pathways and increase pain sensitivity by overstimulation. These mechanisms are similar to those induced in IBD. Musculoskeletal, joint, or abdominal pain induced by inflammation may often be increased by intense PA because of its inflammatory effect or impact. Soreness may therefore lead to avoidance of movement, initiating a deconditioning spiral that contributes to a decrease in PA level. Maggio et al.35 indicate that the fear of movement is a greater deterrent in PA than the pain itself. Armbrust et al.36 and Bohr et al.25 found a positive correlation between pain and fatigue, and Bos et al.37 found a negative correlation between pain and well-being. All these findings are correlated with low PA level and high level of sedentary behaviors, demonstrating that pain may contribute to decreased daily PA in children with health disorders.
Mechanisms and consequences of fatigue
These pathologies are characterized by high level of tumor necrosis factor (TNF)-alpha, which lead to decreased insulin sensitivity.38 These metabolic disorders will impair metabolic flexibility, leading to prompter fatigue, and thereby to reduced exercise tolerance.
Inflammatory entities produced in these disorders (such as TNF-alpha) are known to be implicated as mediators in muscle wasting.39 The decreased muscle mass may oblige children with JIA or IBD to use more energy than healthy children to generate the same strength level.40 This overwork then leads to acute neuromuscular fatigue, amplifies generalized fatigue, and finally results in decreased PA level. Mechanisms of inflammation have many negative effects on the body, affecting central nervous system activity, and resulting in stress and depression linked to fatigue.41
These considerations may explain why sick children declare a higher energy expenditure and vigorous PA when assessed with a subjective method4 (here, the International Physical Activity Questionnaire), regardless of disease status. These results may stem from lower physical fitness, which contributes to exacerbated fatigue during exercise and a consequent overestimation of the effort expended. Finally, decreased performance and depreciated self-worth, combined with an apprehension of pain (on the part of the medical and social environment, as well as the children themselves) lead to decreased participation in PA.13,42
More and more studies have been assessing the effects of different PA programs in JIA and IBD during therapies. The review by Catania et al.43 and that of Narula and Fedorak44 highlight beneficial effects of PA on symptoms of JIA and IBD, respectively. These findings underline the need to increase PA level and limit sedentary behavior in this population.
Conclusion
Children with JIA or IBD are less active than healthy children and spend more time in sedentary behaviors. Generally, symptoms induced by JIA and IBD (mainly pain) contribute to decreased exercise tolerance and degrade physical fitness, decreasing PA level in children with JIA or IBD. However, specific attention needs to be paid to the design of studies that can tend to induce bias, especially for the assessment of PA. Objective methods are more robust in highlighting differences between sick and healthy populations, but they also are very heterogeneous because of the diversity of the variables and measurements used. Studies assessing sedentary behaviors do not show such a method-related effect. It might be useful to adopt an overall approach to the assessment of PA level using both objective and subjective methods to assess the quantitative and qualitative elements of PA level. Further research is needed to establish a gold standard method for assessing PA level for these population.
References
Harrold, L. R. et al. Incidence and prevalence of juvenile idiopathic arthritis among children in a managed care population, 1996–2009. J. Rheumatol. 40, 1218–1225 (2013).
Thierry, S., Fautrel, B., Lemelle, I. & Guillemin, F. Prevalence and incidence of juvenile idiopathic arthritis: a systematic review. Jt. Bone Spine 81, 112–117 (2014).
Rosen, M. J., Dhawan, A. & Saeed, S. A. Inflammatory bowel disease in children and adolescents. JAMA Pediatr. 169, 1053–1060 (2015).
Mählmann, L. et al. Psychological wellbeing and physical activity in children and adolescents with inflammatory bowel disease compared to healthy controls. BMC Gastroenterol. 17, 160 (2017).
Benchimol, E. I. et al. Epidemiology of pediatric inflammatory bowel disease: a systematic review of international trends. Inflamm. Bowel Dis. 17, 423–439 (2011).
Saeed, S. A. & Kugathasan, S. in Pediatric Inflammatory Bowel Disease (eds Mamula, P., Markowitz, J. E. & Baldassano, R. N.) Ch. 4 (Springer, New York, 2017).
Claar, R. L. et al. Psychological distress and quality of life in pediatric crohn disease: impact of pain and disease state. J. Pediatr. Gastroenterol. Nutr. 65, 420–424 (2017).
Lundberg, V. & Eriksson, C. Health-related quality of life among Swedish children with juvenile idiopathic arthritis: parent-child discrepancies, gender differences and comparison with a European cohort. Pediatr. Rheumayol. Online J. 15, 26 (2017).
Tong, A., Jones, J., Craig, J. C. & Singh-Grewal, D. Children’s experiences of living with juvenile idiopathic arthritis: a thematic synthesis of qualitative studies. Arthritis Care Res. 64, 1392–1404 (2012).
Rashid, A. et al. Patterns of pain over time among children with juvenile idiopathic arthritis. Arch. Dis. Child 103, 437–443 (2018).
Sawczenko, A. & Sandhu, B. Presenting features of inflammatory bowel disease in Great Britain and Ireland. Arch. Dis. Child 88, 995–1000 (2003).
Szigethy, E., McLafferty, L. & Goyal, A. Inflammatory bowel disease. Child Adolesc. Psychiatr. Clin. N. Am. 19, 301–318 (2010).
Hutzal, C. E., Wright, F. V., Stephens, S., Schneiderman-Walker, J. & Feldman, B. M. A qualitative study of fitness instructors’ experiences leading an exercise program for children with juvenile idiopathic arthritis. Phys. Occup. Ther. Pediatr. 29, 409–425 (2009).
Ostlie, I. L., Aasland, A., Johansson, I., Flatö, B. & Möller, A. A longitudinal follow-up study of physical and psychosocial health in young adults with chronic childhood arthritis. Clin. Exp. Rheumatol. 27, 1039–1046 (2009).
Minden, K. et al. Long-term outcome in patients with juvenile idiopathic arthritis. Arthritis Rheumatol. 46, 2392–2401 (2002).
Marcus, S. B. et al. Fatigue and health-related quality of life in pediatric inflammatory bowel disease. Clin. Gastroenterol. Hepatol. 7, 554–561 (2009).
Caspersen, C. J., Powell, K. E. & Christenson, G. M. Physical activity, exercise, and physical fitness: definitions and distinctions for health-related research. Public Health Rep. 100, 126–131 (1985).
Bilski, J., Brzozowski, B., Mazur-Bialy, A., Sliwowski, Z. & Brzozowski, T. The role of physical exercise in inflammatory bowel disease. Biomed. Res. Int. 2014, 429031 (2014).
Rochette, E., Duché, P. & Merlin, E. Juvenile idiopathic arthritis and physical activity: possible inflammatory and immune modulation and tracks for interventions in young populations. Autoimmun. Rev. 14, 726–734 (2015).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 6, e1000097 (2009).
Elm, E. et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. PLOS Med. 4, e296 (2007).
Cavallo, S., Majnemer, A., Duffy, C. M. & Feldman, D. E. Participation in leisure activities by children and adolescents with juvenile idiopathic arthritis. J. Rheumatol. 42, 1708–1715 (2015).
Lelieveld, O. T. H. M. et al. Physical activity in adolescents with juvenile idiopathic arthritis. Arthritis Care Res. 59, 1379–1384 (2008).
Walker, R. G. et al. Sedentary time and screen-based sedentary behaviors of children with a chronic disease. Pediatr. Exerc Sci. 27, 219–225 (2015).
Bohr, A.-H., Nielsen, S., Müller, K., Karup Pedersen, F. & Andersen, L. B. Reduced physical activity in children and adolescents with juvenile idiopathic arthritis despite satisfactory control of inflammation. Pediatr. Rheumatol. Online J. 13, 57 (2015).
Henderson, C. J., Lovell, D. J., Specker, B. L. & Campaigne, B. N. Physical activity in children with juvenile rheumatoid arthritis: quantification and evaluation. Arthritis Care Res. 8, 114–119 (1995).
Gualano, B., Bonfa, E., Pereira, R. M. R. & Silva, C. A. Physical activity for paediatric rheumatic diseases: standing up against old paradigms. Nat. Rev. Rheumatol. 13, 368–379 (2017).
Limenis, E., Grosbein, H. A. & Feldman, B. M. The relationship between physical activity levels and pain in children with juvenile idiopathic arthritis. J. Rheumatol. 41, 345–351 (2014).
McGhee, J. L., Burks, F. N., Sheckels, J. L. & Jarvis, J. N. Identifying children with chronic arthritis based on chief complaints: absence of predictive value for musculoskeletal pain as an indicator of rheumatic disease in children. Pediatrics 110, 354–359 (2002).
Greenley, R. N., Kunz, J. H., Schurman, J. V. & Swanson, E. Abdominal pain and health related quality of life in pediatric inflammatory bowel disease. J. Pediatr. Psychol. 38, 63–71 (2013).
Wojtowicz, A. A., Greenley, R. N., Gumidyala, A. P., Rosen, A. & Williams, S. E. Pain severity and pain catastrophizing predict functional disability in youth with inflammatory bowel disease. J. Crohns Colitis 8, 1118–1124 (2014).
Leegaard, A., Lomholt, J. J., Thastum, M. & Herlin, T. Decreased pain threshold in juvenile idiopathic arthritis: a cross-sectional study. J. Rheumatol. 40, 1212–1217 (2013).
La Hausse de Lalouvière, L., Ioannou, Y. & Fitzgerald, M. Neural mechanisms underlying the pain of juvenile idiopathic arthritis. Nat. Rev. Rheumatol. 10, 205–211 (2014).
Cornelissen, L. et al. Pain hypersensitivity in juvenile idiopathic arthritis: a quantitative sensory testing study. Pediatr. Rheumatol. Online J. 12, 39 (2014).
Maggio, A. B. R. et al. Reduced physical activity level and cardiorespiratory fitness in children with chronic diseases. Eur. J. Pediatr. 169, 1187–1193 (2010).
Armbrust, W. et al. Fatigue in patients with juvenile idiopathic arthritis: relationship to perceived health, physical health, self-efficacy, and participation. Pediatr. Rheumatol. Online J. 14, 65 (2016).
Bos, G. J. F. J. et al. Physical activity in children with juvenile idiopathic arthritis compared to controls. Pediatr. Rheumatol. Online J. 14, 42 (2016).
Stagakis, I. et al. Anti-tumor necrosis factor therapy improves insulin resistance, beta cell function and insulin signaling in active rheumatoid arthritis patients with high insulin resistance. Arthritis Res. Ther. 14, R141 (2012).
Langen, R. C. J. et al. Tumor necrosis factor-alpha inhibits myogenic differentiation through MyoD protein destabilization. FASEB J. 18, 227–237 (2004).
Semmler, J. G. Motor unit activity after eccentric exercise and muscle damage in humans. Acta Physiol. (Oxf.) 210, 754–767 (2014).
Louati, K. & Berenbaum F. Fatigue in chronic inflammation - a link to pain pathways. Arthritis Res. Ther. 17, 254 (2015).
Race, D. L. et al. ‘It might hurt, but you have to push through the pain’: perspectives on physical activity from children with juvenile idiopathic arthritis and their parents. J. Child Health Care 20, 428–436 (2016).
Catania, H., Fortini, V. & Cimaz, R. Physical exercise and physical activity for children and adolescents with juvenile idiopathic arthritis: a literature review. Pedia. Phys. Ther. 29, 256–260 (2017).
Narula, N. & Fedorak, R. N. Exercise and inflammatory bowel disease. Can. J. Gastroenterol. 22, 497–504 (2008).
Gueddari, S. et al. Physical activity, functional ability, and disease activity in children and adolescents with juvenile idiopathic arthritis. Clin. Rheumatol. 33, 1289–1294 (2014).
Hulsegge, G. et al. Fundamental movement skills, physical fitness and physical activity among Australian children with juvenile idiopathic arthritis. J. Paediatr. Child Health 51, 425–432 (2015).
Nørgaard, M. et al. Accelerometer-assessed daily physical activity in relation to pain cognition in juvenile idiopathic arthritis. Scand. J. Rheumatol. 46, 22–26 (2017).
Tarakci, E., Yeldan, I., Kaya Mutlu, E., Baydogan, S. N. & Kasapcopur, O. The relationship between physical activity level, anxiety, depression, and functional ability in children and adolescents with juvenile idiopathic arthritis. Clin. Rheumatol. 30, 1415–1420 (2011).
Werkstetter, K. J. et al. Lean body mass, physical activity and quality of life in paediatric patients with inflammatory bowel disease and in healthy controls. J. Crohns Colitis 6, 665–673 (2012).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bourdier, P., Saidi, O., Rochette, E. et al. Physical activity and sedentary levels in children with juvenile idiopathic arthritis and inflammatory bowel disease. A systematic review and meta-analysis. Pediatr Res 86, 149–156 (2019). https://doi.org/10.1038/s41390-019-0409-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-019-0409-5
This article is cited by
-
Impaired cardiorespiratory and neuromuscular fitness in children and adolescents with juvenile idiopathic arthritis: a cross-sectional case–control study in the era of biologic drug therapies
Pediatric Rheumatology (2023)
-
Analyse von Bewegungsstörungen in der Kinder- und Jugendrheumatologie
Die Orthopädie (2023)
-
Daily physical activity patterns in children and adolescents with inflammatory bowel disease
Pediatric Research (2021)
-
Assessing the Management of Excessive Daytime Sleepiness by Napping Benefits
Sleep and Vigilance (2020)